Streamline Clinical Operations with the Best CRM Software

Elevate Clinical Operations with a Purpose-Built CRM
Managing clinical operations without a CRM is like navigating complex trials blindfolded. Critical details slip through, timelines falter, and communication breaks down.
Common challenges faced without CRM software include:
- Fragmented patient and site information — lack of centralized data creates confusion and delays.
- Difficulty tracking regulatory submissions — missed deadlines and incomplete documentation risk compliance.
- Inefficient coordination across multidisciplinary teams — unclear roles and responsibilities cause bottlenecks.
- Unreliable monitoring of clinical trial progress — status updates scattered and inconsistent.
- Poor communication with external vendors and partners — no unified record of interactions or agreements.
- Onboarding new team members without context — no accessible history of project decisions or contacts.
- Manual follow-ups leading to missed actions — critical tasks fall through the cracks.
- Scattered data across emails, spreadsheets, and systems — hard to maintain audit trails and transparency.
Transform Clinical Management Beyond Traditional Methods
Traditional Methods
- Patient and site info scattered across spreadsheets and emails
- Manual tracking of regulatory documents
- Lack of real-time trial status updates
- No centralized communication log
- Missed follow-up reminders
- Difficulty coordinating cross-functional teams
- Data siloed in multiple systems
- Limited visibility into vendor performance
ClickUp CRM for Clinical Ops
- Consolidate patient, site, and vendor data in one platform
- Automate regulatory submission tracking with reminders
- Visualize trial progress with customizable dashboards
- Log all communications and decisions for audit readiness
- Automate follow-up tasks and deadlines
- Streamline team collaboration with task assignments and comments
- Integrate data sources for a unified view
- Monitor vendor contracts and performance metrics
Unlock Powerful CRM Use Cases for Clinical Operations Managers
Centralize Patient and Site Information
Streamline Regulatory Submission Tracking
Coordinate Multidisciplinary Teams with Transparency
Monitor Clinical Trial Milestones
Manage Vendor and Partner Communications
Automate Patient Recruitment and Follow-Up
Facilitate Training and Onboarding
Integrate Data for Comprehensive Reporting
Turn Meeting Notes into Actionable Tasks
Who Benefits Most from ClickUp CRM in Clinical Operations?
Global Clinical Trial Managers
Site Coordinators and Research Nurses
Regulatory Affairs Specialists

How ClickUp CRM Empowers Clinical Operations Managers
Build a Comprehensive Patient and Site Database
Centralize all contact info, documents, and activity logs for patients, clinical sites, and research partners.
Automate Regulatory Workflow Tracking
Set up pipelines and reminders for IRB submissions, protocol amendments, and safety reports.
Coordinate Team Tasks and Communication
Assign responsibilities, track progress, and maintain transparent communication channels.
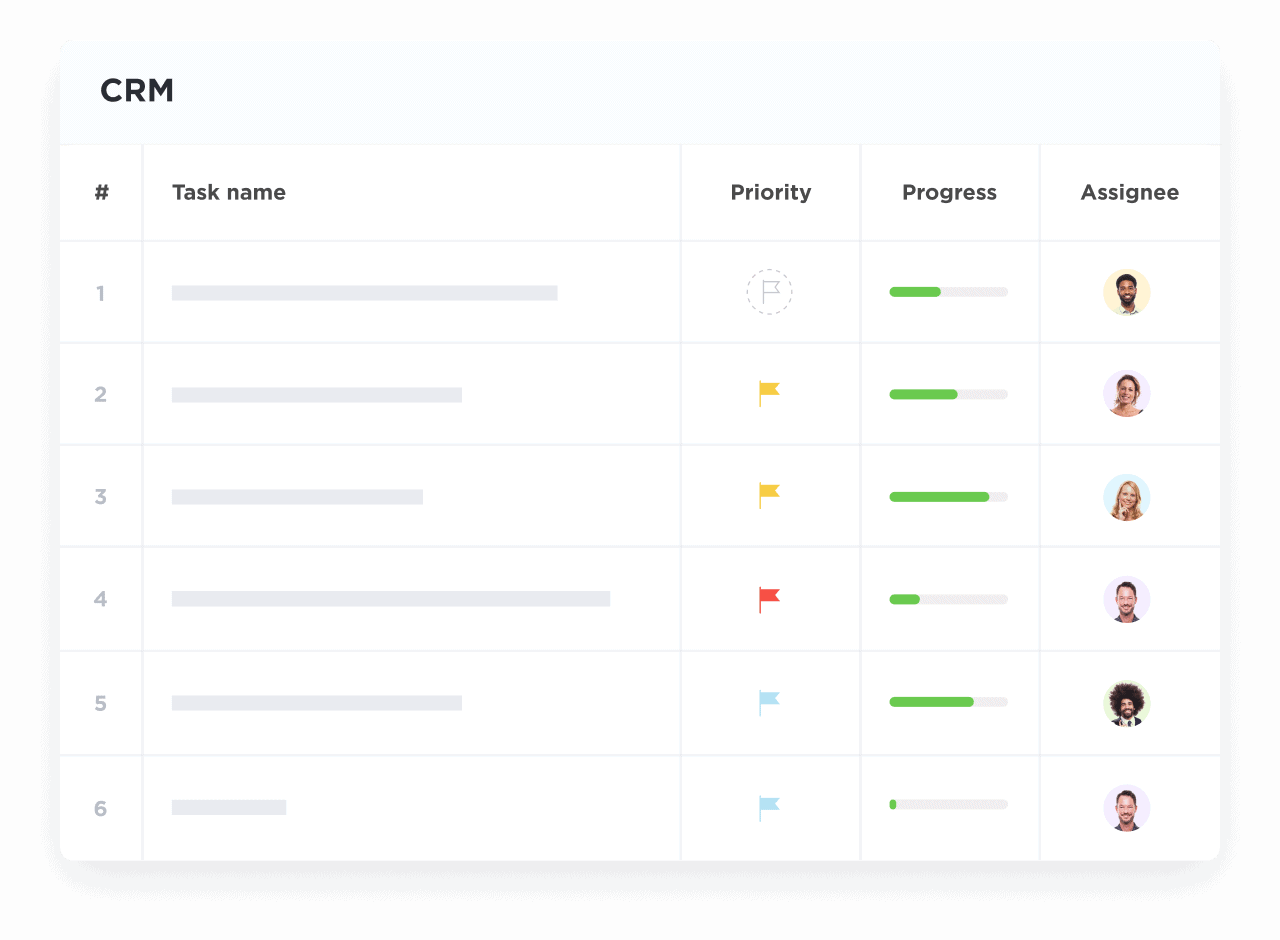
Visualize Trial Progress with Dashboards
Monitor enrollment, data collection, and milestone completion in real time.
Integrate External Systems and Documents
Attach lab results, monitoring reports, and consent forms directly to CRM records.
Ensure Timely Follow-Ups and Reporting
Use automated alerts and task assignments to keep the trial on schedule and audit-ready.