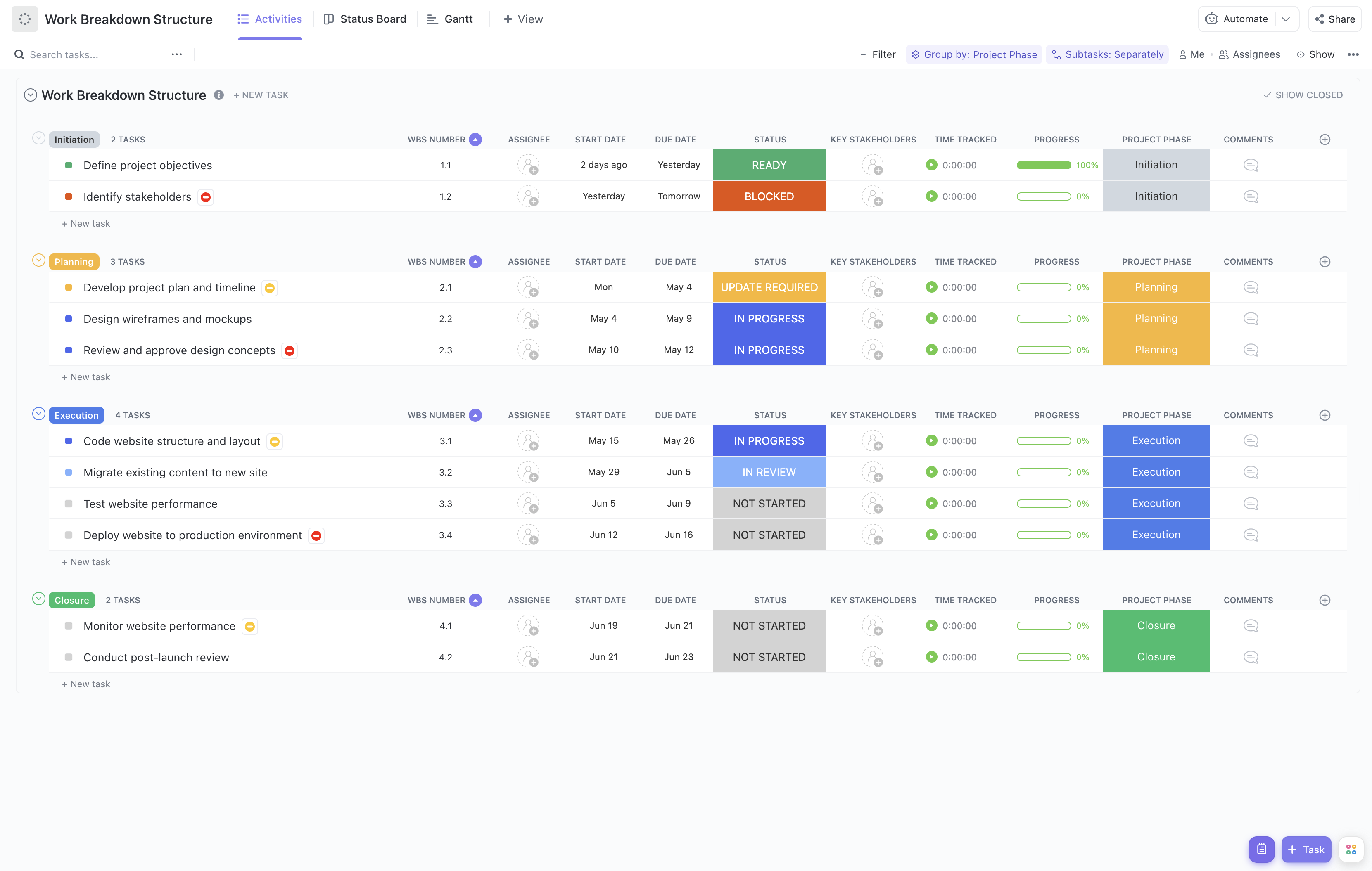
Managing regulatory submissions involves coordinating numerous detailed tasks, strict deadlines, and compliance requirements. Breaking down these complex processes into manageable components is essential for success. That's where our Regulatory Submission Work Breakdown Structure Template comes in!
This specialized template helps you:
- Organize submission activities into clear, manageable sections for enhanced oversight
- Align regulatory, clinical, quality, and legal teams to their specific responsibilities efficiently
- Track submission milestones and regulatory agency feedback with real-time updates
Best of all, no coding or specialized software skills are needed—just ClickUp to streamline your regulatory project management.
Benefits of a Regulatory Submission Work Breakdown Structure Template
Using a work breakdown structure tailored for regulatory submissions provides critical advantages for regulatory affairs professionals. It visually maps out the entire submission process, ensuring all regulatory requirements are addressed on time and within scope. Key benefits include:
- Clearly defines and organizes submission deliverables such as dossiers, validation reports, and correspondence
- Clarifies roles and responsibilities among cross-functional teams including regulatory affairs, clinical operations, and quality assurance
- Enhances communication between internal teams and external stakeholders such as regulatory agencies and consultants
- Improves overall efficiency by identifying dependencies and potential bottlenecks in the submission workflow
Main Elements of the Regulatory Submission Work Breakdown Structure Template
This template is structured to cover all critical phases of regulatory submission projects:
- Planning and Preparation: Define submission scope, identify regulatory requirements, and assemble the submission team.
- Document Compilation: Organize and prepare technical documents, clinical data, and quality reports required for submission.
- Review and Quality Control: Conduct internal reviews, cross-functional approvals, and ensure compliance with regulatory standards.
- Submission and Follow-up: Submit to regulatory agencies, track acknowledgments, respond to queries, and manage amendments.
- Project Monitoring: Use integrated dashboards and status boards to monitor progress, risks, and deadlines in real-time.
By leveraging this template, regulatory teams can maintain a clear overview of complex submission projects, ensuring all tasks are completed accurately and on schedule.