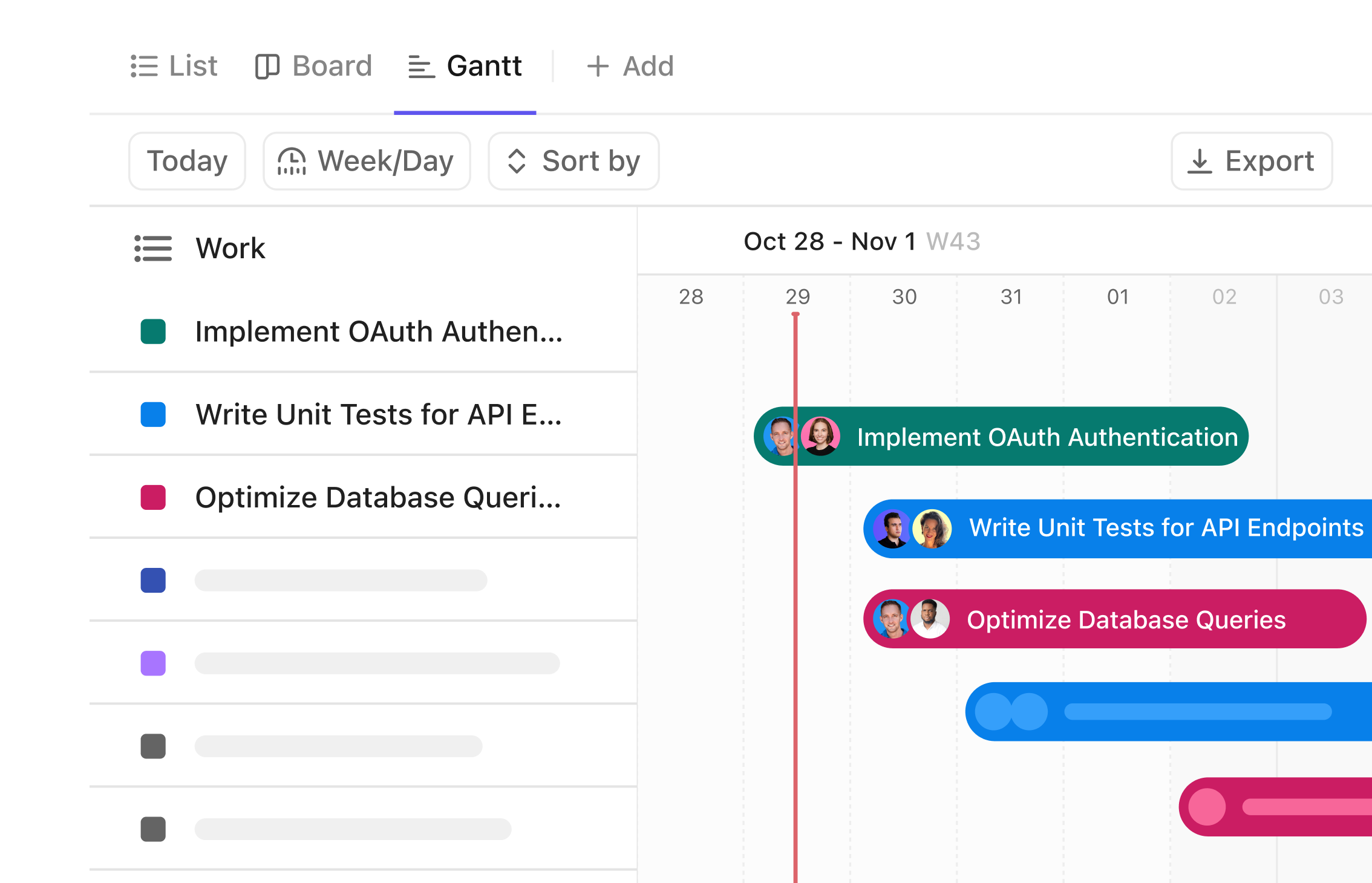
Gantt Chart for Drug Safety Associates
Visualize Drug Safety Timelines with Precision
Track every safety assessment, coordinate with cross-functional teams, and manage regulatory deadlines—all on a clear Gantt chart that streamlines your drug safety workflow.
