Empower Your Life Sciences Teams with Industry-Leading CRM Software

Why Life Sciences Organizations Rely on Specialized CRM Solutions
Managing collaborations in life sciences without a dedicated CRM is like navigating clinical data without proper protocols — errors and delays are inevitable.
Common challenges without a CRM include:
- Fragmented partner communications — losing track of research agreements and clinical contacts.
- Complex trial pipelines lacking transparency — missing enrollment milestones and regulatory deadlines.
- Inefficient subject recruitment processes — scattered outreach and inconsistent documentation.
- Stalled industry partnerships — unclear next steps and no centralized follow-up system.
- Regulatory feedback buried in emails — difficult to audit and respond effectively.
- Communications scattered across multiple platforms — obstructing historical context and compliance.
- External vendors and CROs poorly tracked — risking delays and budget overruns.
- New team members onboard without project history — causing knowledge gaps and duplicated efforts.
Why ClickUp CRM Outperforms Conventional Life Sciences Management Tools
Traditional Methods
- Contacts spread across emails and spreadsheets
- No consolidated communication logs
- Trial pipelines tracked manually
- Subject recruitment lacks automation
- Regulatory feedback lost in inboxes
- Limited visibility into partner progress
- Documents scattered across drives
- No automated reminders
ClickUp CRM Advantages
- Centralize all collaborators, trial subjects, and vendors
- Log calls, emails, decisions, and documents in organized timelines
- Visualize clinical trial stages with customizable pipelines
- Automate recruitment outreach and follow-ups
- Link regulatory feedback directly to tasks and timelines
- Dashboards provide real-time progress and ownership
- Store all essential documents attached to CRM records
- Automatic reminders prevent missed deadlines
How CRM Software Accelerates Life Sciences Success
Centralizing Research Partners & Clinical Subjects
Managing Clinical Trial Pipelines Efficiently
Streamlining Subject Recruitment and Retention
Capturing Regulatory and Ethics Committee Feedback
Coordinating Multi-Site and Cross-Functional Teams
Tracking Vendor and CRO Communication
Monitoring Journal Editors and Peer Review Processes
Enhancing Industry Collaborations and Licensing Agreements
Transforming Meetings into Actionable Research Steps
Who Benefits Most from Life Sciences CRM Software
Designed for professionals managing complex collaborations, compliance, and communications in life sciences sectors.
For Global Research Consortia
ClickUp CRM creates a unified communications framework that bridges institutions, countries, and time zones seamlessly.
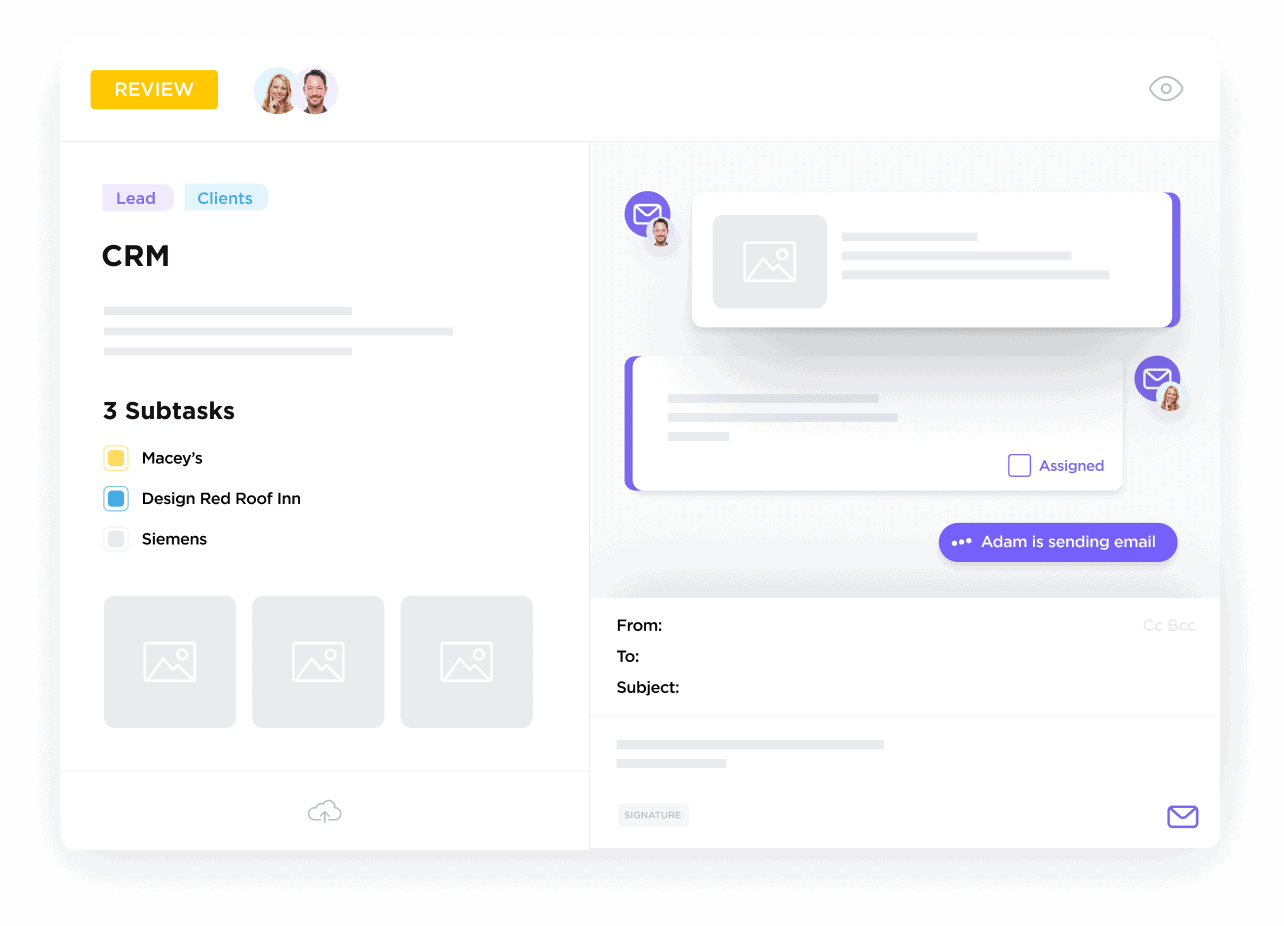
For Laboratory and Clinical Researchers
Effortlessly coordinate participants, vendors, collaborators, and facility resources to keep studies on track.
