Build a Centralized Knowledge Base for Clinical Trial Operations
Create a comprehensive, searchable hub for protocols, SOPs, regulatory documents, site communications, and study updates—ensuring your teams access the most current clinical trial insights.
Why ClickUp Advances Clinical Trial Knowledge Management
Unify trial knowledge and operations in one scalable platform.
Traditional clinical trial tools
- Information scattered across disconnected systems, leading to outdated knowledge
- Manual updates increase risk of errors and delays
- Limited visibility into document revisions and task ownership
- Fragmented communication between sites and teams
- Time-intensive processes for maintaining SOPs and protocols
Using ClickUp for clinical trial knowledge base
- Integrate documents, tasks, and communications seamlessly
- Automate updates and assign action items with ClickUp Brain and Brain Max
- Link protocols, deviations, and site feedback in one dynamic workspace
- Granular permissions for internal teams, CROs, and regulatory partners
- AI-powered summaries and insights speed decision-making and compliance
How to build a knowledge base for clinical trial operations
Follow these 6 essential steps to maintain accurate, accessible, and compliant trial documentation.
1. Identify key users and clinical documentation objectives
- Define end users: clinical teams, monitors, regulatory reviewers
- Determine documentation scope: protocols, monitoring guides, training materials
- Assign knowledge stewards for ongoing content governance
2. Design a logical knowledge base framework
- Structure hubs for study phases, sites, and therapeutic areas
- Create standardized sections for protocols, SOPs, deviations, and reports
- Implement consistent naming conventions and navigation aids
