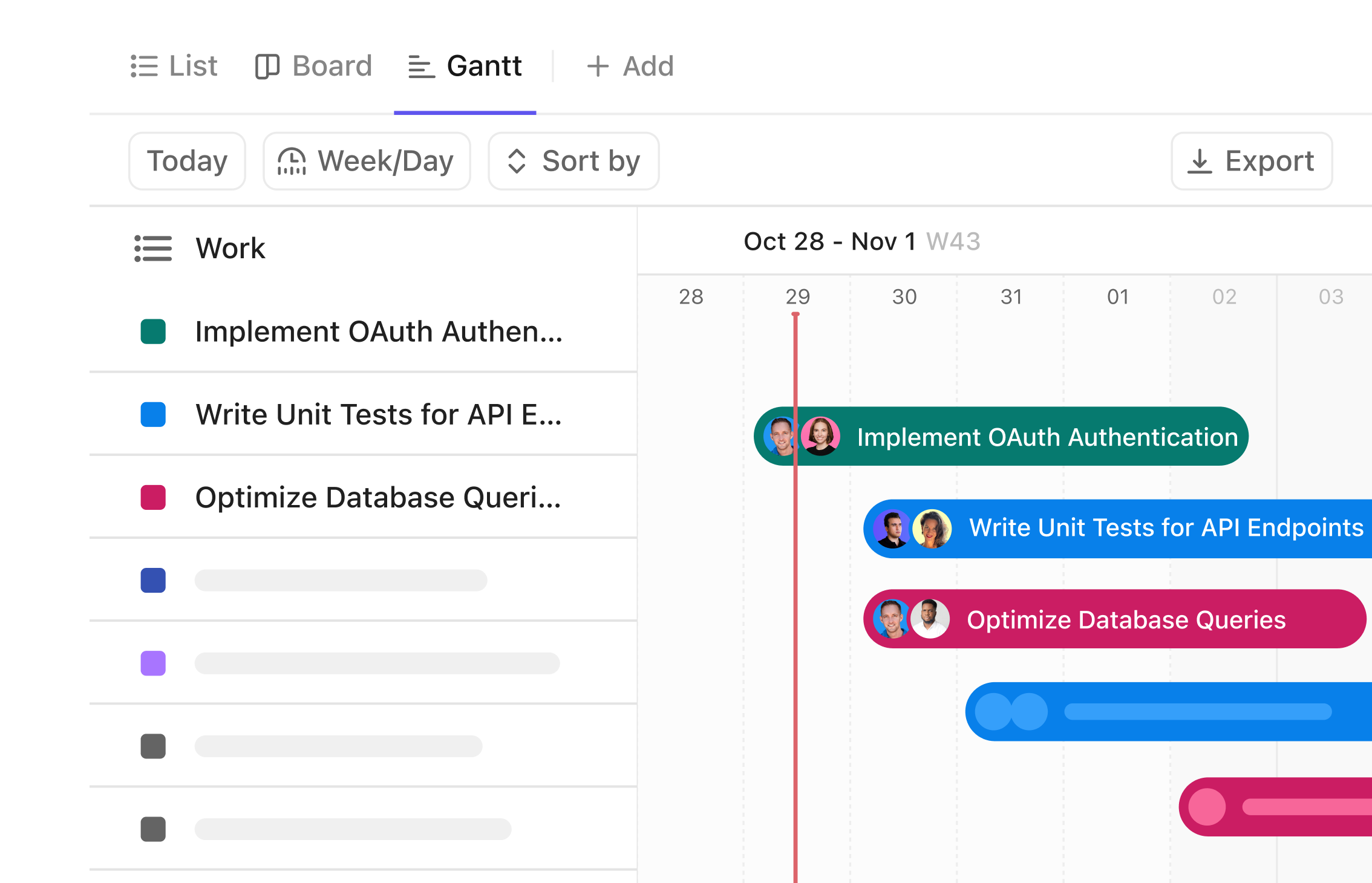
Gantt Chart for Biomedical Technicians
Transform Your Equipment Maintenance Workflow with Gantt Charts
Visualize every repair, calibration, and inspection across your biomedical equipment lifecycle. Coordinate tasks and timelines effortlessly with a dynamic Gantt chart that keeps your lab running smoothly.
