Precision Time Tracking for Clinical Research Organisations
No credit card.
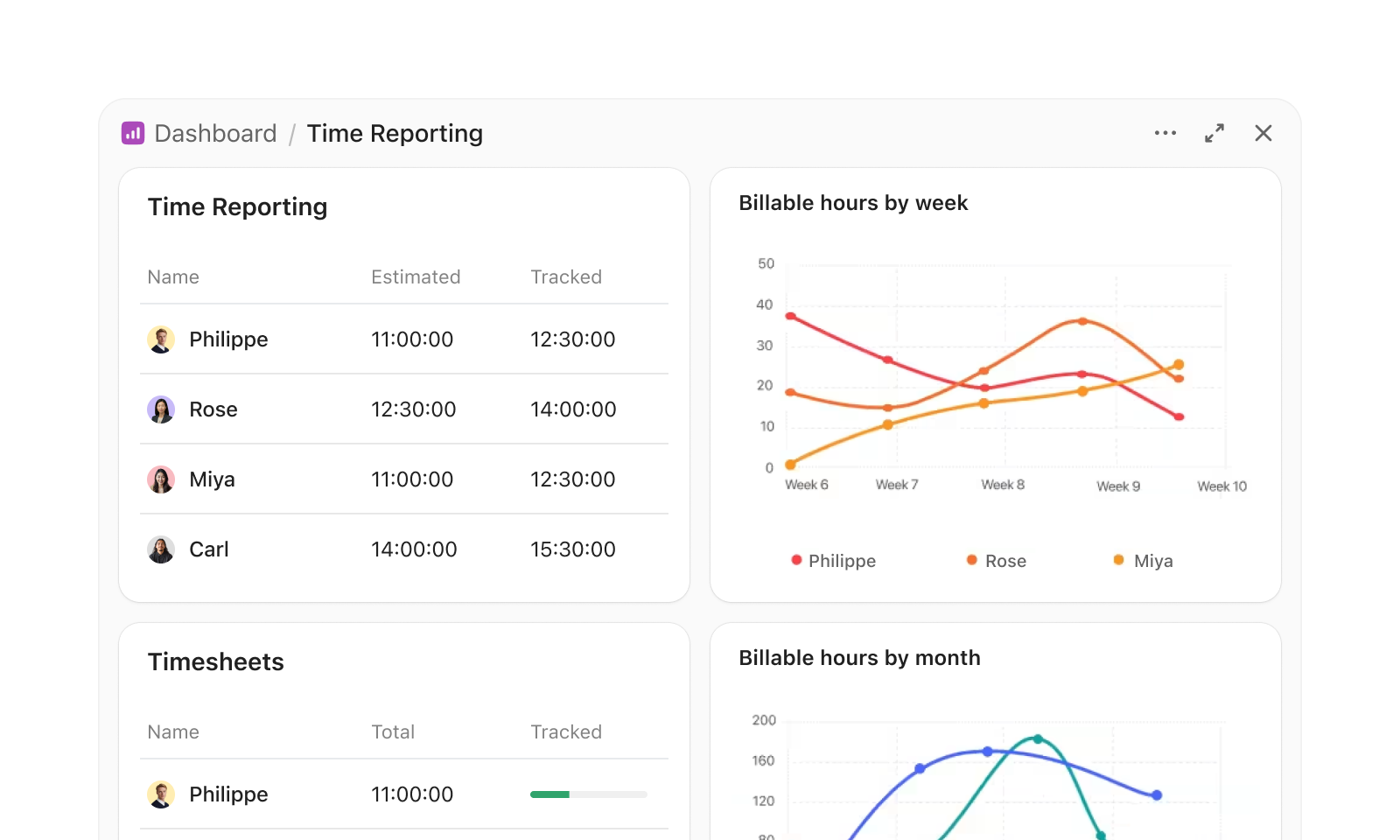
Navigating Time Tracking Challenges in Clinical Research
Without dedicated time tracking tools, clinical research teams face serious operational risks:
- Inconsistent timesheet submissions — jeopardizing study timelines and budgets
- Manual entry errors — risking data integrity and costing resources
- Lack of visibility into staff workload — increasing burnout and turnover
- Regulatory compliance gaps — exposing your organisation to audit failures
- Excessive administrative overhead — pulling focus from critical research tasks
- Disconnected time data and project management — impairing decision-making
- Difficulty verifying contract research hours — leading to payment disputes
- Unreliable labour cost reporting — complicating grant and budget management
Why Traditional Time Tracking Falls Short in Clinical Research
Traditional Approaches
- Timesheets collected via emails or spreadsheets, manually compiled
- Limited visibility into submissions until deadlines pass
- Approval workflows managed through disconnected communications with no audit trail
- Time data isolated from study tasks and milestones
- Capacity planning based on assumptions, not real data
- Compliance documentation incomplete and difficult to retrieve
