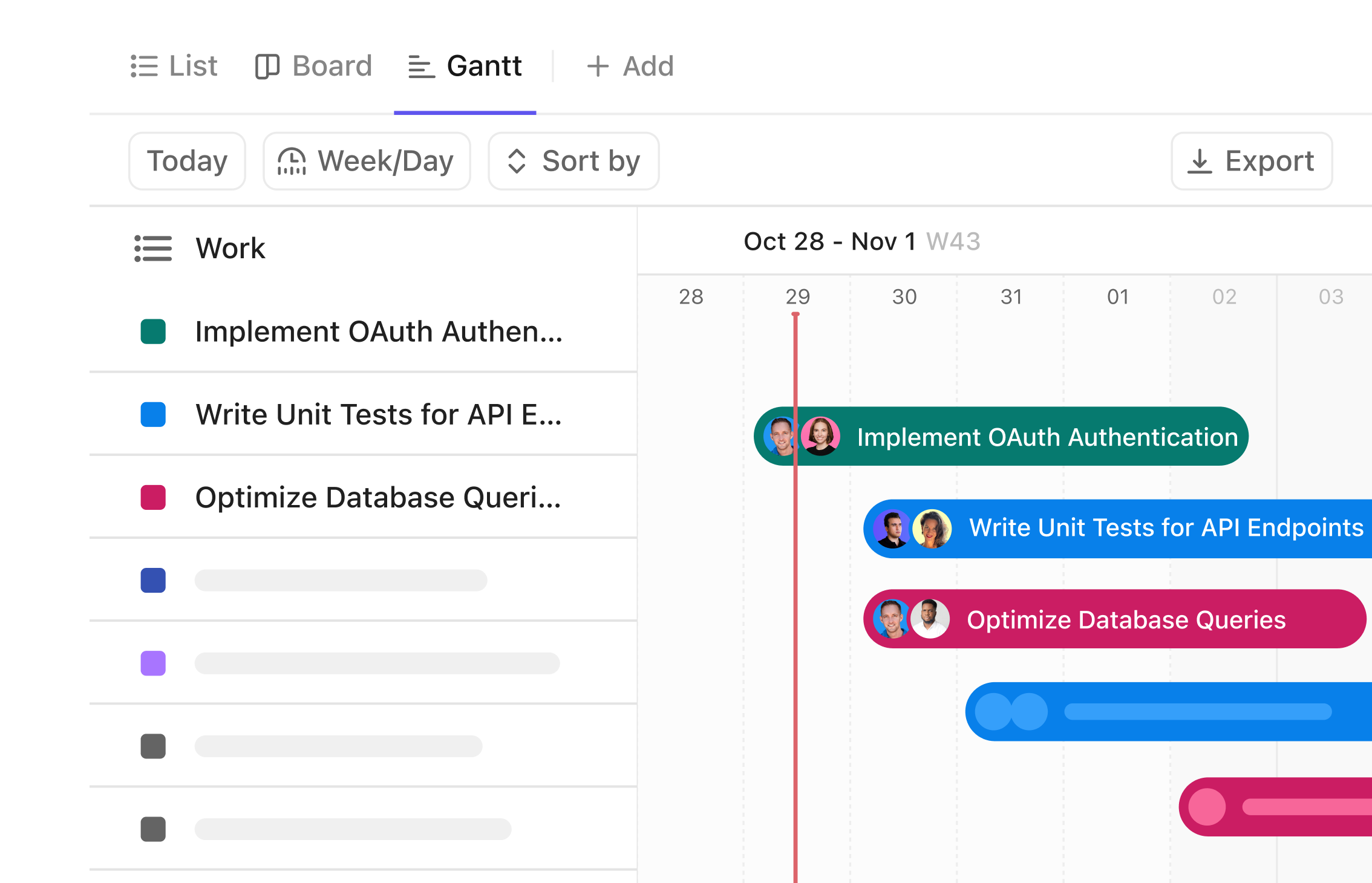
Gantt Chart Software for Clinical Trial Managers
Visualize Your Clinical Trial Progress with Precision
Track every phase from protocol design to patient follow-up, manage timelines across months or years, coordinate cross-functional teams, and gain a clear visual workflow that stays out of your head and into your hands.
Trusted by the best
The Importance of Gantt Charts
Why Clinical Trial Managers Rely on Gantt Chart Software
Managing clinical trials without a comprehensive timeline is like navigating a maze in the dark. Tasks overlap, deadlines loom unexpectedly, and team coordination suffers.
Common challenges faced without timeline visibility include:
- Unclear trial phase transitions — without a timeline, it’s difficult to track study start, enrollment, or close-out stages.
- Regulatory submissions get delayed — missing critical IRB or FDA deadlines can halt progress.
- Resource allocation conflicts — overlapping site visits, patient appointments, or equipment usage cause bottlenecks.
- Data collection inconsistencies — lack of sequencing and dependencies leads to protocol deviations.
- Communication breakdowns — dispersed information across emails and spreadsheets impedes alignment.
- Delayed milestone tracking — progress becomes opaque, risking trial timelines and budgets.
- Complex multi-site coordination — synchronizing tasks across locations becomes overwhelming.
- Inefficient risk management — potential issues aren’t flagged early without clear visualization.
Traditional vs ClickUp Gantt
Why Traditional Tools Fall Short for Clinical Trial Management
Explore how ClickUp’s Gantt Chart software offers unmatched clarity compared to conventional methods.
Traditional Methods
- Rely on disparate spreadsheets and emails
- Manual tracking of patient enrollment and site visits
- No clear visualization of trial phase dependencies
- Difficulty managing regulatory submission deadlines
- Overlapping resource and staff scheduling
- Limited collaboration across teams and sites
ClickUp Gantt Charts
- Unified view of tasks, timelines, and dependencies
- Schedule patient enrollment, monitoring, and data collection
- Visualize trial phases and milestones with clarity
- Automated reminders for regulatory and site deadlines
- Coordinate resources and personnel seamlessly
- Integrate documents, protocols, and communications
Use Cases
How Gantt Chart Software Empowers Clinical Trial Managers
Discover practical ways a visual timeline streamlines clinical trial execution and oversight.
#UseCase1
Eliminating Phase Overlaps and Delays
ClickUp’s Gantt charts map out every trial phase, highlighting dependencies so delays are caught early and steps proceed smoothly.
#UseCase2
Ensuring Regulatory Compliance on Schedule