Trusted by the best
Why Choose Gantt Charts
Why Drug Safety Associates Need Dedicated Gantt Chart Software
Managing drug safety without an organized timeline is like navigating a maze blindfolded. Critical safety checks might overlap or be missed, and deadlines can catch you off guard.
Here’s what typically goes wrong without a timeline view tailored for drug safety:
- Complex safety assessments become tangled — tracking adverse event reports and follow-ups grows confusing.
- Regulatory submissions risk delays — no clear visibility into due dates and dependencies.
- Cross-team communication falters — safety data and updates scattered across emails and spreadsheets.
- Monitoring plans lose clarity — unclear sequencing leads to duplicated or skipped safety evaluations.
- Audit readiness suffers — documentation and timelines aren’t easily accessible or updated.
- Resource allocation misfires — coordinating pharmacovigilance tasks without a timeline causes bottlenecks.
- Progress tracking is difficult — hard to see which safety activities are complete or pending.
- Risk of non-compliance grows — missed timelines can lead to regulatory penalties.
Traditional vs ClickUp
How ClickUp Gantt Outperforms Traditional Drug Safety Tools
Experience the clarity and control that standard methods can’t deliver in drug safety management.
Traditional Approaches
- Safety tasks tracked in isolated spreadsheets and emails
- Manual follow-ups increase risk of missed deadlines
- No unified view of assessment sequences
- Communication gaps between pharmacovigilance teams
- Regulatory timelines managed reactively
- Documentation scattered across platforms
ClickUp Gantt Charts
- Centralized task and timeline visualization
- Automated reminders for critical safety milestones
- Dependency tracking ensures correct task sequencing
- Clear assignment and ownership for all activities
- Integrated documentation linked directly to tasks
- Real-time collaboration across teams and departments
Use Cases
Unlock How Gantt Chart Software Empowers Drug Safety Associates
Discover how a visual timeline eliminates confusion and strengthens drug safety compliance.
#UseCase1
Eliminating Oversight in Long-Term Safety Monitoring
Visualize every safety check and report on a timeline—spotting delays and ensuring timely follow-ups across drug lifecycles.
#UseCase2
Building Transparent Regulatory Submission Timelines
Map all submission steps from data collection to filing—keeping stakeholders aligned and audit-ready.
#UseCase3
Tracking Adverse Event Investigations Efficiently
Update investigation stages with dependencies and priority shifts to keep safety evaluations on track.
#UseCase4
Preventing Data Gaps with Sequenced Pharmacovigilance Tasks
Define task sequences clearly so no safety signal or report is overlooked or duplicated.
#UseCase5
Coordinating Cross-Functional Compliance Activities
Manage timelines across clinical, regulatory, and quality teams for seamless collaboration.
#UseCase6
Managing Risk Minimization Measures and Follow-ups
Visualize all risk management steps with dependencies and deadlines to ensure effective implementation.
#UseCase7
Avoiding Missed Safety Reporting Deadlines
Track all report submissions with alerts to meet regulatory requirements on time.
#UseCase8
Organizing Safety Data Review Cycles
Assign review tasks and sequence data analysis stages clearly on your Gantt timeline.
#UseCase9
Turning Team Meetings into Actionable Safety Plans
Convert discussions into prioritized, scheduled tasks to maintain momentum and accountability.
Key Beneficiaries
Who Benefits Most from ClickUp Gantt Chart Software in Drug Safety
Ideal for professionals managing complex safety timelines, multiple teams, and stringent regulatory demands.
If you’re a Pharmacovigilance Specialist
Coordinate safety signal detection, case processing, and compliance reviews across departments with clear visual workflows.

If you’re a Drug Safety Scientist
Manage study timelines, data analysis, and adverse event tracking with precision and transparency.

If you’re a Regulatory Affairs Manager
Oversee submission schedules, documentation deadlines, and cross-team dependencies to ensure regulatory compliance.

How ClickUp Supports You
Step Into Streamlined Drug Safety with ClickUp Gantt Charts
Master your drug safety processes with clear timelines and actionable insights.
Centralize Safety Data and Documentation
Link adverse event reports, risk management plans, and protocols directly to tasks for easy access.