Trusted by the best
Why Choose Gantt Charts
Why Biomedical Technicians Benefit from Gantt Chart Software
Managing biomedical equipment without a clear timeline invites delays, miscommunications, and compliance risks.
Here’s what typically goes wrong without a visual workflow:
- Maintenance schedules become inconsistent — leading to unexpected equipment failures.
- Calibration deadlines get missed — risking inaccurate diagnostics or treatments.
- Repair tasks overlap or get forgotten — causing bottlenecks and downtime.
- Team coordination falters — unclear task ownership causes duplicated efforts.
- Regulatory compliance timelines slip — increasing audit risks and penalties.
- Progress tracking is unclear — making it hard to report status or justify budgets.
- Communication scatters across emails and logs — complicating accountability.
- Resource conflicts arise — tools and parts may be double-booked or unavailable.
Conventional Methods vs ClickUp
Why Traditional Tracking Falls Short Compared to ClickUp Gantt
Discover how ClickUp Gantt Charts bring clarity and control to biomedical equipment management unlike any spreadsheet or manual logbook.
Traditional Approaches
- Paper logs and spreadsheets prone to errors
- Maintenance dates tracked separately, often missed
- No clear visualization of overlapping tasks
- Repair priorities unclear among teams
- Compliance deadlines tracked manually
- Scattered notes and incomplete records
Use Cases
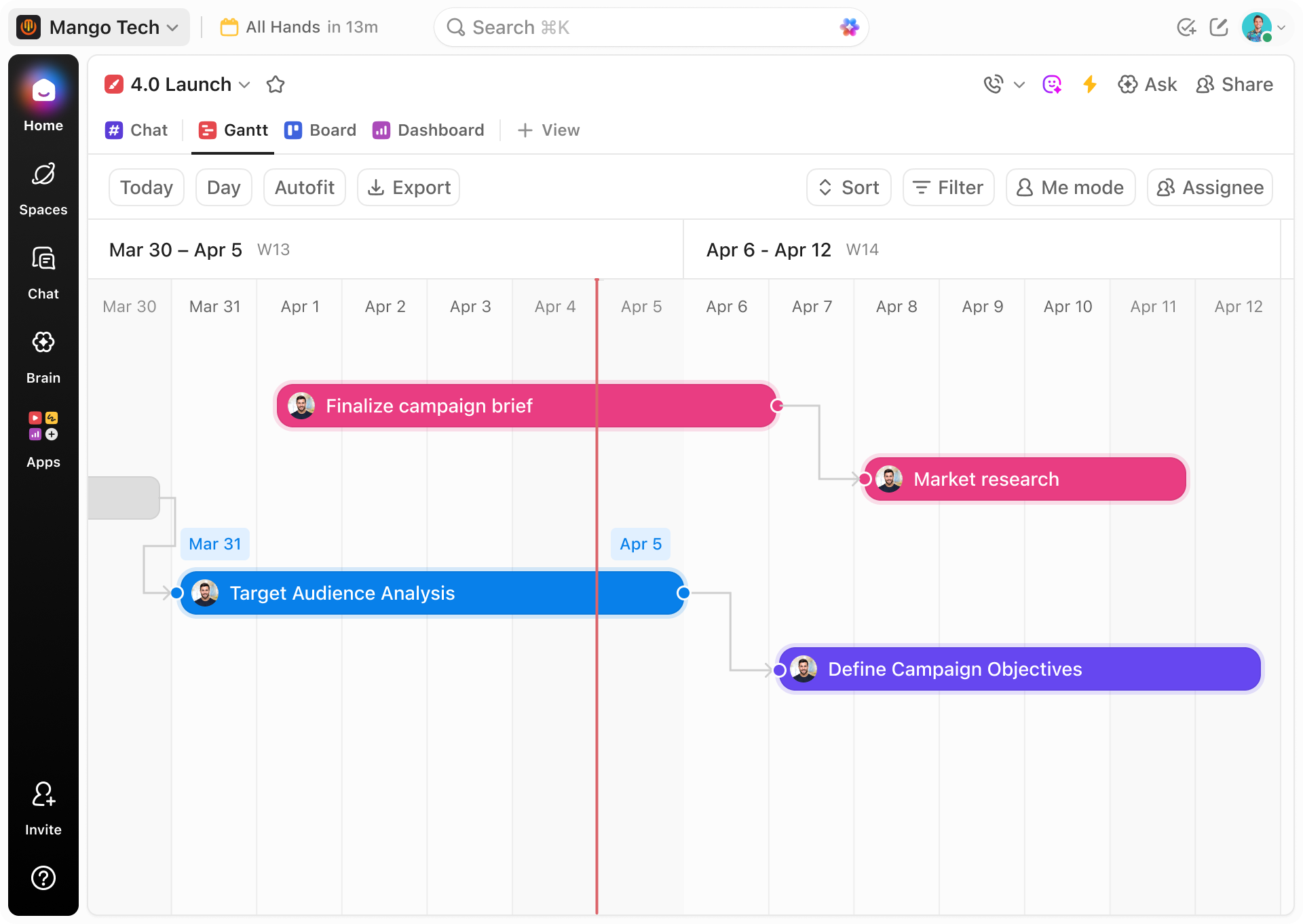
How Gantt Chart Software Empowers Biomedical Technicians
Gain a clear overview of your equipment lifecycle and coordinate maintenance with precision.
#UseCase1
Visualizing Preventive Maintenance Schedules
ClickUp’s Gantt charts help you map recurring maintenance activities, ensuring no equipment is overlooked or serviced late.
#UseCase2
Managing Calibration Timelines
Track calibration due dates and dependencies to maintain accurate diagnostic performance and compliance.
#UseCase3
Coordinating Repair and Replacement Tasks
Assign repair jobs, sequence tasks, and monitor progress to minimize equipment downtime.
#UseCase4
Tracking Regulatory Compliance Deadlines
Map inspection and certification dates with automated reminders to stay audit-ready.
#UseCase5
