Real-World Evidence Knowledge Base
Streamline Your Real-World Evidence Repository
Create a centralized, searchable platform for clinical data, study protocols, patient outcomes, and regulatory insights—ensuring your team accesses accurate evidence anytime.
Trusted by the best
ClickUp vs Traditional Platforms
Elevate Real-World Evidence Management Beyond Conventional Tools
Unify data and workflows in a single adaptable system.
Traditional Tools Limitations
- Disconnected data silos impede comprehensive insights
- Manual updates cause delays and inaccuracies
- Difficult collaboration across clinical and research teams
- Rigid access controls hinder information sharing
- Time-consuming documentation processes
Advantages of ClickUp for Real-World Evidence
- Integrate data, documentation, and tasks seamlessly
- Automate updates with ClickUp Brain and AI-driven workflows
- Facilitate multidisciplinary collaboration with Brain Max
- Tailor access for internal teams, partners, and regulators
- Accelerate evidence synthesis with ClickUp 4.0's advanced tools
Building Your Evidence Repository
Transforming Real-World Evidence Knowledge Base: Step-by-Step
Follow this 6-phase approach to keep your evidence structured, accessible, and current.
1. Define stakeholders and evidence objectives
- Identify clinical teams, researchers, and compliance officers
- Outline goals for data collection, analysis, and reporting
- Assign responsibilities to maintain data integrity
2. Develop a modular knowledge base architecture
- Establish core sections: patient data, study designs, outcomes, regulatory documents
- Implement clear navigation with subpages and indexing
