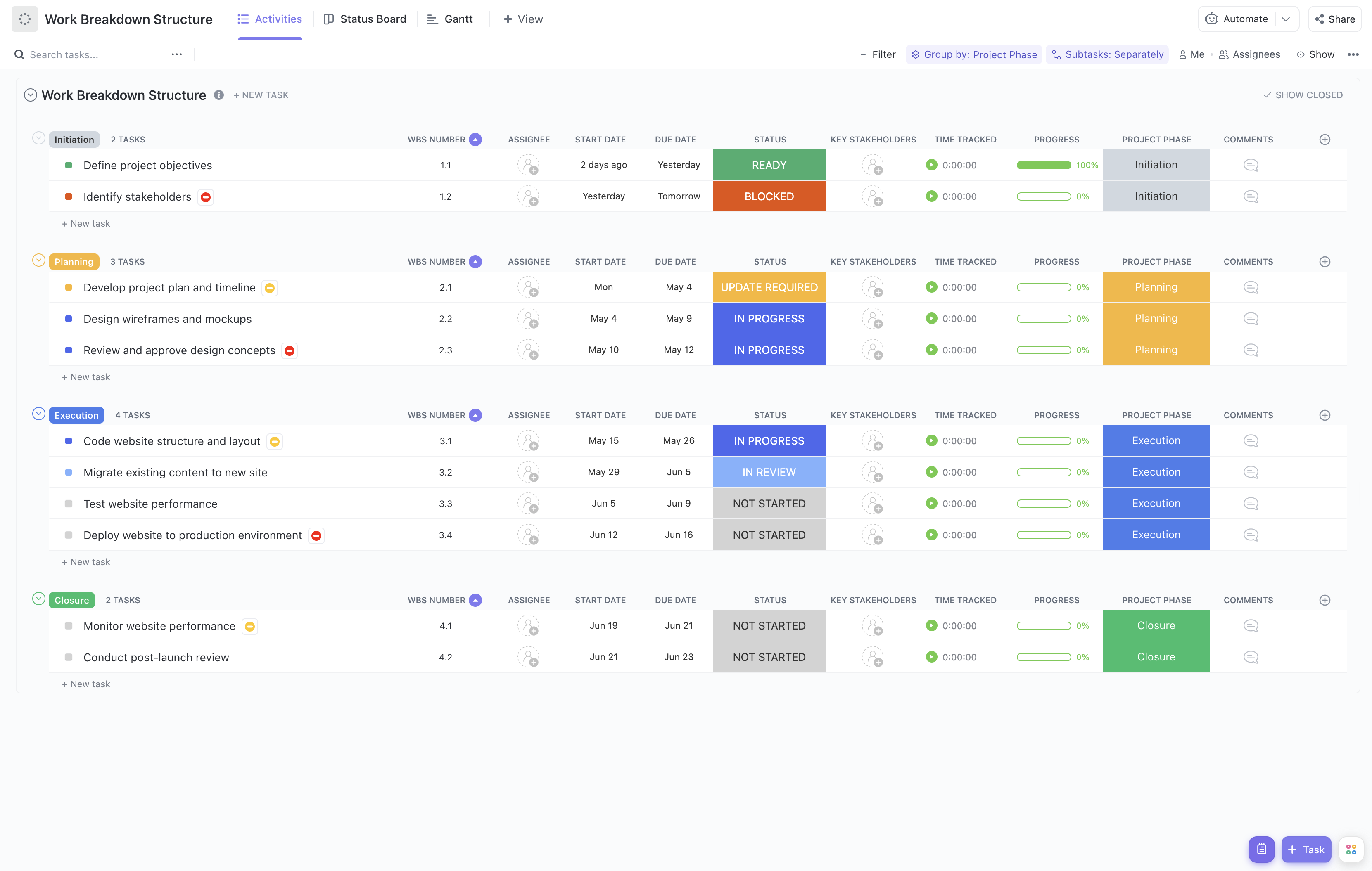
Managing a regulatory readiness program involves navigating complex compliance requirements, coordinating multiple stakeholders, and adhering to strict timelines. To streamline this process, our Regulatory Readiness Program Work Breakdown Structure Template offers a structured approach to decompose regulatory activities into actionable components.
This specialized template empowers your team to:
- Systematically organize regulatory tasks such as documentation preparation, submission planning, and audit readiness.
- Assign responsibilities to regulatory affairs specialists, quality assurance, legal, and other key stakeholders with clarity.
- Track milestones and regulatory submission progress in real-time to anticipate and mitigate risks.
Best of all, this template requires no coding or specialized software knowledge—just ClickUp and your regulatory expertise to drive compliance success.
Benefits of a Regulatory Readiness Work Breakdown Structure Template
Utilizing a WBS template tailored for regulatory readiness programs offers significant advantages, including:
- Clear definition and organization of regulatory deliverables such as dossier compilation, labeling reviews, and certification submissions.
- Enhanced visibility into team roles and responsibilities, ensuring accountability across departments involved in regulatory compliance.
- Improved communication channels between regulatory affairs, clinical teams, manufacturing, and external regulatory bodies.
- Greater efficiency in managing timelines, reducing the risk of missed deadlines or incomplete submissions.
Main Elements of the Regulatory Readiness Work Breakdown Structure Template
This template is structured to cover all critical phases of regulatory readiness:
- Regulatory Strategy Development:
Define regulatory pathways, identify applicable guidelines, and establish submission timelines.
- Documentation Management:
Break down tasks for compiling technical files, safety reports, and quality documentation.
- Submission Preparation:
Coordinate reviews, approvals, and finalization of regulatory dossiers.
- Stakeholder Engagement:
Assign and track responsibilities for internal teams and external consultants or agencies.
- Compliance Monitoring:
Set up checkpoints for audits, inspections, and post-submission follow-ups.
By leveraging this template, regulatory teams can ensure a comprehensive, transparent, and efficient approach to regulatory readiness, ultimately facilitating smoother approvals and market access.