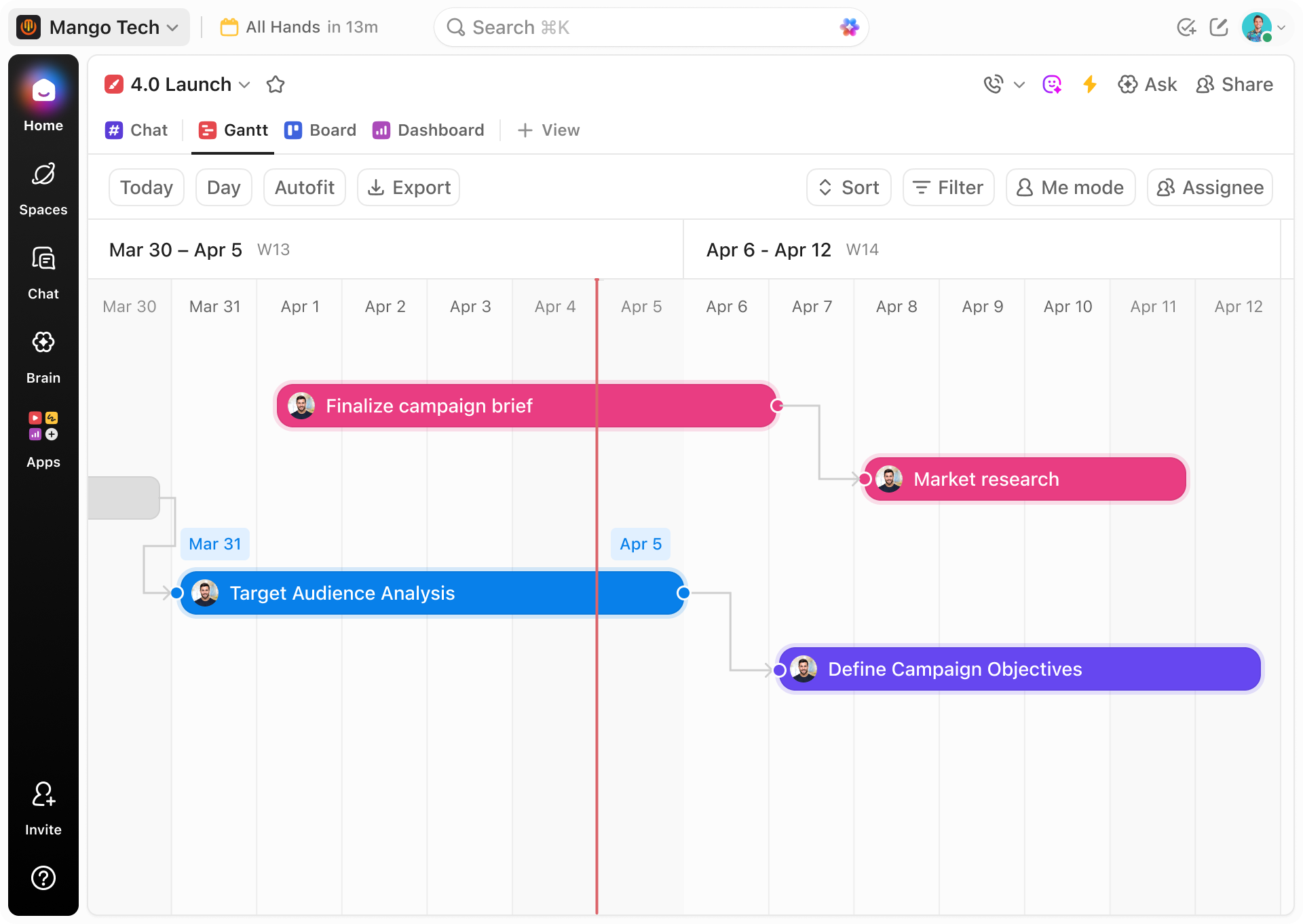
Plan Drug Safety Activities End-to-End
Visualize everything from signal detection to final report submission in one timeline.
Standardize Pharmacovigilance Workflows
Use dependencies and templates to ensure consistent, compliant safety processes.
Collaborate Across Teams Seamlessly
Assign responsibilities, track progress, and communicate clearly with integrated tools.
Turn Meetings into Executable Safety Plans
Transform discussions into scheduled tasks with deadlines and owners to keep safety initiatives moving.
Never Miss Critical Reporting Deadlines
Automated alerts and timeline views help you stay compliant with regulatory requirements.