Task Management for Pharmaceutical Innovators
Task Management Software Tailored for Pharmaceutical Teams
Unify your workflows, monitor clinical milestones, collaborate effortlessly, and gain full transparency across drug development stages.
Trusted by the best
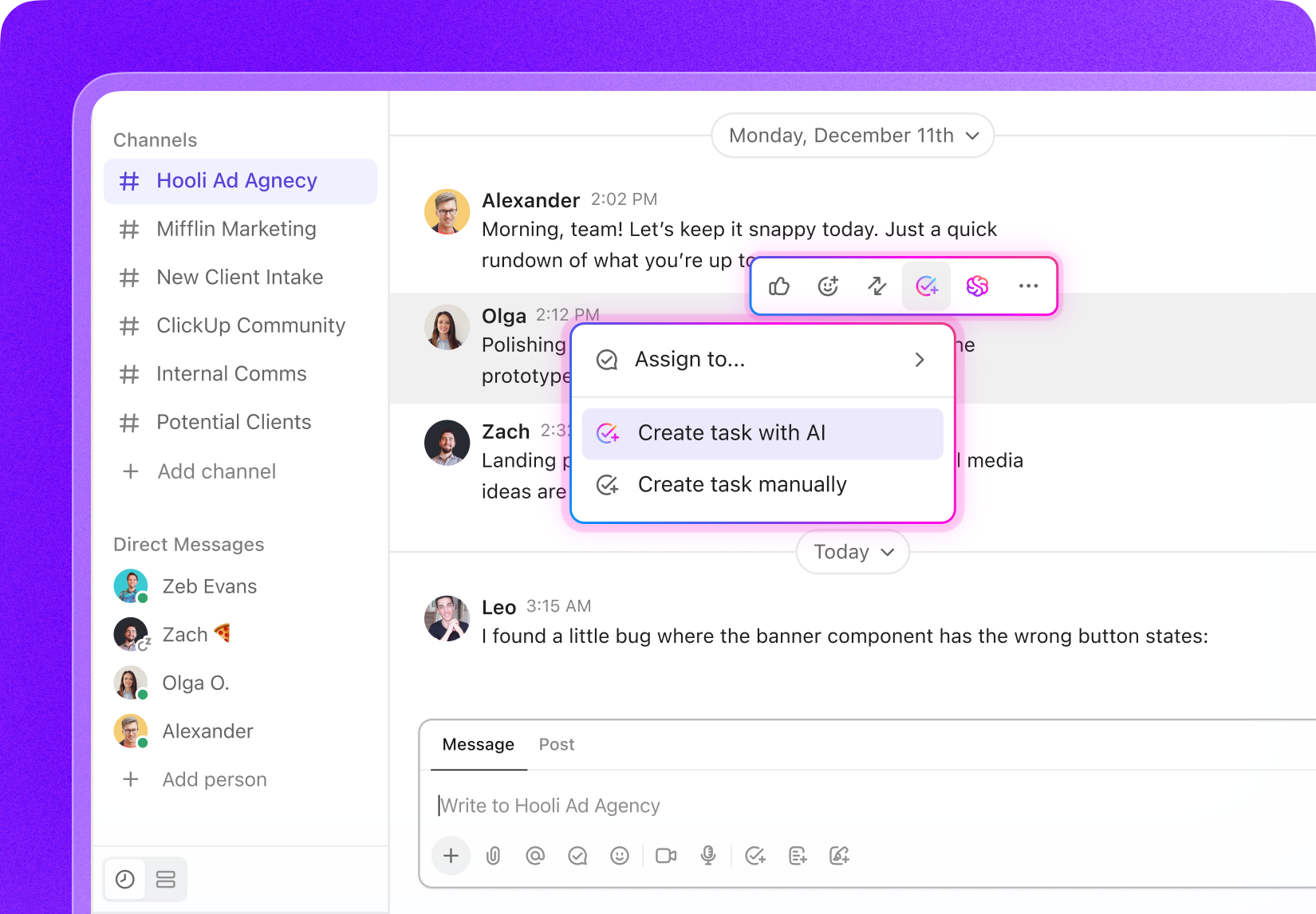
Managing Complex Projects
Why Pharmaceutical Companies Require Specialized Task Management
Relying on outdated tools causes delays, miscommunication, and compliance risks — turning critical projects into overwhelming challenges.
- Clinical trial phases become disjointed — lacking clear status updates and accountability.
- Regulatory documentation scatters — increasing risks of missing submissions or audits.
- Resource allocation gets complicated — laboratory equipment and personnel often double-booked.
- Cross-department collaboration falters — data silos and unclear responsibilities slow progress.
- Deadlines for FDA approvals and patents slip by — risking costly setbacks.
- Progress tracking feels opaque — making it hard to identify bottlenecks or successes.
- Communication breaks down — emails and spreadsheets fail to keep teams aligned.
- Quality assurance becomes inconsistent — protocols may not be followed strictly, risking compliance.
Conventional Approaches vs ClickUp
Why Traditional Systems Fall Short in Pharma
Discover how ClickUp delivers clarity and control where legacy tools cannot.
