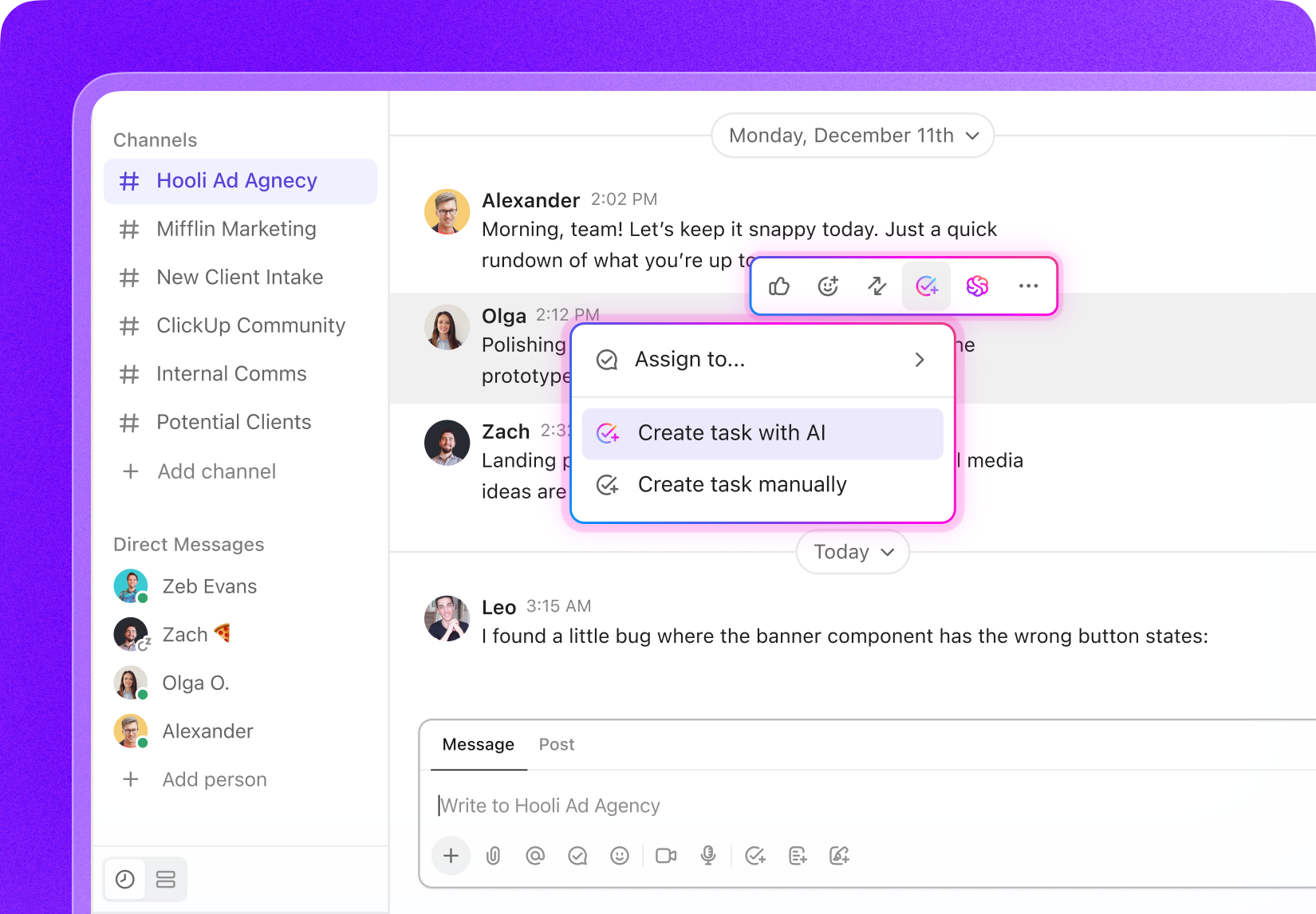
Task Solution for Validation Engineers
Task Management Software Tailored for Validation Engineers
Organize your validation processes, monitor critical checkpoints, collaborate effortlessly with your team, and gain full transparency across every phase of your projects.
Trusted by the best
The Challenge of Managing Validation Tasks
Why Validation Engineers Need Specialized Task Management Tools
Without an effective system, validation tasks become fragmented, deadlines slip, and compliance risks increase, turning complex projects into overwhelming challenges.
- Complex protocols risk inconsistencies — making it difficult to ensure repeatable and compliant validation steps.
- Documentation piles up quickly — scattered records lead to audit failures and lost traceability.
- Coordination gaps occur — multiple teams and stakeholders can cause miscommunication and delays.
- Regulatory deadlines loom — missing submission dates jeopardizes product launches and certifications.
- Progress tracking is unclear — without real-time updates, project status becomes guesswork.
- Change management is tough — tracking updates and approvals manually leads to errors.
- Resource allocation becomes chaotic — equipment and personnel scheduling conflicts slow validation cycles.
- Risk of non-compliance rises — scattered information compromises audit readiness and quality standards.
Conventional Approaches vs ClickUp Solutions
Why Traditional Validation Methods Fall Short
Discover how ClickUp streamlines validation workflows where standard tools struggle.