ClickUp Time Tracking
- Unified platform for timesheet submission with locked entries
- Real-time insight into team hours and project allocation
- Automated approvals with reminders and full audit trails
- Seamless integration of time data with research tasks and study phases
- Workload views that reflect actual capacity versus assignments
- Exportable, tamper-proof records ready for audits and reporting
Unlock the Power of Effective Time Tracking in Clinical Research
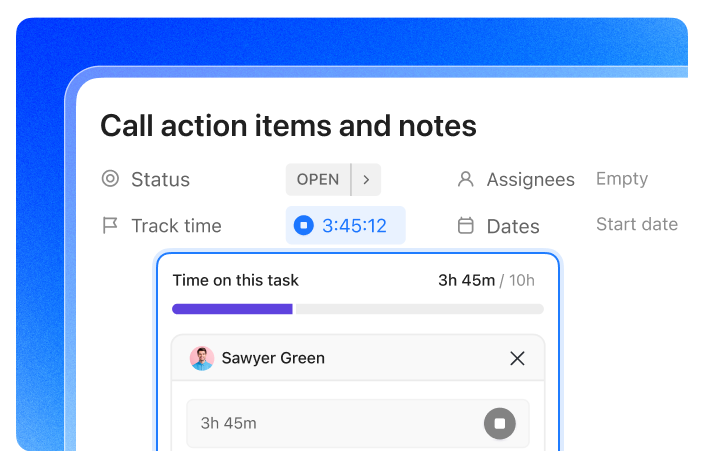
Ensure Every Research Hour Is Accounted For
Automated reminders guarantee timely timesheet submissions ahead of critical study milestones.
Deliver Audit-Ready Timesheets with Confidence
Lock entries with approvals and maintain a full audit trail to meet regulatory standards.
Detect Research Staff Overload Early
Workload insights reveal capacity imbalances so you can redistribute tasks proactively.
Simplify Compliance with Instant Exportable Records
Access comprehensive, tamper-proof logs for audits and regulatory reviews anytime.
Track Labor Costs by Project and Funding Source
Tag hours by study or grant to provide transparent, detailed financial reporting.
Minimize Administrative Time on Time Tracking
Automate follow-ups and summary reports so your team focuses on advancing research.