- Ensure scalability to accommodate growing datasets
3. Standardize data and document formats
- Use consistent templates for study summaries, case reports, and metadata
- Include data provenance, methodology, and validation criteria
- Facilitate cross-study comparisons and meta-analyses
4. Incorporate practical guidance and troubleshooting
- Provide protocols for data collection and quality assurance
- Document common challenges and resolution strategies
- Centralize regulatory compliance checklists and audit trails
5. Link evidence updates to project milestones
- Connect documentation with clinical trial phases and data submissions
- Treat updates as integral to ongoing research workflows
- Maintain version control and audit logs for transparency
6. Manage access and continuous improvement
- Set granular permissions for internal teams, collaborators, and external reviewers
- Regularly review content relevance and accuracy
- Collect feedback to refine and expand the knowledge base
Harness ClickUp for Evidence Excellence
Unlock the potential of ClickUp for real-world evidence
Ensure your knowledge base is comprehensive, accountable, and synchronized with every research update.
Organize
Organized Evidence Documentation with ClickUp Docs
- Patient cohorts, study protocols, outcomes, regulatory filings
- Intuitive tables of contents and nested pages
- Uniform templates for consistent data capture
Why it matters: Teams quickly locate precise evidence, minimizing delays and errors.
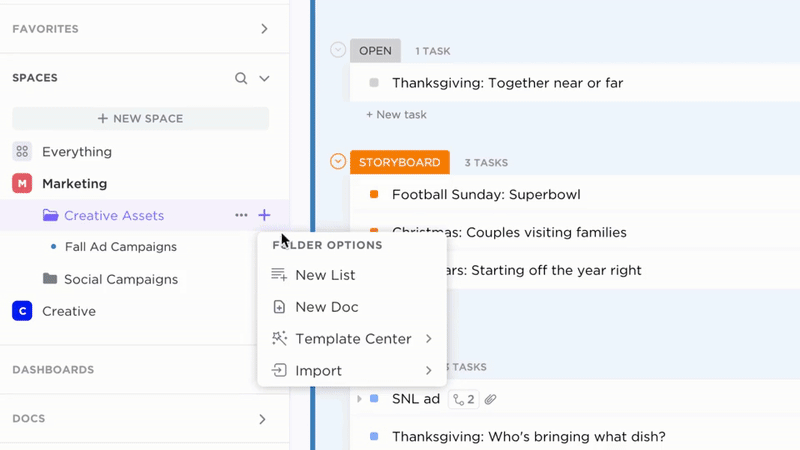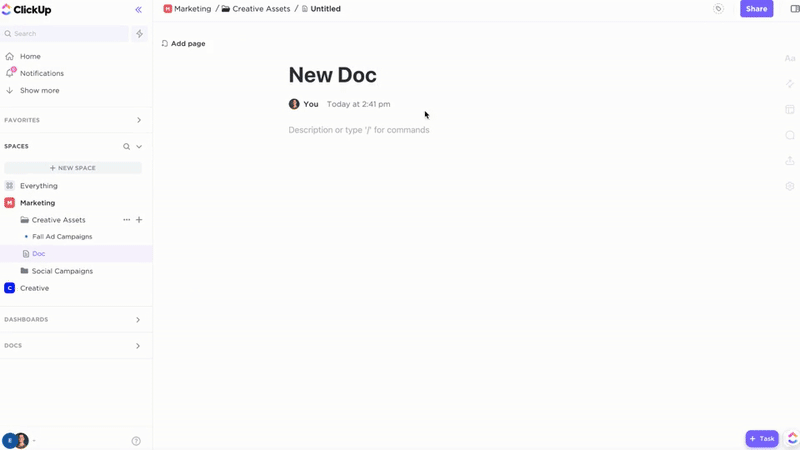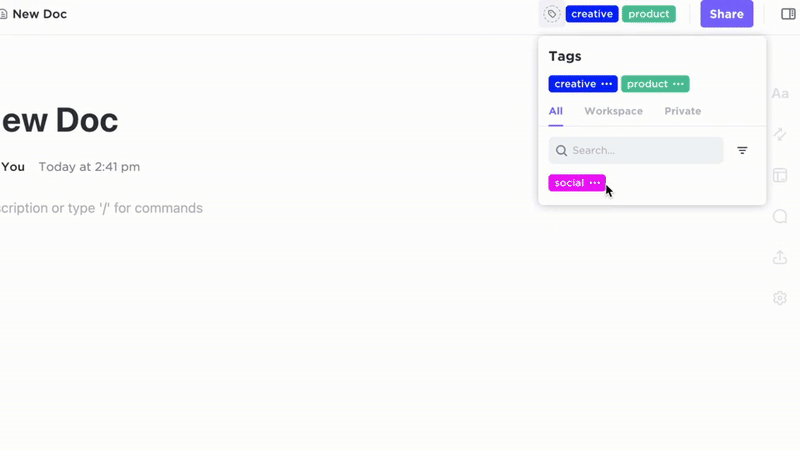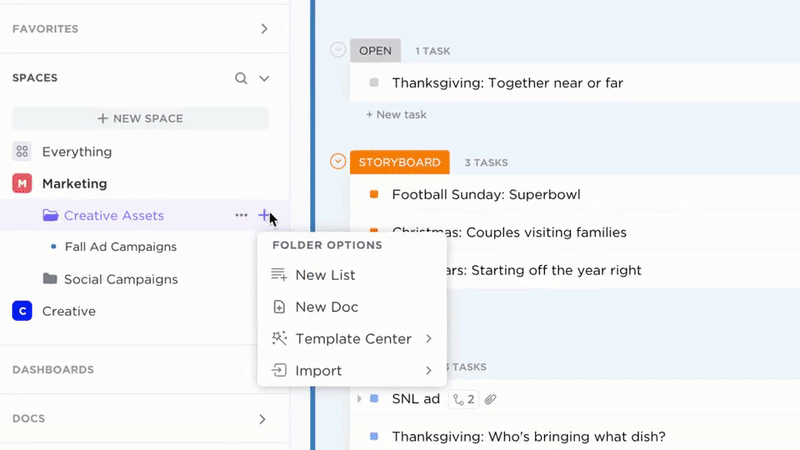
Manage