Legacy Methods
- Tasks spread across emails, spreadsheets, and disconnected tools
- Regulatory files scattered and hard to locate
- Manual tracking of clinical trial steps prone to errors
- Collaboration hindered by unclear task ownership
- Missed deadlines for submissions and approvals
- Inconsistent documentation across teams
ClickUp Task Management
- Centralized task boards with clear statuses and priorities
- Organized regulatory documents with easy retrieval
- Automated clinical trial workflows with reusable templates
- Transparent ownership and real-time collaboration
- Automated alerts for critical deadlines
- Comprehensive, searchable records linked to each task
Key Applications
Unlocking Efficiency: Task Management Use Cases for Pharma
See how streamlined task tracking reduces errors and accelerates drug development.
#UseCase1
Consolidating Protocols and Compliance Documents
ClickUp centralizes SOPs, regulatory filings, and audit trails, ensuring accessibility and reducing compliance risks.
#UseCase2
Maintaining Transparent Clinical Trial Progress
Track every trial phase with timelines, dependencies, and real-time updates to ensure adherence to protocols.
#UseCase3
Capturing Regulatory Feedback and Amendments
Manage comments, change requests, and approvals with version control and clear task assignments.
#UseCase4
Preventing Protocol Deviations in Drug Testing
Use checklists and task dependencies to maintain consistency and document every trial step comprehensively.
#UseCase5
Tracking IRB Approvals and Ethical Compliance
Organize submissions, approvals, and follow-ups with reminders to ensure no compliance deadlines are missed.
#UseCase6
Mapping Complex Manufacturing Workflows
Visualize each stage of production with custom fields and dependencies to avoid delays and quality issues.
#UseCase7
Meeting Regulatory Submission Deadlines
Centralize all submission materials and track due dates with automated alerts to avoid costly penalties.
#UseCase8
Avoiding Redundant Testing and Data Duplication
Track experiments and datasets as tasks with tags and notes to minimize duplicated efforts across teams.
#UseCase9
Turning Cross-Functional Meetings into Action Plans
Convert discussion points into assigned tasks with deadlines, ensuring clear accountability and follow-through.
Key Beneficiaries
Who Gains the Most from ClickUp in Pharma?
Ideal for pharmaceutical professionals seeking integrated task oversight across departments.
If you’re a Clinical Trial Manager
ClickUp helps you coordinate patient enrollment, monitor trial phases, and maintain compliance without juggling spreadsheets.
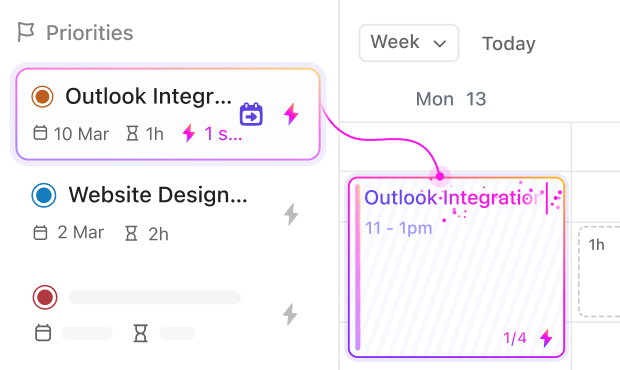
If you’re a Regulatory Affairs Specialist
Stay ahead of submission deadlines, organize documentation, and track approval statuses effortlessly.
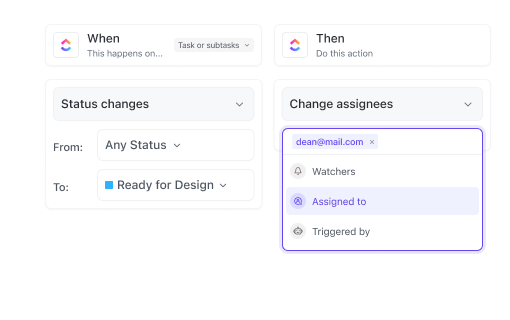
If you’re a Manufacturing Supervisor
Standardize production processes, monitor resource allocation, and ensure quality control with task templates and dashboards.
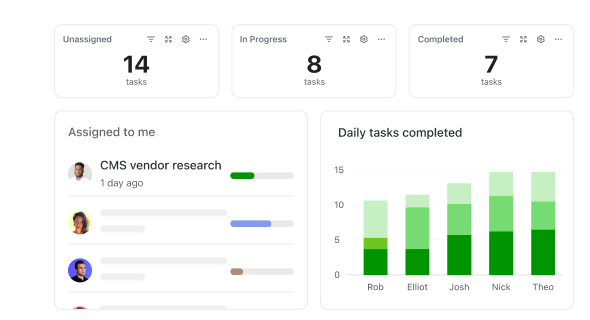
How ClickUp Supports Pharma Teams
Simplify Pharmaceutical Workflow Management
Manage drug development stages, compliance, and cross-team collaboration in one platform.
Centralize Everything
Store literature, datasets, protocols, drafts, and grant docs in one workspace — no more scattered files.
Plan Research in Phases
Break projects into proposal, literature review, experiments, analysis, and writing with task lists and Gantt timelines.
Standardize Experiments & Fieldwork
Use templates and checklists for repeatable, error-free lab or field procedures.
Collaborate Across Teams
Assign tasks to co-authors, lab members, or collaborators. Shared boards and dashboards keep everyone aligned.
Turn Meetings Into Actionable Tasks
Convert supervisor or lab meetings into tasks with owners, checklists, and deadlines.
Stay on Top of Deadlines & Funding
Track grants, conferences, and submissions with automated reminders and calendars.