3. Standardize document templates and content formats
- Use uniform formats for protocol sections, monitoring checklists, and training modules
- Include metadata for version control, approval status, and effective dates
- Embed regulatory references and study-specific notes to reduce queries
4. Incorporate practical guides and troubleshooting resources
- Develop how-to manuals for site setup, data entry, and adverse event reporting
- Provide escalation pathways and FAQ sections tailored to trial phases
- Centralize corrective action plans and deviation management strategies
5. Keep documentation synchronized with trial milestones
- Link knowledge base updates to protocol amendments, site activations, and safety reports
- Use ClickUp workflows and Brain Max to automate review cycles and approvals
- Ensure audit readiness by maintaining comprehensive change logs
6. Manage access controls and continuous content refinement
- Configure permissions for internal teams, external vendors, and regulatory authorities
- Schedule regular content audits and incorporate stakeholder feedback
- Leverage ClickUp AI to summarize complex updates and highlight action items
Unlock Clinical Trial Knowledge with ClickUp
Centralize trial protocols, monitoring plans, and regulatory documents—keeping your teams informed and aligned throughout the study lifecycle.
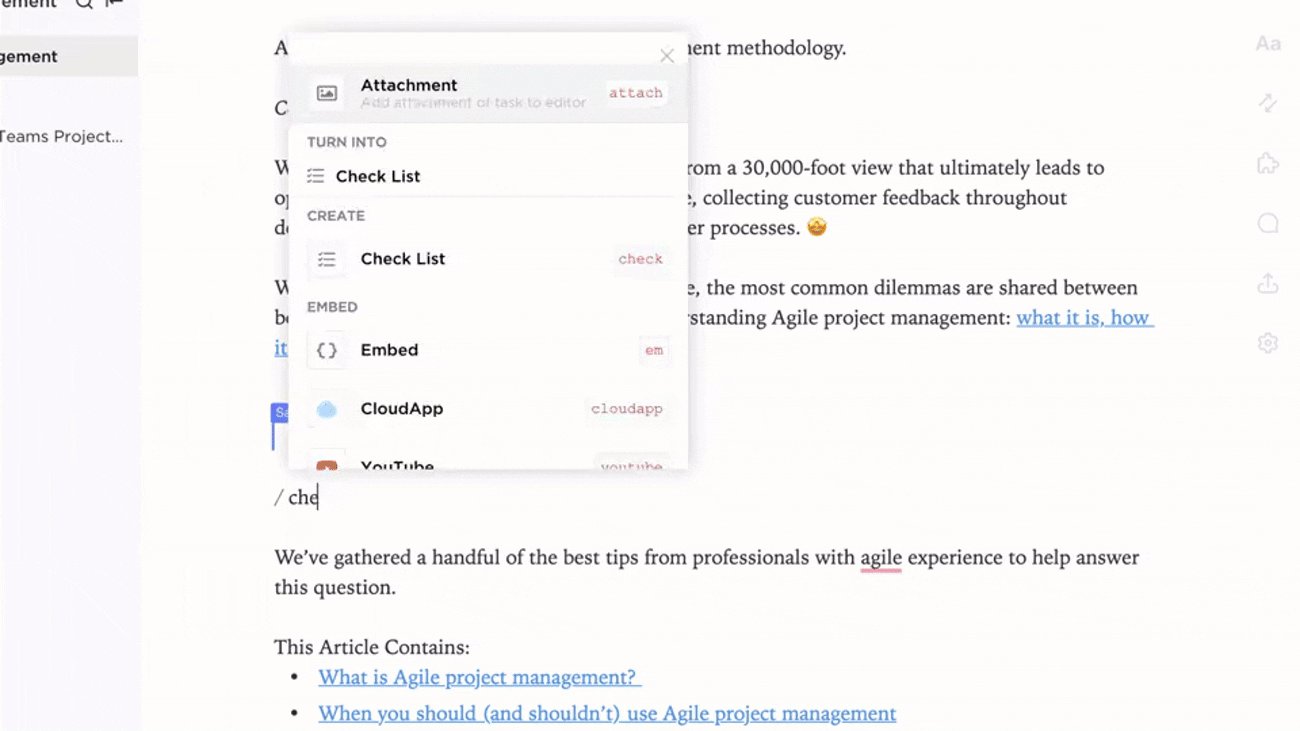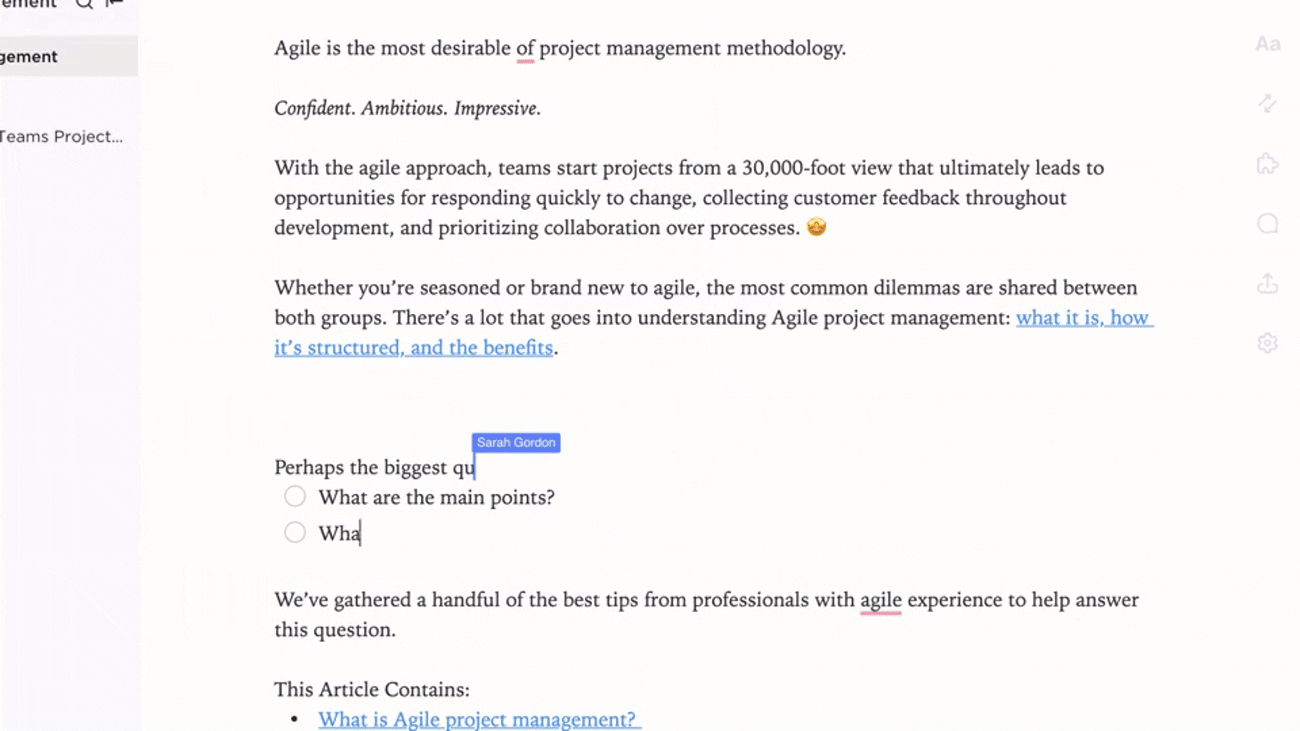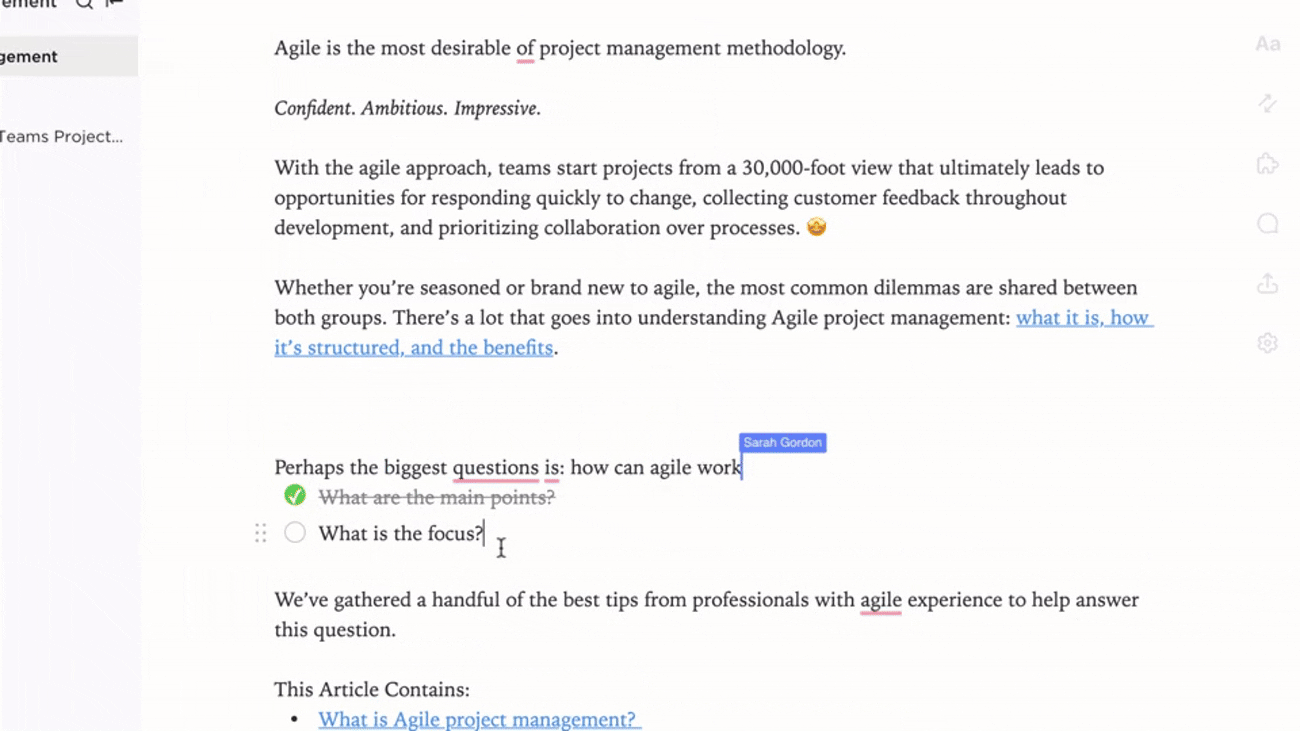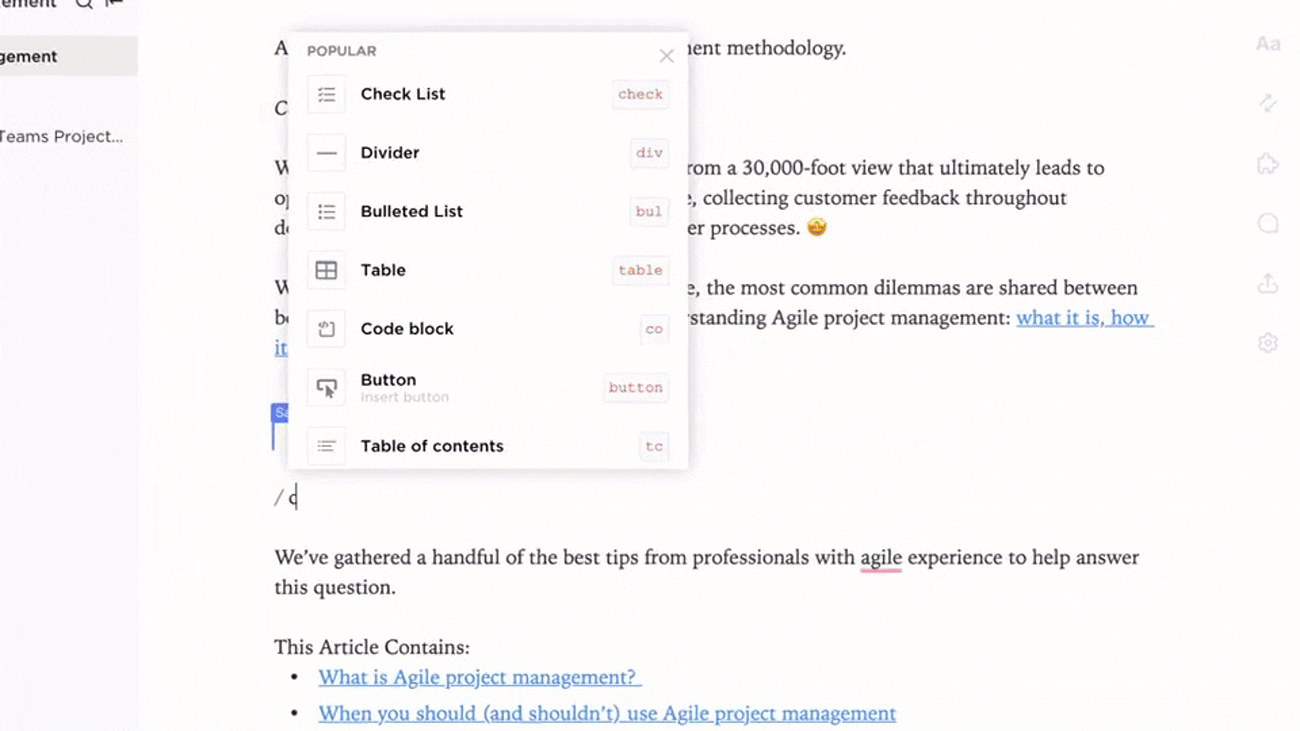
Structured Clinical Documentation in ClickUp Docs
- Protocols, SOPs, Monitoring Plans, Regulatory Submissions
- Table of contents and nested pages for seamless navigation
- Consistent template use for clarity and compliance