For Early-Career Scientists and Graduate Students
Manage supervisors, ethics committees, funding pipelines, and conference contacts all in one centralized workspace.

How ClickUp CRM Optimizes Life Sciences Operations
Create a Comprehensive Research Contact Hub
Organize collaborators, participants, vendors, reviewers, and partners with customizable fields and detailed activity histories.
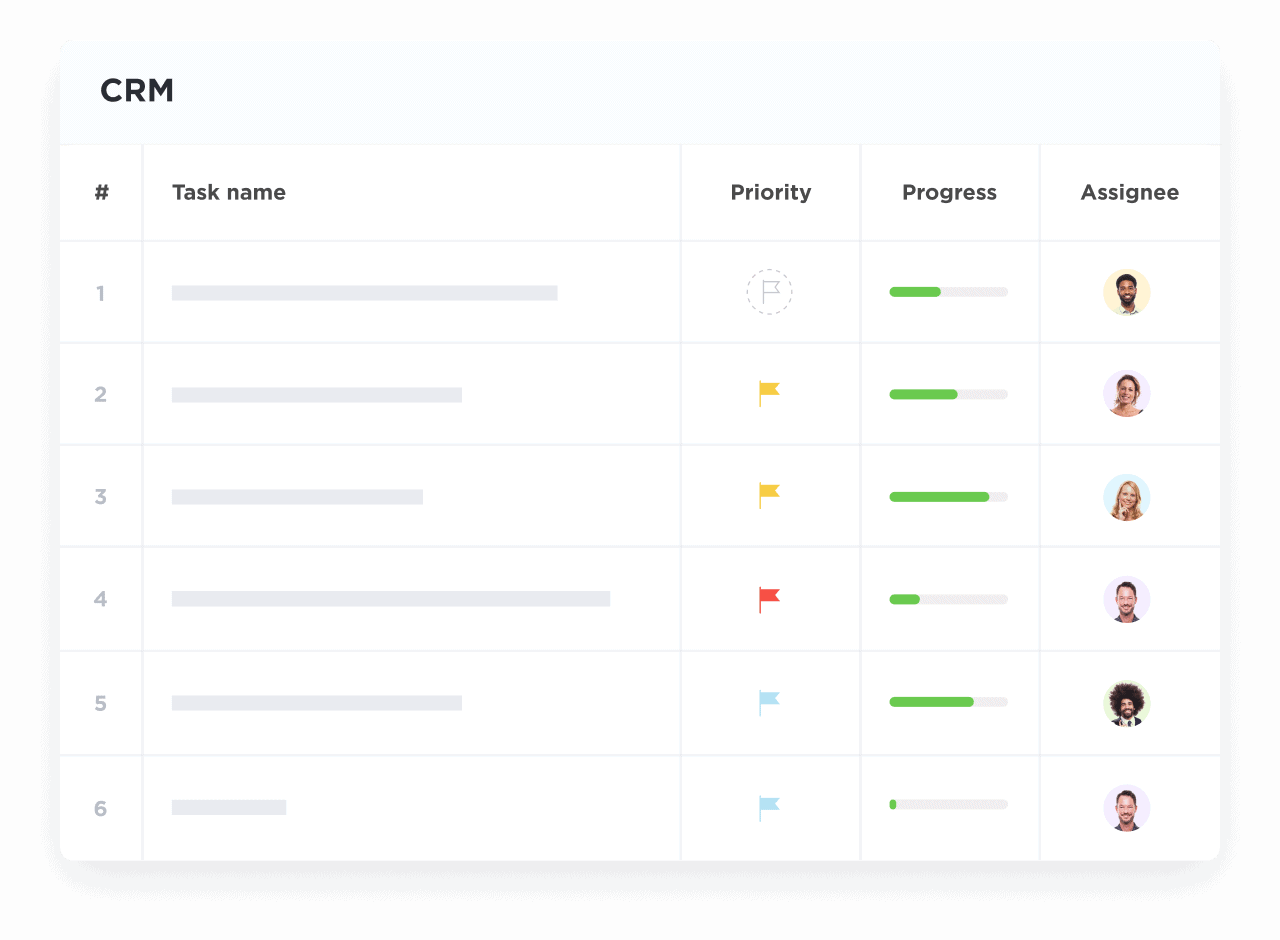
Visualize Complex Research Relationships
Map clinical trial stages, partnership milestones, recruitment progress, and collaboration status visually.
Maintain Complete Communication Records
Log emails, calls, decisions, and file attachments to preserve context and auditability.
Convert Conversations Into Action Items
Turn follow-ups into assigned tasks with due dates, automations, and notifications.
Link Documents and Protocols Directly
Attach consent forms, regulatory submissions, data agreements, and manuscripts directly to CRM entries.
Stay Alert with Real-Time Dashboards
Monitor outstanding actions, compliance checkpoints, and funding deadlines to ensure nothing is overlooked.