Regulatory Affairs Knowledge Management
Streamline Your Regulatory Affairs Knowledge Base
Centralize policies, compliance guidelines, submission protocols, and audit trails into one searchable hub—so your team always accesses the latest regulatory insights.
Trusted by the best
ClickUp vs Legacy Systems
Elevate Regulatory Knowledge Management Beyond Traditional Tools
Unify compliance content and execution in a single platform.
With traditional tools
- Regulatory documents scattered across multiple platforms, causing version confusion
- Manual updates increase risk of outdated compliance information
- Disconnected workflows hinder traceability and accountability
- Complex permission settings lead to redundant document copies
- Time-consuming manual processes for document creation and review
With ClickUp
- Integrated knowledge and task management (Docs + tasks + comments)
- Use AI-powered ClickUp Brain to draft, summarize, and maintain regulatory content
- Link documentation directly to submission deadlines, audits, and compliance tasks
- Granular access controls for internal teams, external partners, and regulators
- Automation accelerates updates and enforces review cycles
Building Your Regulatory Knowledge Base
How to create a regulatory affairs knowledge base using ClickUp
Follow these 6 steps to ensure your compliance data is organized, accessible, and audit-ready.
1. Identify stakeholders and compliance objectives
- Define users from regulatory, legal, and quality teams
- Outline key compliance areas: submissions, labeling, audits, reporting
- Assign knowledge ownership for accountability
2. Develop a comprehensive knowledge base framework
- Create sections for guidelines, SOPs, submission templates, correspondence
- Ensure intuitive navigation with tables of contents and subpages
- Include version history and change logs for traceability
3. Standardize document formats and metadata