Allocating Tools and Parts Effectively
Visualize resource availability to prevent scheduling conflicts and optimize inventory use.
#UseCase6
Reporting Equipment Status to Stakeholders
Use Gantt views to generate clear progress reports for management and regulatory bodies.
#UseCase7
Integrating ClickUp Brain for Smart Task Suggestions
Leverage AI-powered insights to predict maintenance needs and optimize scheduling.
#UseCase8
Streamlining Team Communications
Keep conversations, notes, and files linked directly to tasks, reducing information loss.
#UseCase9
Enhancing Workflow with Brain Max Automation
Automate task dependencies and reminders, freeing your team to focus on critical repairs.
Key Beneficiaries
Who Gains the Most from ClickUp’s Gantt Chart Software
Perfect for biomedical technicians juggling multiple devices, compliance standards, and team coordination.
If you supervise hospital equipment across departments
ClickUp synchronizes tasks and timelines across units, ensuring consistent maintenance and quick issue resolution.

If you manage calibration and quality assurance
Use Gantt charts to track calibration cycles, certificate renewals, and audit preparations seamlessly.

If you coordinate repair teams and vendors
Assign clear tasks, monitor progress, and align schedules to reduce equipment downtime effectively.

How ClickUp Supports Biomedical Technicians
Step-by-Step: Streamlining Equipment Maintenance with ClickUp Gantt
Harness the full power of ClickUp Gantt Charts tailored for biomedical workflows
Centralize Equipment Records
Link manuals, maintenance logs, and calibration certificates directly to tasks for easy access.
Plan Maintenance Cycles Visually
Schedule recurring tasks with dependencies to avoid missed or premature servicing.
Standardize Procedures with Templates
Create task templates for common maintenance routines to ensure consistency and quality.
Coordinate Team Assignments
Clearly assign roles and deadlines to technicians, vendors, and quality officers.
Turn Inspections into Actionable Tasks
Convert audit findings into prioritized tasks with deadlines on your Gantt timeline.
Automate Alerts and Follow-Ups
Use Brain Max to trigger reminders and automate task progression, minimizing manual oversight.