Elevate Clinical Data Management with ClickUp CRM

Why Clinical Data Managers Demand Specialized CRM Software
Handling clinical data without a dedicated CRM is like navigating a trial blindfolded. Missteps lead to costly delays and compliance risks.
Common pitfalls when clinical data managers rely on manual tracking:
- Data inconsistencies multiply — losing track of patient records, visit logs, or monitoring forms.
- Trial timelines slip — missed deadlines for data lock, monitoring visits, or regulatory submissions.
- Communication gaps widen — fragmented messages across emails, calls, and documents.
- Sponsor and CRO coordination falters — unclear responsibilities and no centralized status updates.
- Audit trails become incomplete — difficult to reconstruct data queries, resolutions, or protocol deviations.
- Onboarding new team members is inefficient — absence of a unified knowledge base and contact history.
- Vendor and site management is scattered — tracking shipments, invoices, and site communications is error-prone.
- Regulatory compliance risks increase — no centralized repository for essential documentation and approvals.
Unlock Precision: Why Traditional Methods Fall Short for Clinical Data Management
Conventional Tracking
- Patient info scattered across spreadsheets and emails
- No comprehensive communication logs
- Missed trial milestones and data lock dates
- Manual follow-ups prone to oversight
- Limited visibility into site or vendor status
- Documents housed in multiple unlinked locations
- No automated alerts for critical events
- Difficulty maintaining audit-ready records
ClickUp CRM
- Centralized patient, site, and vendor database
- Comprehensive logs of calls, emails, and data queries
- Visual timelines for trial phases and milestones
- Automated reminders for follow-ups and deadlines
- Linked documents and real-time status dashboards
- Streamlined collaboration between sponsors, CROs, and sites
- Robust audit trails for compliance
- Integration with ClickUp Brain for predictive insights and workflow optimization
How CRM Software Empowers Clinical Data Managers
Centralize Patient & Site Information
Manage Clinical Trial Milestones
Streamline Data Query Resolution
Coordinate Sponsor & CRO Communications
Oversee Vendor & Supply Chain Interactions
Maintain Regulatory Compliance Records
Track Monitoring Visits & Reports
Facilitate Team Onboarding & Training
Automate Follow-Up Actions from Meetings
Who Benefits Most from ClickUp CRM in Clinical Data Management
If You Coordinate Global Clinical Trials
If You Manage Site Data and Vendor Relations
If You’re a Clinical Data Manager Leading Protocol Compliance

Simplify Clinical Data Management with ClickUp CRM
Create a Comprehensive Trial Contact Directory
Store investigators, coordinators, monitors, vendors, and regulatory contacts with custom fields and interaction history.
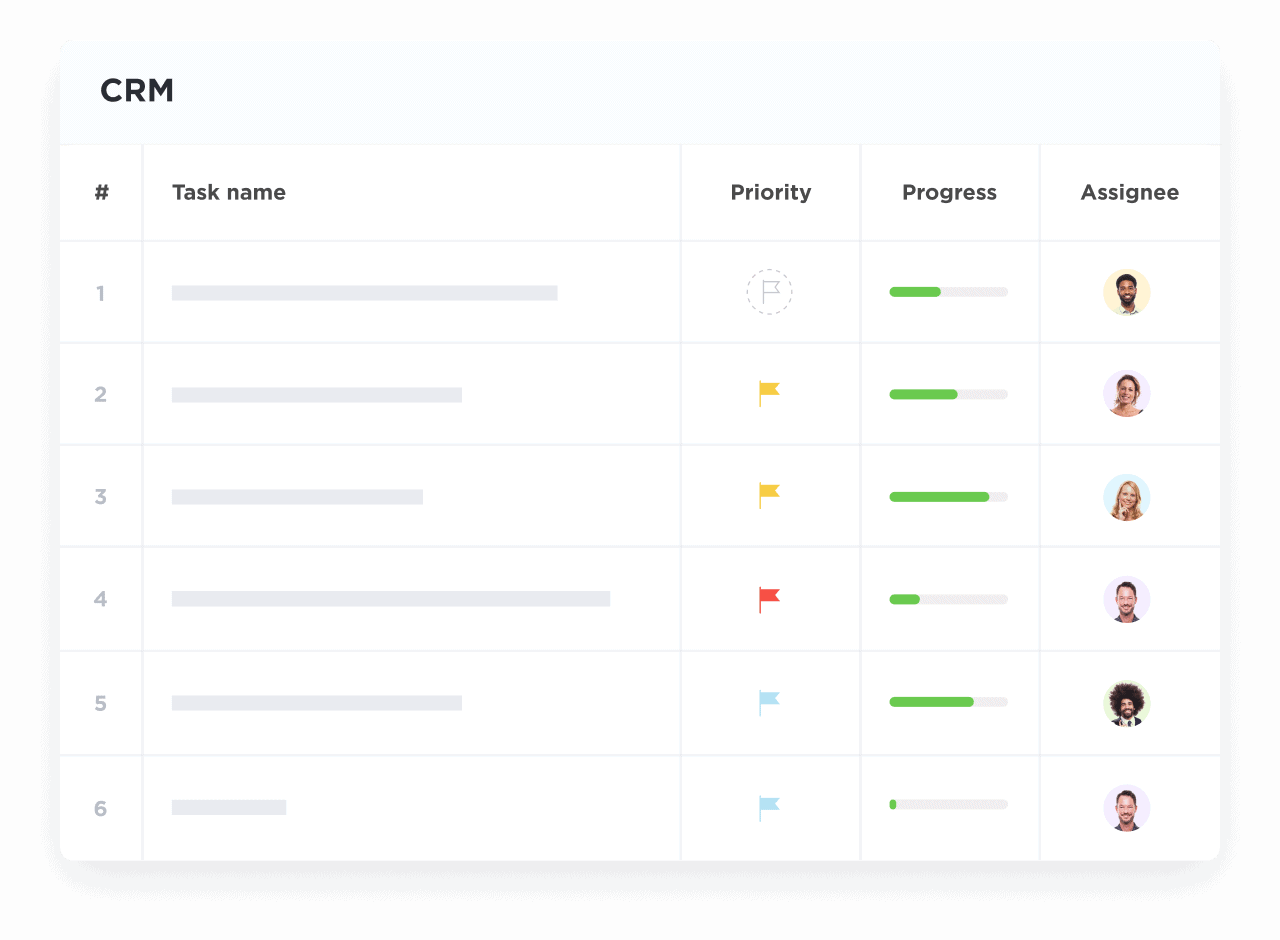
Visualize Trial Progress and Milestones
Map enrollment, data collection, monitoring, and submission stages with interactive dashboards.
Log Communication and Data Queries
Capture all correspondence and issue resolutions to maintain audit-ready documentation.
Turn Discussions into Trackable Tasks
Assign follow-ups with clear ownership, deadlines, and automated reminders.
Attach Critical Documents Securely
Link protocols, consent forms, monitoring reports, and regulatory submissions directly to CRM records.
Stay Ahead with Proactive Alerts
Dashboards and notifications ensure no data lock, audit, or compliance deadline is ever missed.