- Use consistent templates for submissions, risk assessments, and reports
- Tag documents with regulatory regions, product lines, and status
- Facilitate quick search and filtering to reduce compliance risks
4. Incorporate practical guides and regulatory updates
- Add how-to content for submissions, audits, and protocol adherence
- Track regulatory changes and update documents accordingly
- Centralize communication logs and correspondence summaries
5. Link knowledge base to workflows and audit timelines
- Connect documents to compliance tasks, deadlines, and review cycles
- Automate notifications for document reviews and regulatory milestones
- Use ClickUp Brain and Brain Max to synthesize compliance data
6. Manage permissions and continuous improvement
- Set access levels for internal staff, external consultants, and regulators
- Collect feedback and monitor document usage for ongoing refinement
- Schedule regular audits and updates to maintain compliance integrity
Your Regulatory Knowledge Companion
Unlock Compliance Confidence with ClickUp
Ensure regulatory documentation is accurate, actionable, and aligned with every submission cycle.
Organize
Structured Regulatory Documentation with ClickUp Docs
- Policies, SOPs, submission dossiers, audit reports
- Intuitive table of contents plus nested subpages
- Uniform document templates and metadata tags
Why it matters: Teams find critical compliance information quickly, reducing risk and delays.
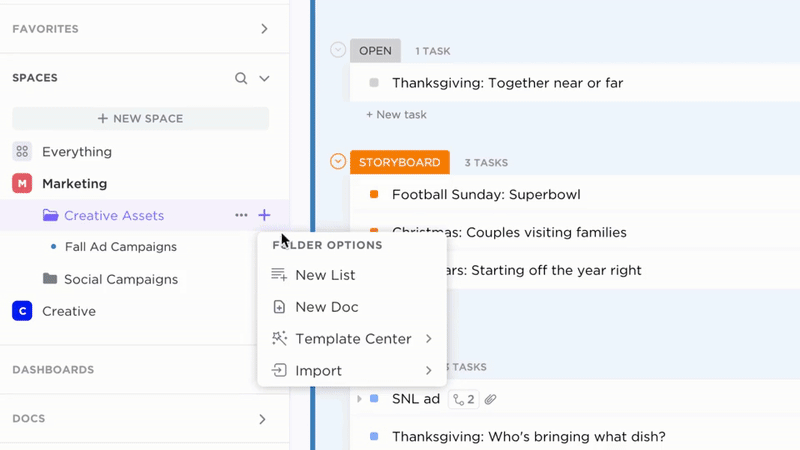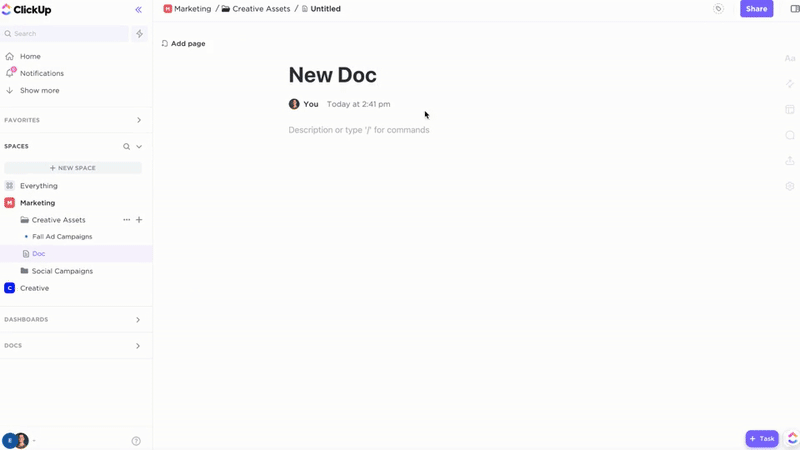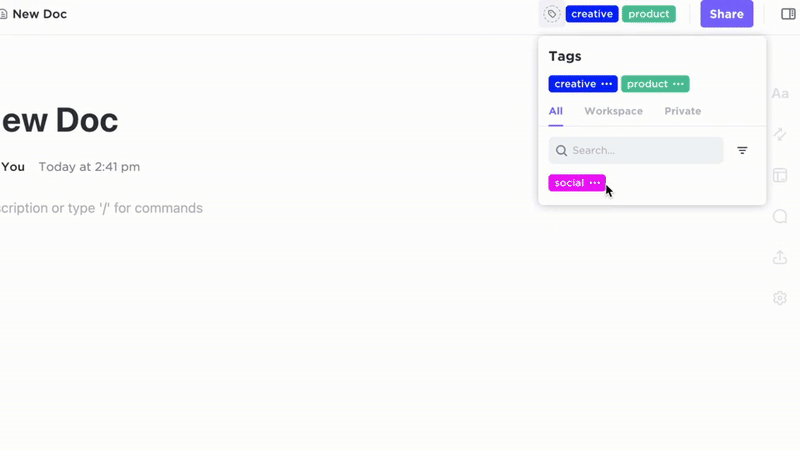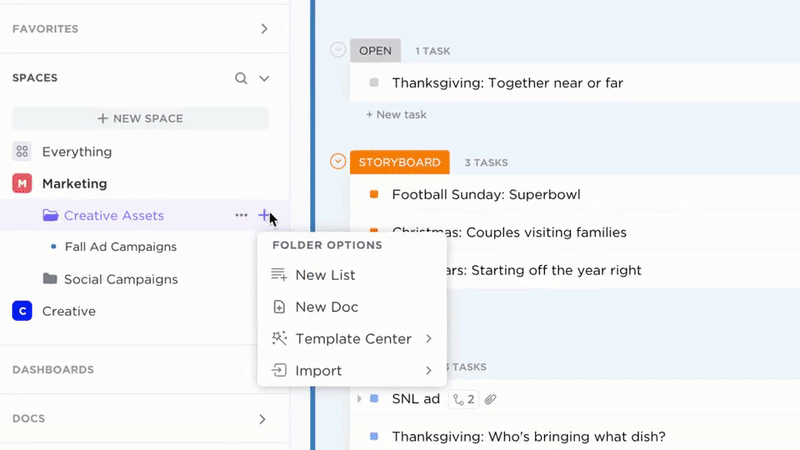
Accountability
Transparent Ownership and Tracking
- Convert documentation gaps into actionable tasks
- Assign responsibility with due dates and review schedules
- Monitor progress like any regulatory deliverable
Why it matters: Compliance documentation stays current because ownership is clear and tracked.