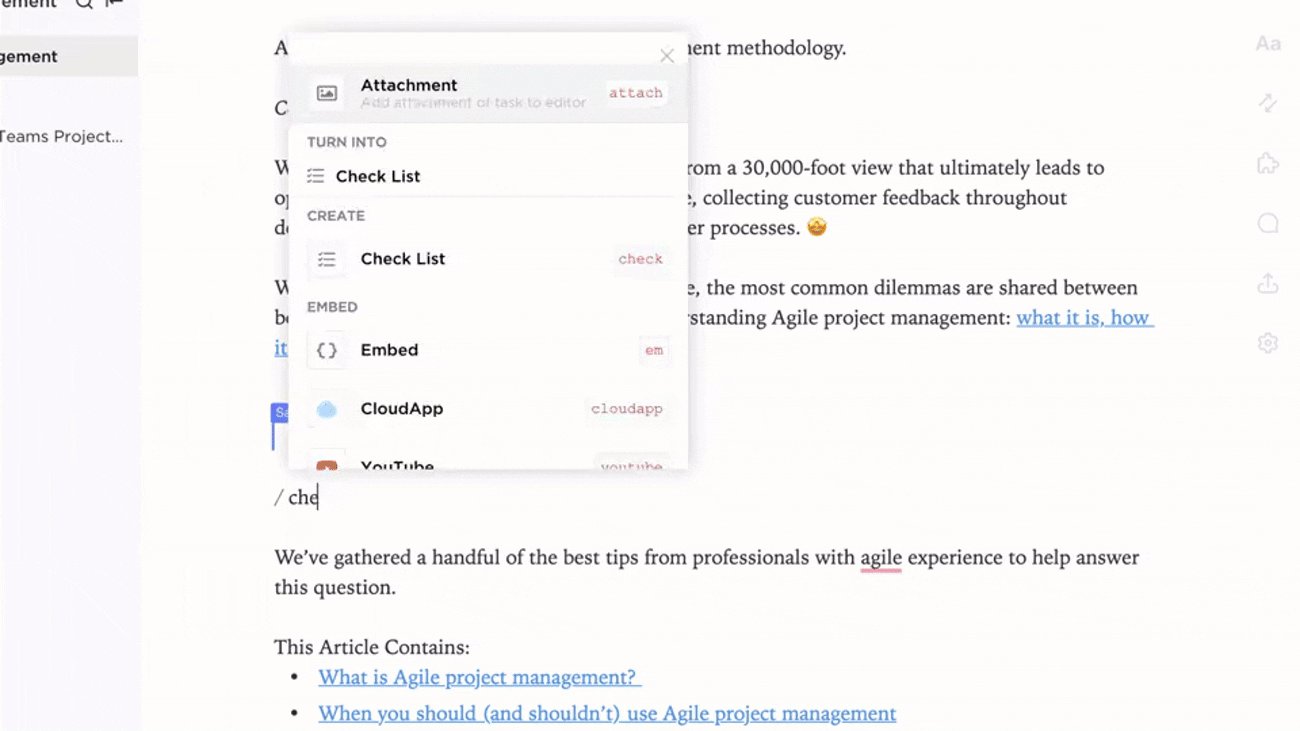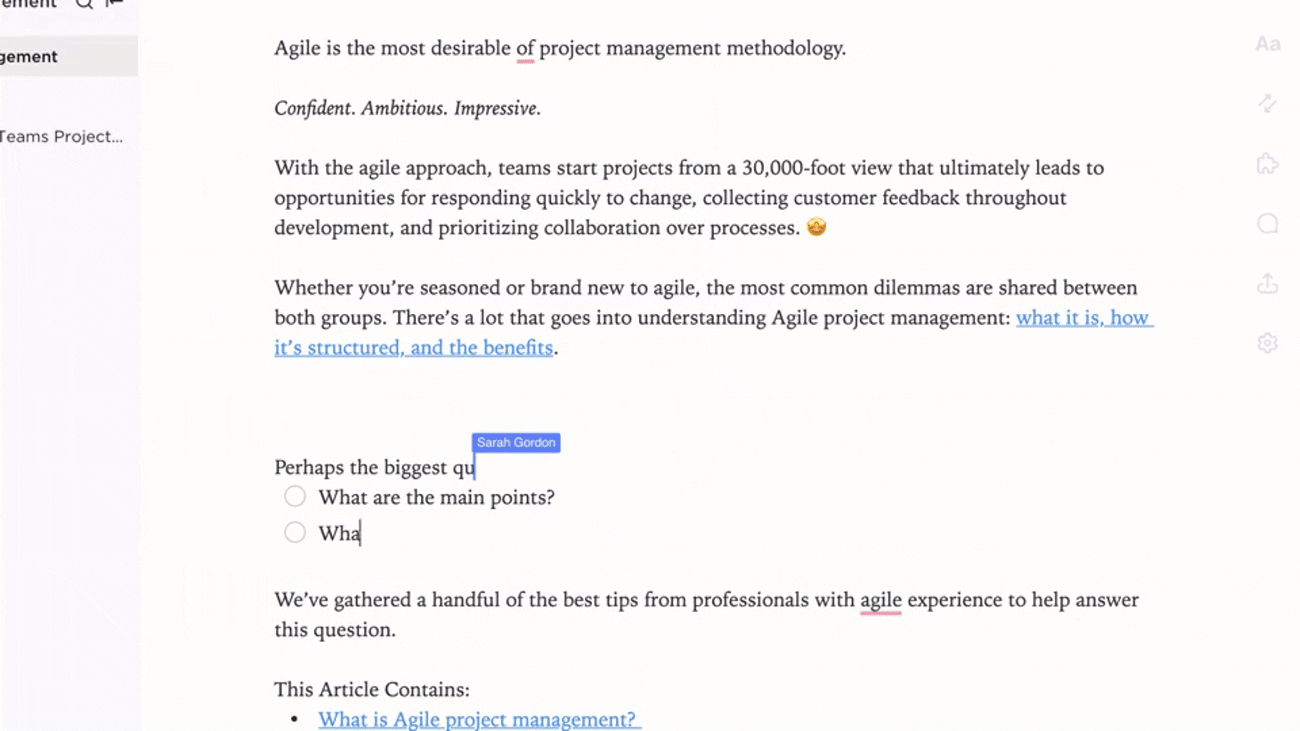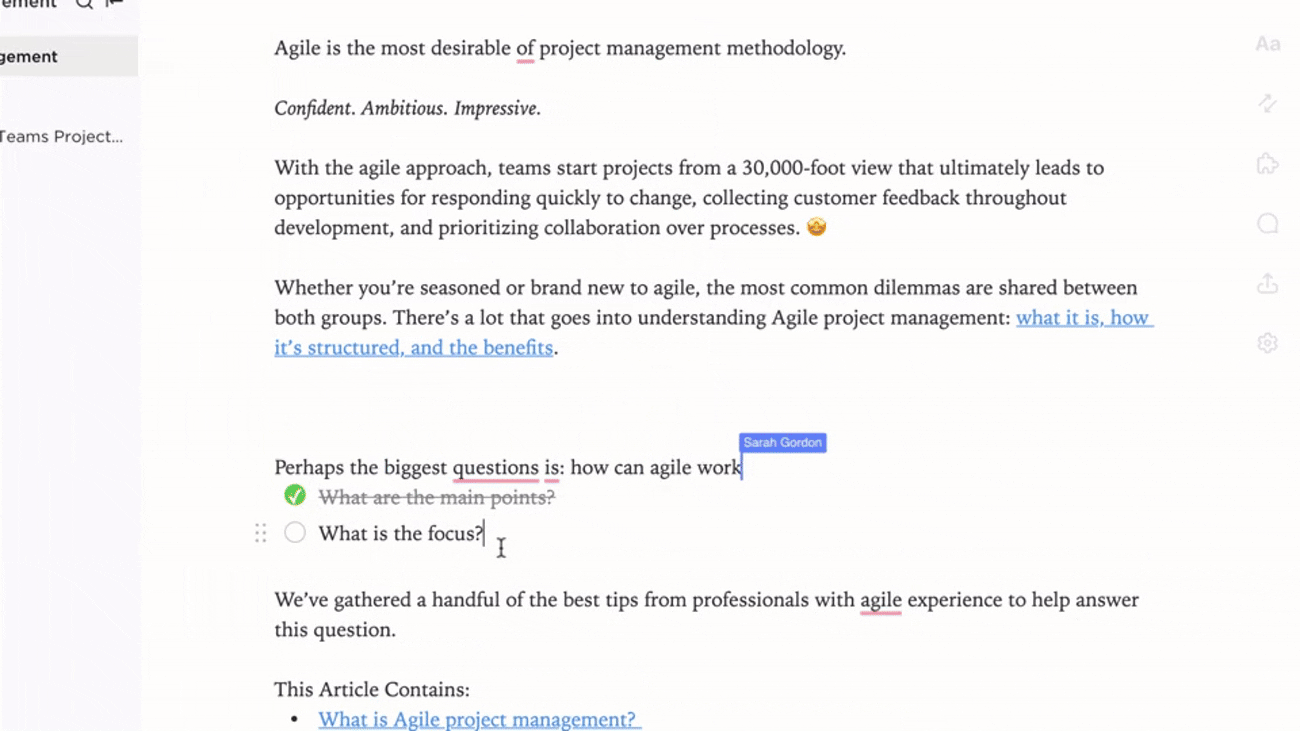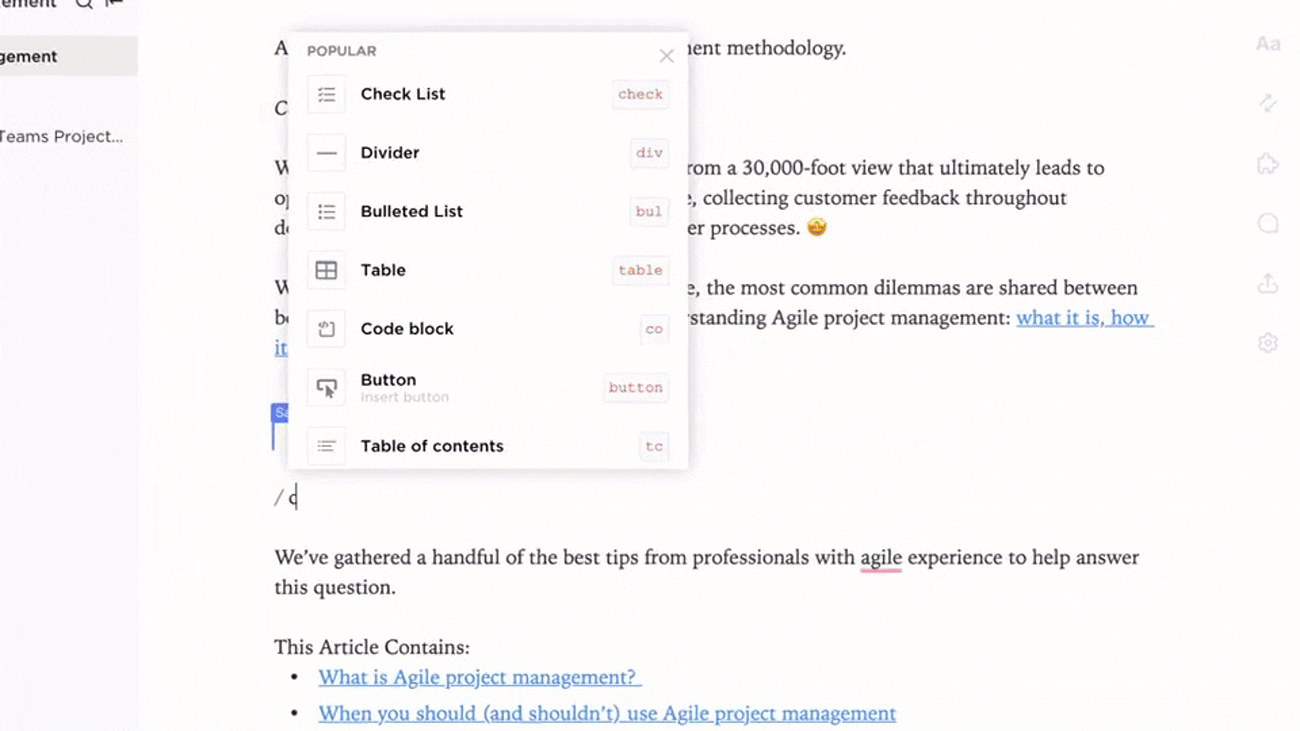
Transparent Ownership and Workflow Tracking
- Identify gaps and create action items with ClickUp Brain
- Assign data stewards with deadlines and review cycles
- Monitor progress as part of broader project management
Why it matters: Accountability ensures your evidence repository remains reliable and up to date.
Integrate
Evidence Linked Directly to Research Milestones
- Connect documentation to trial phases, data analyses, and regulatory submissions
- Link adverse event reports, post-market surveillance, and feedback
- Use Brain Max to surface insights and automate data synthesis
Why it matters: Documentation evolves alongside your research, reducing compliance risks.
Real-World Evidence Knowledge Base with ClickUp