Track IRB submissions, FDA approvals, and audit preparations with automated alerts to avoid costly missed deadlines.
#UseCase3
Coordinating Multi-Site Activities Transparently
Visual timelines synchronize site visits, patient enrollments, and monitoring visits, ensuring cohesive multi-location collaboration.
#UseCase4
Managing Resource Allocation Efficiently
Avoid conflicts by scheduling equipment, staff, and lab time with clear visibility into overlapping demands.
#UseCase5
Tracking Patient Enrollment and Retention
Map recruitment targets, monitor retention rates, and schedule follow-ups with precise timeline controls.
#UseCase6
Facilitating Data Collection and Analysis Milestones
Sequence data entry, quality checks, and analysis phases to maintain protocol adherence and integrity.
#UseCase7
Anticipating and Mitigating Risks Early
Visualize potential bottlenecks and resource constraints to proactively manage trial risks before they escalate.
#UseCase8
Streamlining Communication Across Teams
Centralize updates, feedback, and documentation linked directly to timeline tasks to keep everyone aligned.
#UseCase9
Optimizing Budget and Timeline Forecasting
Use real-time progress tracking to adjust forecasts and keep the trial on budget and on schedule.
Key Beneficiaries
Who Benefits Most from ClickUp Gantt Charts in Clinical Trials
Ideal for professionals managing complex timelines, diverse teams, and critical deadlines in clinical research.
If You’re a Clinical Project Manager
Coordinate across multiple sites and teams seamlessly with a centralized timeline that updates in real-time.

If You’re a Site Coordinator
Manage patient visits, data collection, and regulatory tasks with clear scheduling to minimize errors and overlaps.

If You’re a Data Manager
Track data entry timelines, quality control tasks, and analysis milestones to ensure trial data integrity.

How ClickUp Advances Clinical Trial Management
6 Ways ClickUp Gantt Charts Enhance Your Trial Workflow
Elevate your clinical trial oversight with ClickUp’s powerful timeline features.
Centralize Trial Documentation
Link protocols, consent forms, and monitoring reports directly to timeline tasks for easy access.
Visualize Study Phases End-to-End
Track protocol development, recruitment, treatment, and analysis in a single, interactive timeline.
Standardize Task Sequences and Dependencies
Use templates and dependencies to enforce protocol adherence and prevent procedural errors.
Facilitate Cross-Functional Team Coordination
Assign responsibilities with deadlines and monitor progress across clinical operations and data teams.
Convert Meetings into Actionable Task Lists
Turn team discussions into sequenced tasks on your Gantt chart—so nothing falls through the cracks.
Receive Automated Alerts for Critical Deadlines
Stay ahead of IRB submissions, site initiations, and close-out visits with timely notifications.