Why it matters: Teams locate critical information quickly, reducing delays and errors.
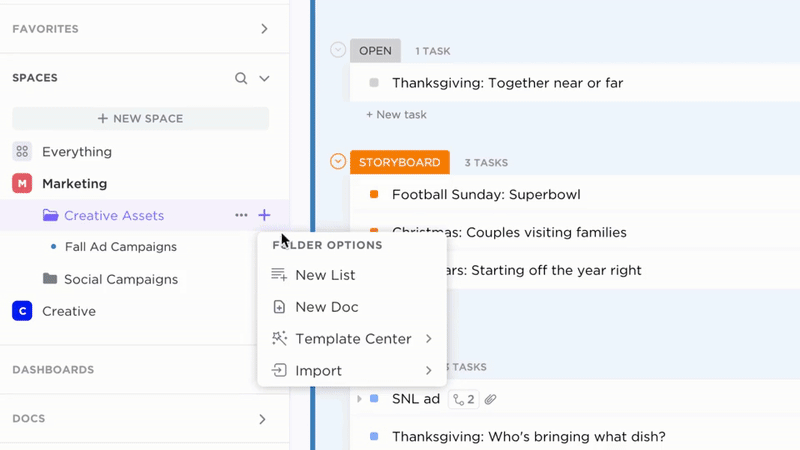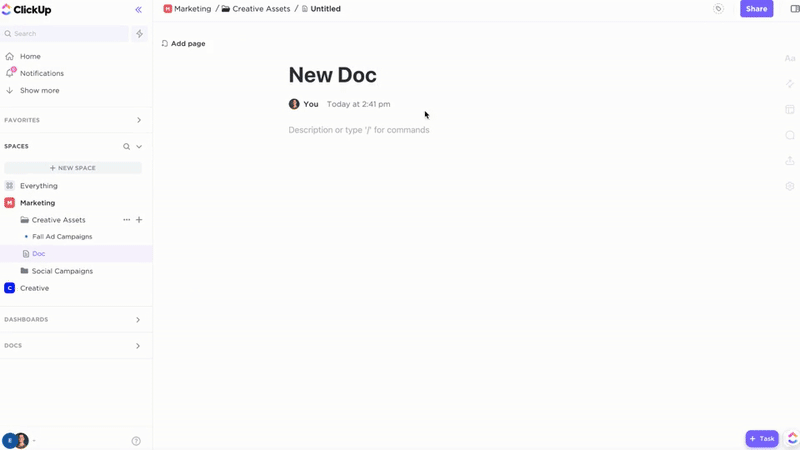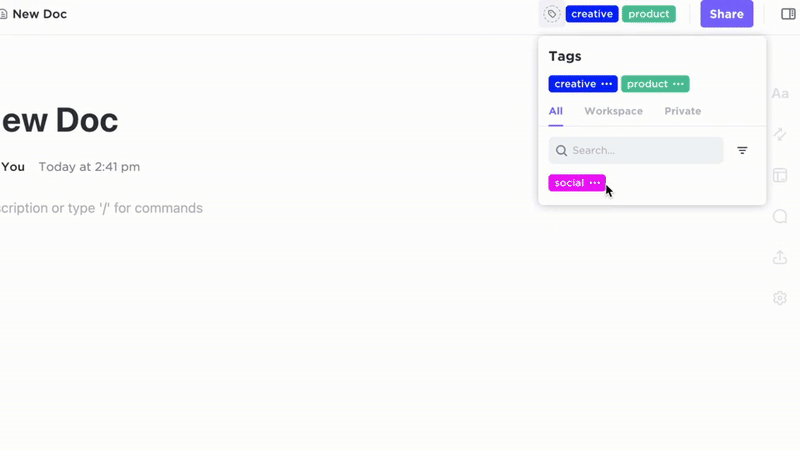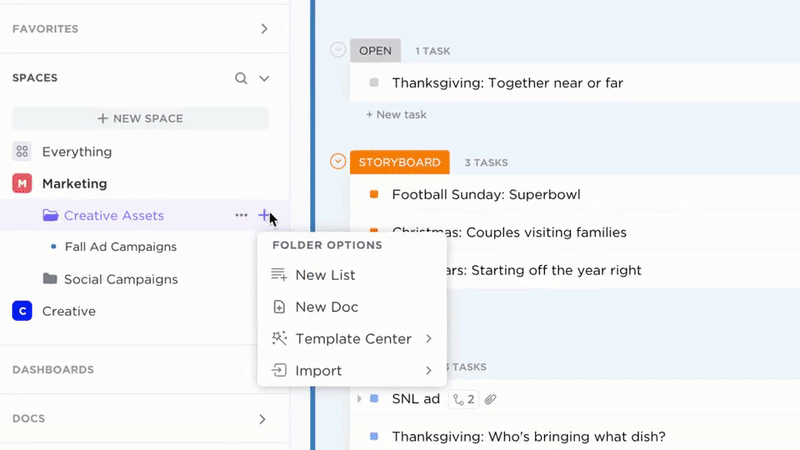
Transparent Documentation Ownership and Tracking
- Convert gaps or updates into actionable tasks
- Assign document owners, due dates, and periodic reviews
- Monitor progress and accountability alongside operational tasks
Why it matters: Knowledge stays current with clear responsibility and workflows.
Integrated Documentation Aligned with Trial Events
- Link documents to protocol amendments, monitoring visits, and safety reports
- Connect deviations, CAPAs, and site feedback to knowledge base entries
- Attach regulatory correspondence and post-study reports
Why it matters: Documentation evolves in sync with the trial, ensuring accuracy and compliance.