Traditional Validation Practices
- Tasks managed by spreadsheets and emails
- Protocols stored in disconnected folders
- Manual tracking prone to errors
- Collaboration via scattered communication channels
- Deadlines managed by memory or informal reminders
- Documentation scattered across systems
ClickUp Task Management for Validation Engineers
- Centralized task lists with clear priorities and statuses
- Structured protocol templates and checklists
- Automated tracking and audit trails
- Real-time collaboration and notifications
- Integrated deadline reminders and calendar syncing
- Unified document storage linked to tasks
Use Cases
Unlocking Efficiency: How Task Management Software Empowers Validation Engineers
See how focused task tracking reduces errors and streamlines regulatory compliance.
#UseCase1
Centralizing Validation Protocols and Results
Keep all validation procedures, test results, and related documents organized in one accessible workspace, ensuring nothing is overlooked.
#UseCase2
Maintaining Complete Audit Trails for Compliance
Track every step, change, and approval with timestamped comments and version history to meet regulatory requirements effortlessly.
#UseCase3
Managing Cross-Functional Team Communication
Coordinate between engineering, quality assurance, and production teams with clear task ownership and real-time updates.
#UseCase4
Preventing Validation Protocol Deviations
Use templates and checklists to enforce standard procedures, reducing protocol drift and ensuring consistent execution.
#UseCase5
Tracking Regulatory Submission Deadlines
Automate reminders for FDA, ISO, and other important submission timelines to avoid costly delays or penalties.
#UseCase6
Streamlining Equipment Qualification Processes
Organize installation, operational, and performance qualification tasks with dependencies and status tracking.
#UseCase7
Reducing Rework through Early Issue Identification
Identify bottlenecks or discrepancies early with dashboards and reports, allowing prompt corrective actions.
#UseCase8
Integrating Change Control and Validation Activities
Link change requests to validation tasks to ensure all modifications undergo proper review and approval.
#UseCase9
Simplifying Training and Competency Tracking
Assign and monitor training tasks for validation team members to maintain compliance with industry standards.
Key Beneficiaries
Who Gains the Most from ClickUp’s Validation Software?
For validation engineers aiming for precision and compliance in every project.
If you’re a Validation Engineer in Pharmaceuticals
ClickUp helps you manage complex drug validation protocols, ensuring regulatory standards and audit readiness without data loss.
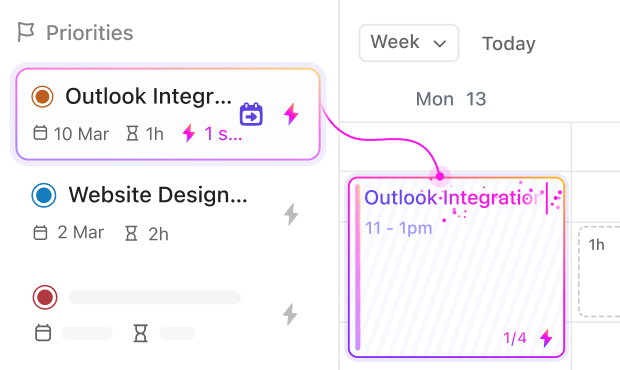
If you work in Medical Device Validation
Standardize device qualification processes, track risk assessments, and coordinate cross-department reviews seamlessly.
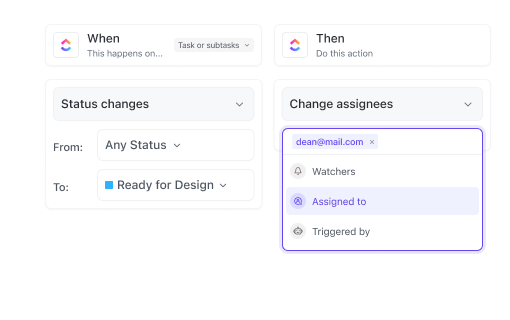
If you’re part of a Validation Team in Manufacturing
Coordinate equipment and process validations, assign corrective actions, and maintain traceability across shifts and locations.
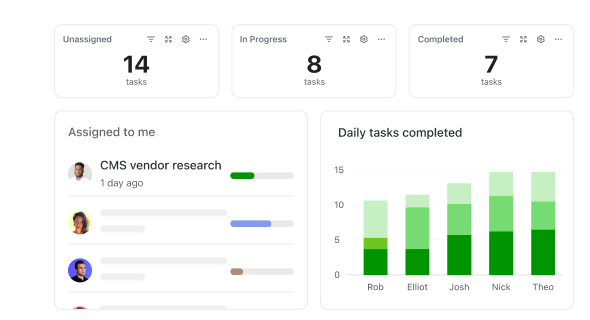
How ClickUp Supports Validation Engineers
Optimize Every Phase of Your Validation Process
Manage protocols, approvals, and documentation without juggling multiple systems.
Centralize Everything
Store literature, datasets, protocols, drafts, and grant docs in one workspace — no more scattered files.
Plan Research in Phases
Break projects into proposal, literature review, experiments, analysis, and writing with task lists and Gantt timelines.
Standardize Experiments & Fieldwork
Use templates and checklists for repeatable, error-free lab or field procedures.
Collaborate Across Teams
Assign tasks to co-authors, lab members, or collaborators. Shared boards and dashboards keep everyone aligned.
Turn Meetings Into Actionable Tasks
Convert supervisor or lab meetings into tasks with owners, checklists, and deadlines.
Stay on Top of Deadlines & Funding
Track grants, conferences, and submissions with automated reminders and calendars.