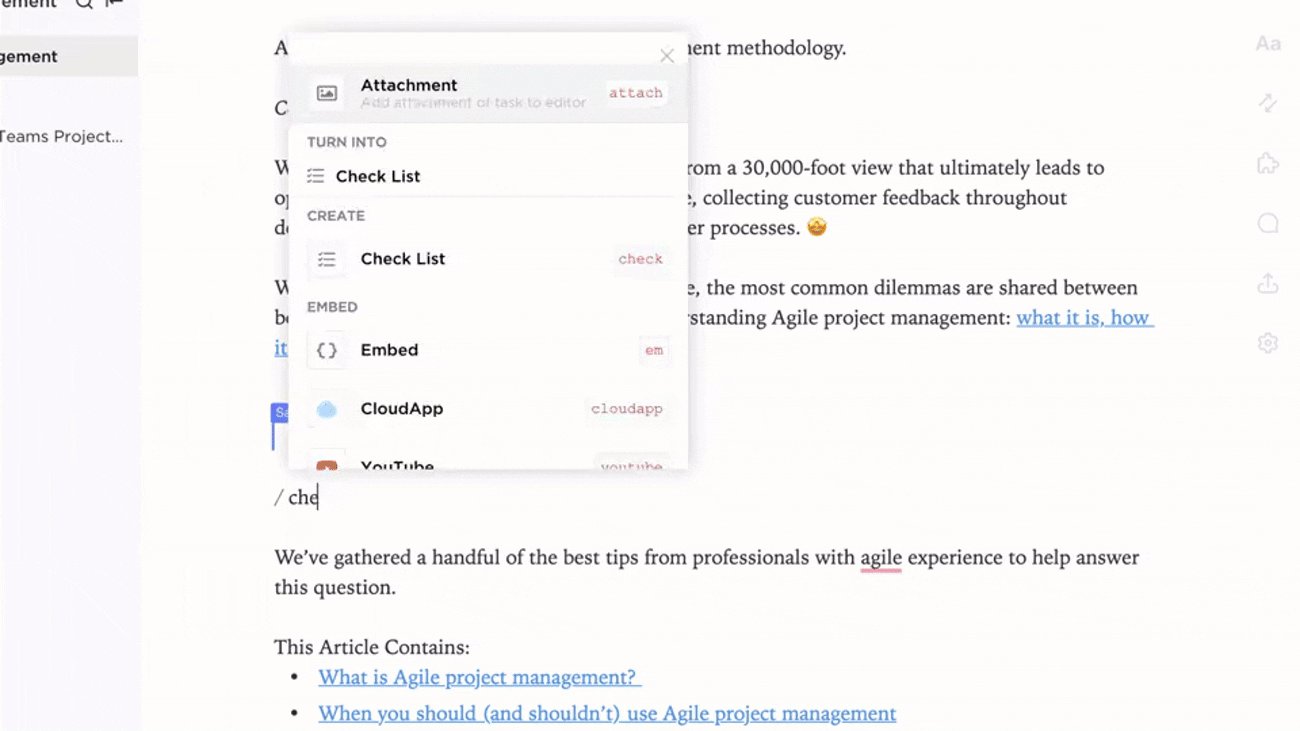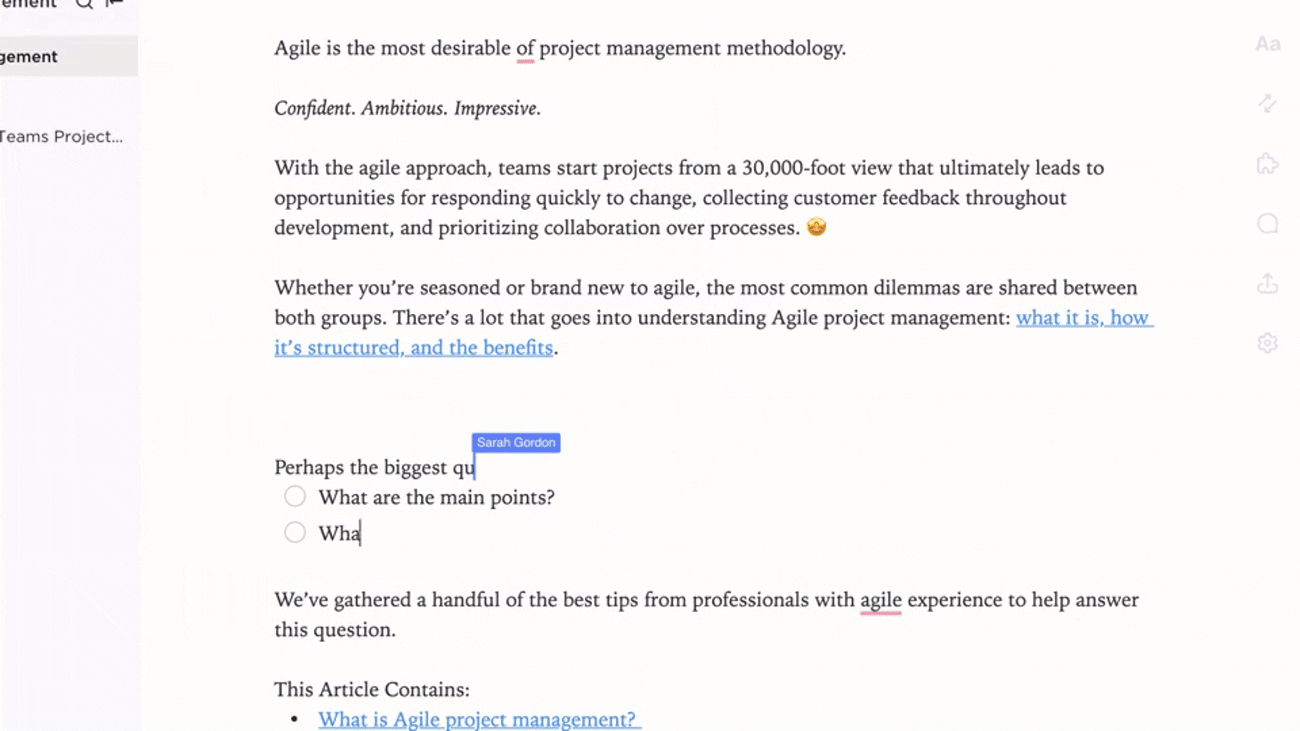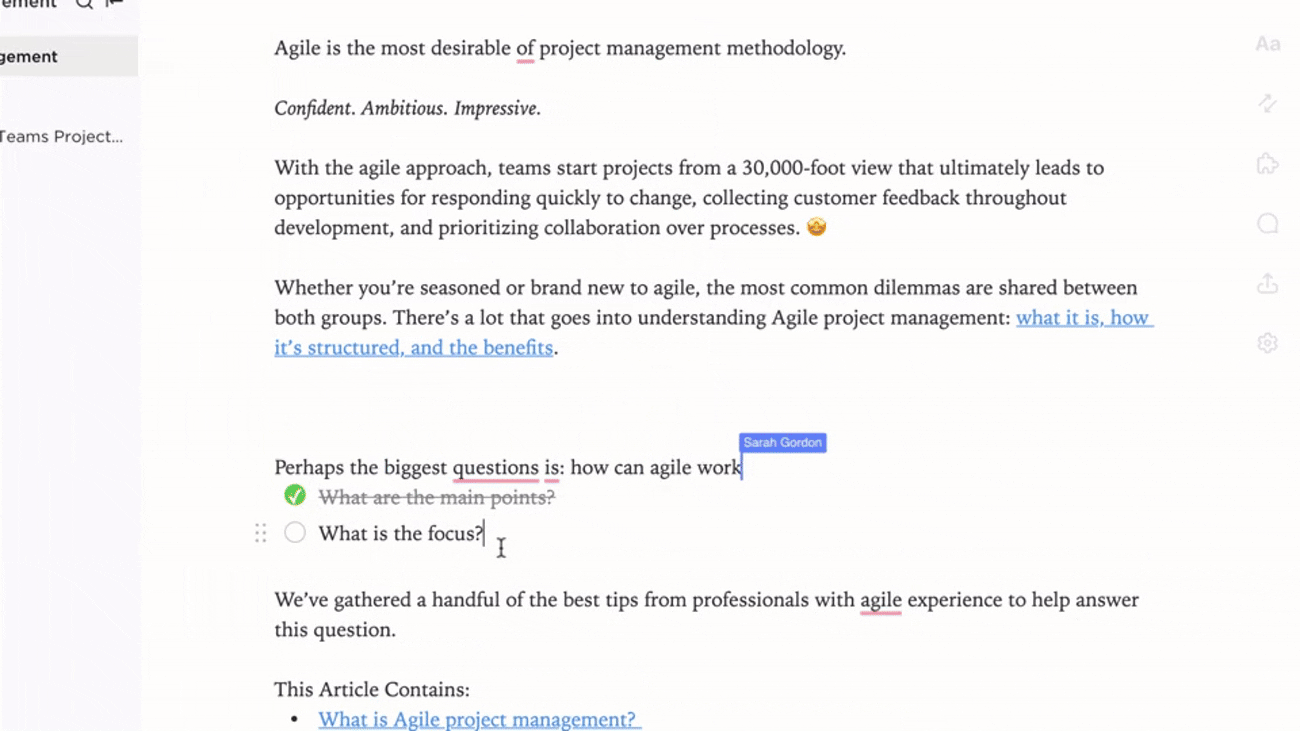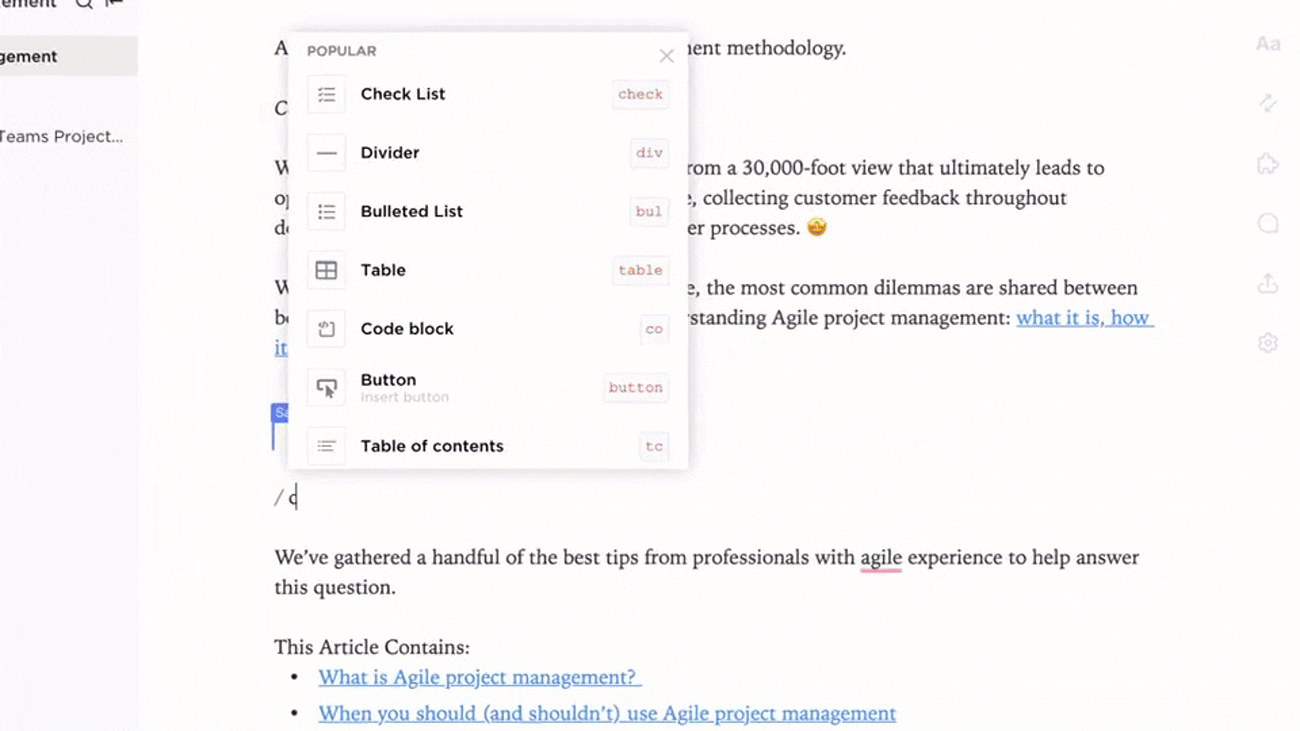
Integration
Documentation Linked to Regulatory Workflows
- Connect documents to submission deadlines and audit timelines
- Tie changes to regulatory updates, incident reports, and feedback
- Leverage AI tools to keep content fresh and aligned
Why it matters: Documentation evolves with your regulatory landscape, reducing compliance gaps.
Regulatory Knowledge Base FAQs

