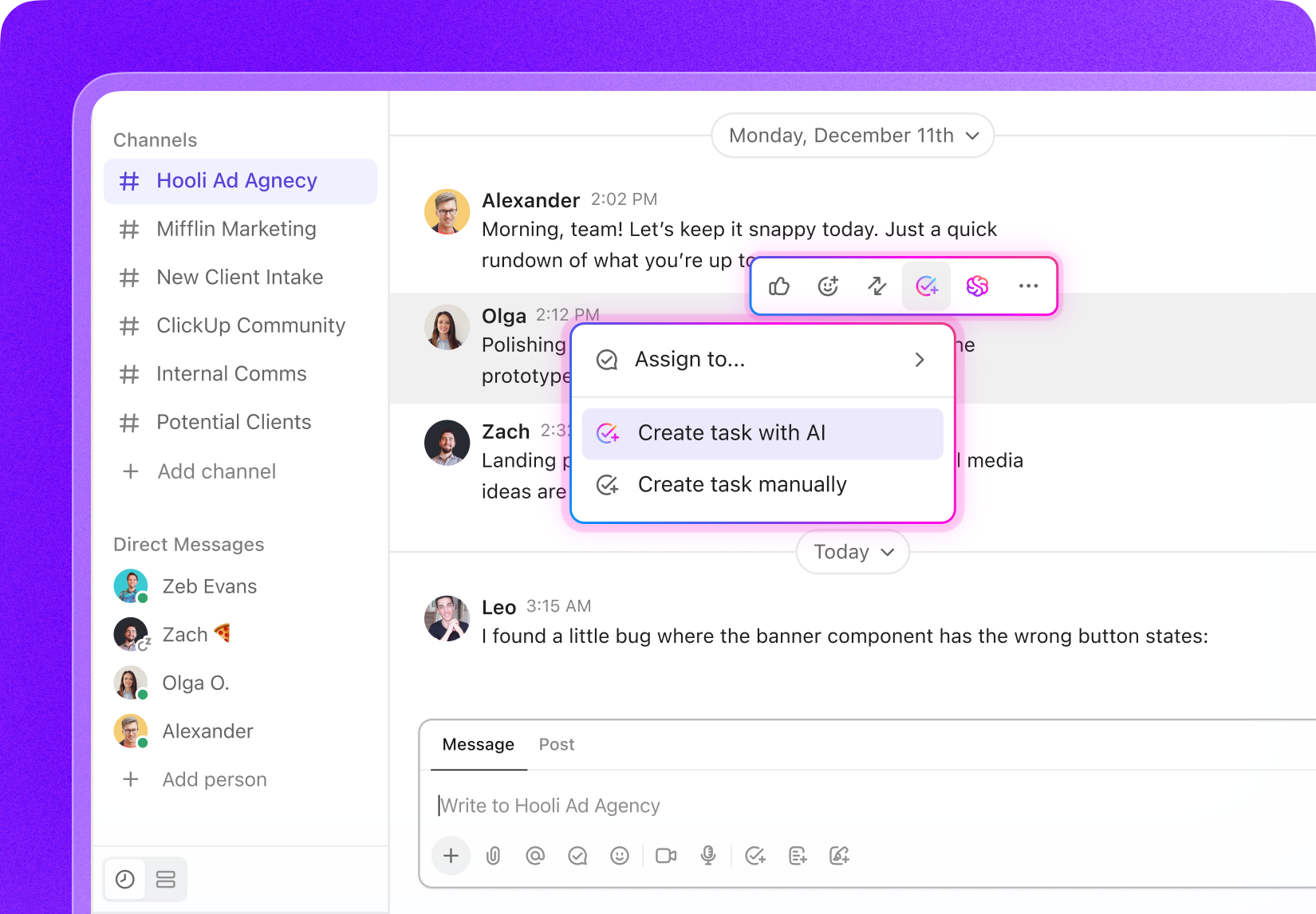
Task Management Software Tailored for Site Visit Documentation
Why You Need Specialized Task Management for Site Visit Documentation
Relying on outdated methods for site visit documentation can cause missed details, scattered records, and growing confusion — turning routine visits into logistical headaches.
- Untracked inspections cause compliance gaps — missing critical observations or follow-up actions.
- Paper reports get lost or damaged — making audit trails incomplete.
- Inconsistent data entry risks errors — jeopardizing report accuracy.
- Communication breakdowns slow resolutions — delays between field teams and office staff.
- Deadlines for corrective actions slip — risking penalties or project delays.
- Progress remains unclear — making it hard to assess site status at a glance.
- Resource scheduling gets complicated — overlapping site visits or unavailable personnel.
- Data fragmentation across devices — causing version conflicts and lost notes.
Why Legacy Site Visit Tools Fall Short
Conventional Processes
- Manual notes scattered across paper and emails
- Lost or incomplete inspection reports
- Error-prone data entry with inconsistent formats
- Communication delays between field and office
- Missed deadlines for compliance and follow-ups
- Files and photos stored in separate locations
ClickUp Task Management
- Centralized tasks with clear statuses and priorities
- Digital forms and checklists standardize inspections
- Real-time collaboration with instant updates
- Automated reminders for deadlines and follow-ups
- Integrated media attachments linked to tasks
- Searchable, organized documentation accessible anywhere
How Task Management Software Enhances Site Visit Documentation
Consolidating Inspection Data Across Devices & Teams
Maintaining Clear Compliance Records and Audit Trails
Capturing Real-Time Feedback from Field Teams
Preventing Oversight in Multi-Phase Site Inspections
Tracking Permits, Safety Approvals & Compliance Deadlines
Coordinating Resource Allocation and Site Visit Schedules
Meeting Regulatory Reporting Requirements on Time
Reducing Redundant Data Collection and Entry
Transforming Site Visit Notes into Clear Action Plans
Who Benefits Most from ClickUp for Site Visit Documentation?
If you’re a Field Inspector
ClickUp helps you capture detailed observations, assign follow-ups, and manage deadlines without losing critical information in transit.
If you’re a Project Manager
Coordinate site visits, track compliance status, and allocate resources efficiently to keep projects on schedule.
If you’re a Compliance Officer
Monitor inspection results, manage regulatory deadlines, and maintain audit-ready documentation with ease.
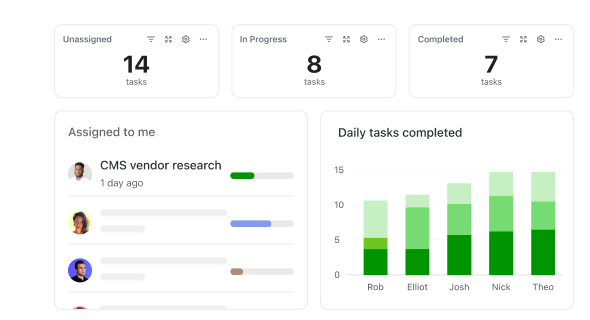
How ClickUp Simplifies Every Aspect of Site Visit Documentation
Manage inspections, compliance, and reporting—all in one centralized platform.
Centralize Everything
Plan Research in Phases
Break projects into proposal, literature review, experiments, analysis, and writing with task lists and Gantt timelines.
Standardize Experiments & Fieldwork
Collaborate Across Teams
Assign tasks to co-authors, lab members, or collaborators. Shared boards and dashboards keep everyone aligned.