Begin Accurate Time Tracking for Clinical Research Today
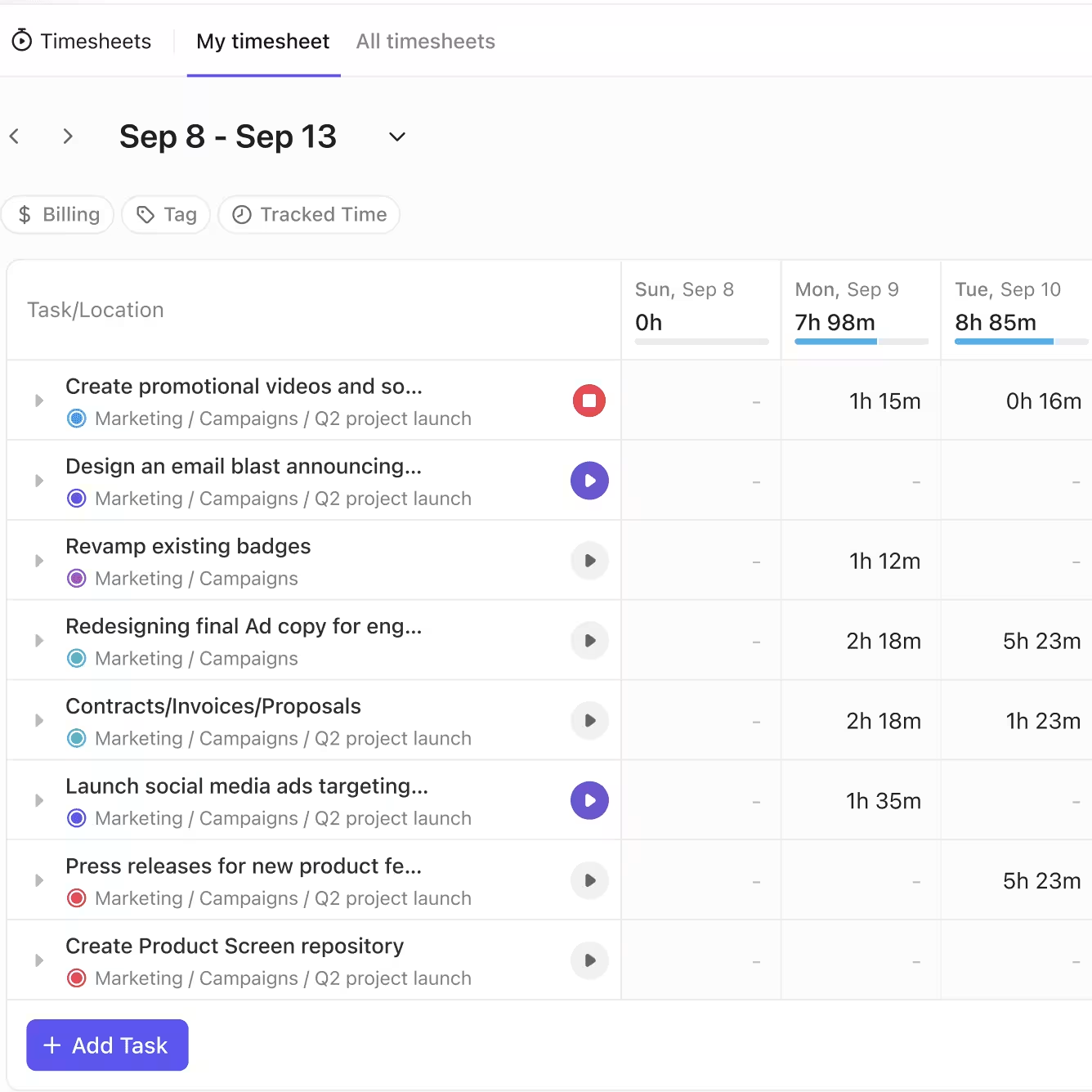
Clinical Research Teams That Gain the Most from Time Tracking Software
If You’re a Clinical Trial Manager
- End the chase for delayed timesheets. Automated reminders keep submissions on schedule
- Quickly identify incomplete or missing entries without manual tracking
- Approve hours with confidence. Locked and audited timesheets protect data integrity
- Ensure all study hours are verified before budget reconciliation
If You’re a Research Coordinator
- Monitor team workload to prevent burnout and maintain productivity
- Reassign tasks easily through integrated workload views
- Eliminate reminder emails. Brain manages follow-ups automatically
- Approve your team’s recorded hours swiftly, freeing you to focus on research coordination
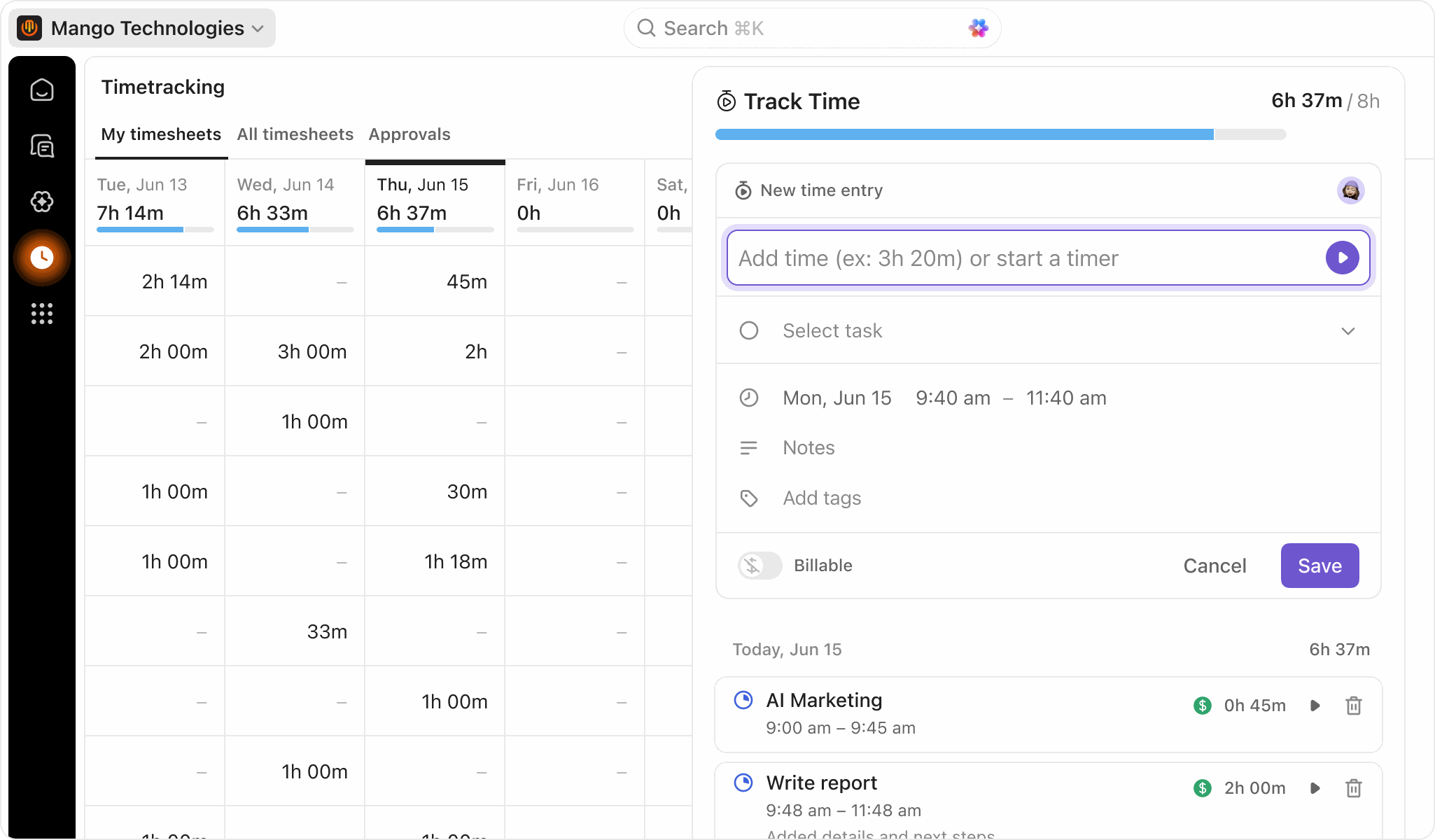
Streamlining Time Tracking With ClickUp Brain and AI
Forget Manual Timesheet Follow-Ups
Set deadlines once and Brain sends automatic reminders and flags missing submissions.
Instantly Query Time Tracking Data
Ask Brain questions like “Who hasn’t submitted this week?” or “Time spent per study?” and get answers instantly.
Arrive at Meetings Prepared
Receive AI-generated summaries of tracked hours, workload, and project progress before reviews.
Capture Hidden Research Activities
Automatically log meetings and discussions, linking them to the correct study tasks.
Automate Compliance Workflows
Brain detects overtime, triggers follow-ups, and generates compliance reports without manual input.
Identify Issues Before They Impact Studies
Brain spots missing timesheets and unusual time patterns early, reducing risk to research timelines.
FAQs on Time Tracking for Clinical Research Organisations
Stop chasing timesheets. Start with ClickUp today.
