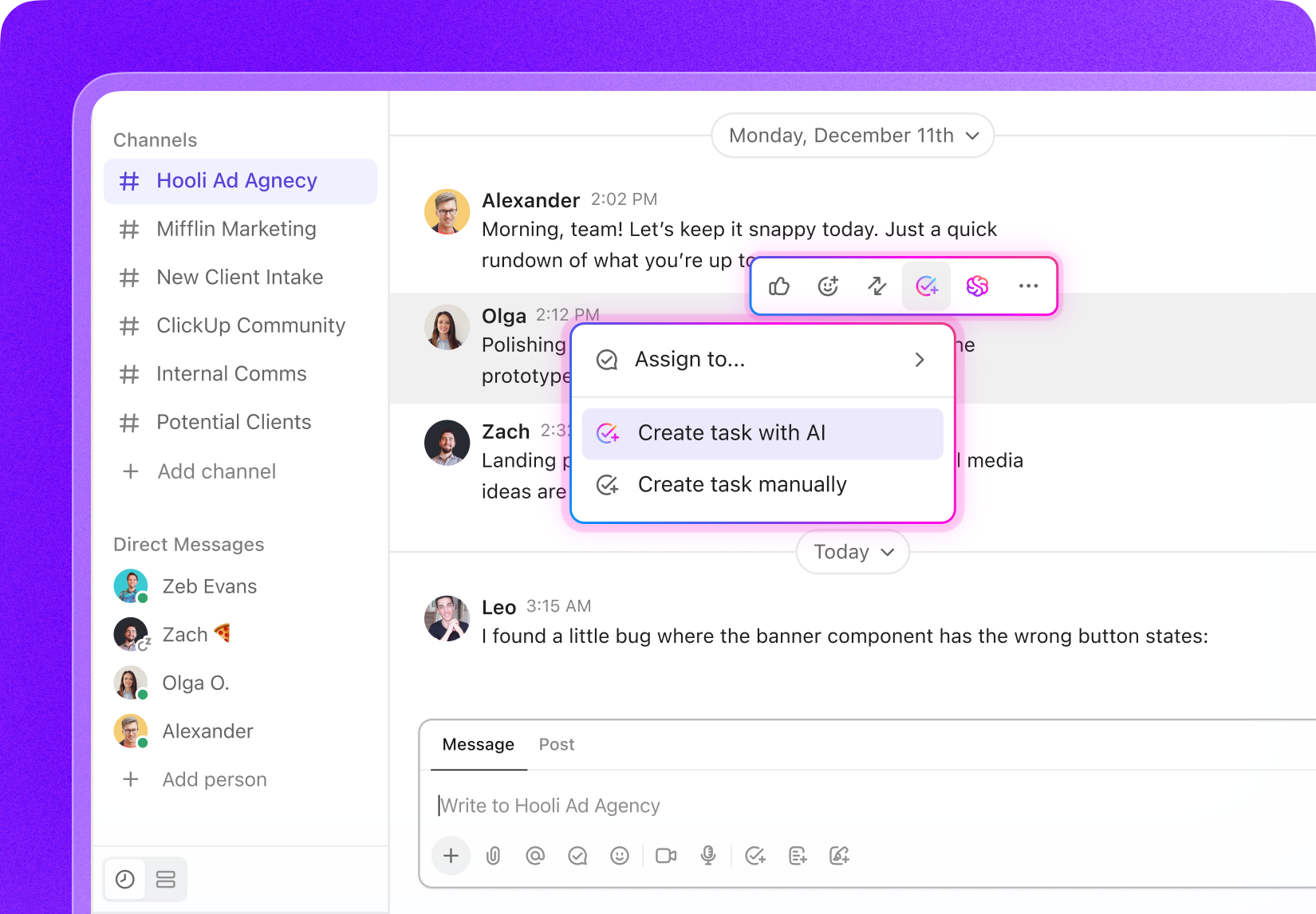
Task Management for Biomedical Innovators
Custom Task Management Software Designed for Biomedical Engineers
Unify your project tasks, monitor critical milestones, collaborate effortlessly with your team, and gain full oversight of every phase in your biomedical projects.
Trusted by the best
The Challenge of Managing Complex Projects
Why Biomedical Engineers Need Specialized Task Management Software
Handling biomedical projects without a dedicated system causes missed deadlines, scattered data, and operational inefficiencies — turning crucial tasks into uphill battles.
- Multistage device development blurs timelines — making it difficult to track design, testing, and regulatory approval phases.
- Data from experiments accumulates rapidly — risking loss or misplacement of critical results.
- Protocol inconsistencies arise — leading to unreliable test outcomes and costly repeats.
- Collaboration hurdles slow innovation — unclear responsibilities and version conflicts disrupt progress.
- Compliance deadlines loom — missing FDA or ISO submission dates can halt projects.
- Progress tracking is challenging — long development cycles feel stagnant without transparent updates.
- Communication gaps increase risk — fragmented notes and emails cause misalignment among cross-functional teams.
- Resource scheduling conflicts happen — lab equipment and trial timelines overlap, causing delays.
Traditional Approaches vs ClickUp
Where Conventional Tools Fall Short in Biomedical Engineering
Discover how ClickUp delivers precision and clarity beyond standard project tools.
Conventional Methods
- Tasks scattered across spreadsheets, emails, and paper notes
- Experimental data managed manually, prone to errors
- Protocols documented inconsistently
- Collaboration hindered by unclear task ownership
- Regulatory deadlines often missed
- Files and documents dispersed across platforms
ClickUp Task Management
- Centralized task tracking with clear priorities and statuses
- Structured data and protocol documentation with templates
- Consistent experimental workflows using checklists
- Transparent ownership with real-time team collaboration
- Automated alerts for compliance deadlines
- Unified document storage accessible to all stakeholders
Practical Use Cases
How Task Management Empowers Biomedical Engineers
See how streamlined workflows reduce errors and accelerate innovation.
#UseCase1
Unifying Experimental Data and Team Inputs
Consolidate protocols, test results, and design notes in one searchable workspace, ensuring data integrity and accessibility.
#UseCase2
Maintaining an Audit Trail for Compliance and Quality Assurance
Track every modification, comment, and submission to support regulatory audits and maintain product quality standards.
#UseCase3
Capturing Evolving Feedback from Cross-Disciplinary Teams
Manage input from clinicians, engineers, and regulatory experts with clear version control and actionable comments.
#UseCase4
Preventing Deviations in Clinical Trial Protocols
Use templates and task dependencies to ensure every trial step follows approved procedures without deviation.
#UseCase5
Organizing Ethical Approvals and Regulatory Submissions
Keep all documentation, checklists, and deadlines visible to guarantee timely IRB and FDA submissions.
#UseCase6
Mapping Complex Device Development Pipelines
Visualize and track each phase from design through validation with custom fields and status workflows.
#UseCase7
Meeting Critical Publication and Patent Deadlines
Manage manuscript preparations, patent filings, and conference abstracts with timeline views and task reminders.
#UseCase8
Avoiding Redundant Literature and Data Review
Assign and track literature as tasks with tags and notes to prevent duplicated efforts within the team.
#UseCase9
Turning Project Meetings into Clear Action Plans
Convert brainstorming sessions into assigned tasks with deadlines to maintain momentum and accountability.
Key Stakeholders
Who Benefits Most from ClickUp in Biomedical Engineering
Engineers, researchers, and teams seeking a unified platform to streamline complex workflows.
Biomedical Engineering Students
Stay organized through coursework, lab projects, and thesis deadlines without juggling scattered notes and tasks.
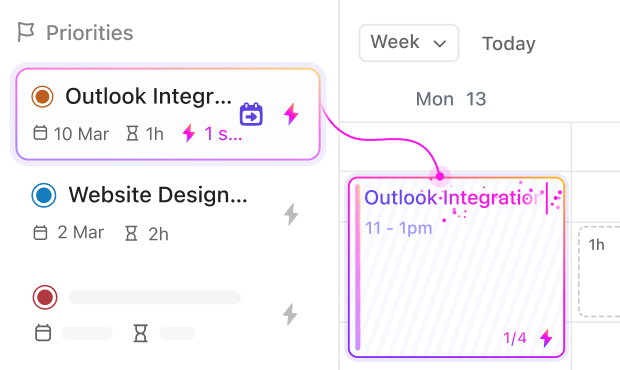
Research and Development Engineers
Standardize design protocols, manage resources, and ensure reproducible testing without manual tracking.
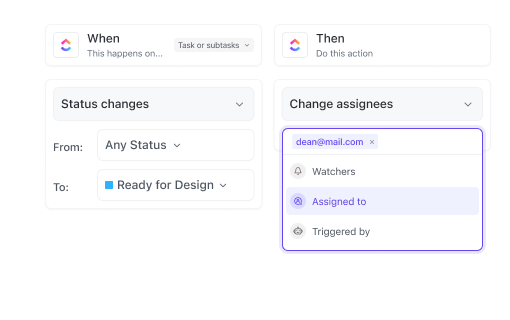
Cross-Functional Biomedical Teams
Coordinate complex tasks, roles, and timelines across departments to avoid bottlenecks and overlaps.
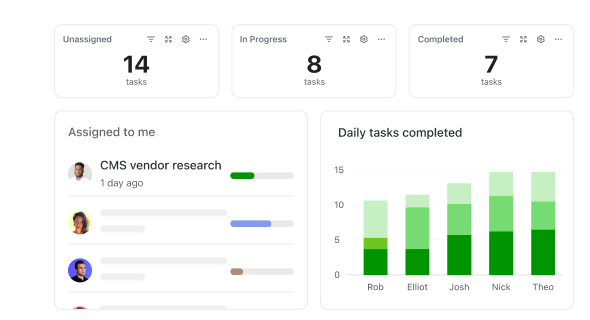
Harness ClickUp’s Full Potential
How ClickUp Accelerates Biomedical Engineering Projects
Manage device development, regulatory compliance, and collaboration without switching tools.
Centralize Everything
Store literature, datasets, protocols, drafts, and grant docs in one workspace — no more scattered files.
Plan Research in Phases
Break projects into proposal, literature review, experiments, analysis, and writing with task lists and Gantt timelines.
Standardize Experiments & Fieldwork
Use templates and checklists for repeatable, error-free lab or field procedures.
Collaborate Across Teams
Assign tasks to co-authors, lab members, or collaborators. Shared boards and dashboards keep everyone aligned.
Turn Meetings Into Actionable Tasks
Convert supervisor or lab meetings into tasks with owners, checklists, and deadlines.
Stay on Top of Deadlines & Funding
Track grants, conferences, and submissions with automated reminders and calendars.