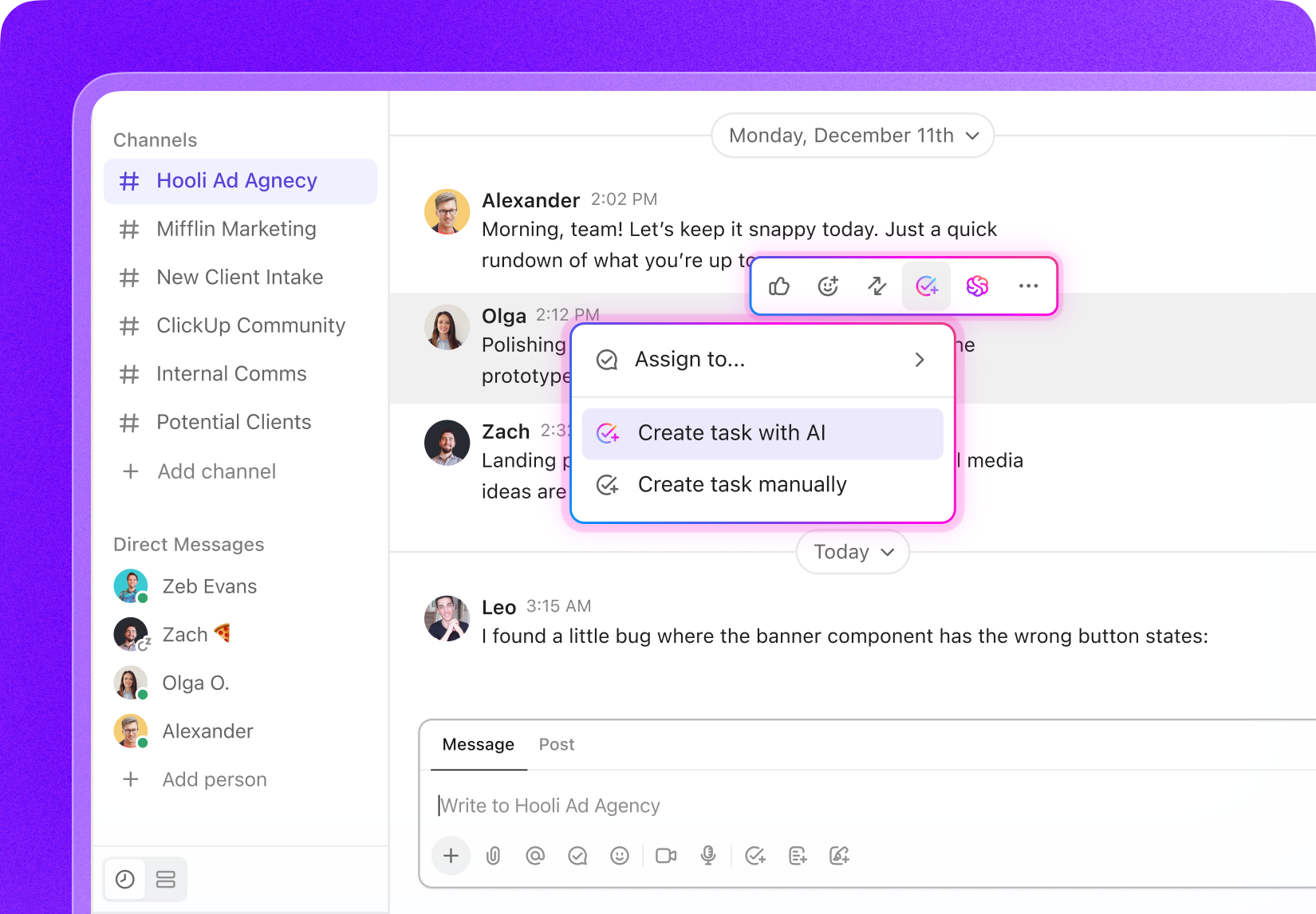
Task Software for Drug Safety Associates
Task Management Software Designed for Drug Safety Associates
Organize your pharmacovigilance activities, monitor safety case workflows, collaborate effortlessly with your team, and maintain full oversight of every compliance step.
Trusted by the best
Why You Need Specialized Task Management
The Challenge of Managing Drug Safety Tasks Efficiently
Without a dedicated system, managing drug safety tasks leads to missed case updates, scattered reports, and regulatory delays — making compliance stressful and error-prone.
- Safety reports get delayed — tracking case progress manually causes bottlenecks and late submissions.
- Adverse event documentation scatters — data spread across emails, spreadsheets, and notes increases risk of loss.
- Regulatory deadlines slip by — without automated reminders, critical timelines are often overlooked.
- Cross-team communication falters — unclear handoffs between pharmacovigilance, medical, and regulatory teams slow processes.
- Quality audits become daunting — lack of centralized documentation complicates readiness and transparency.
- Compliance oversight is limited — without real-time visibility, potential risks can go unnoticed.
- Resource allocation is inefficient — manual tracking leads to duplicated efforts and missed priorities.
- Case follow-ups lack consistency — varied workflows result in protocol deviations and data inconsistencies.
Traditional Methods vs ClickUp
Why Conventional Drug Safety Tools Fall Short
Discover how ClickUp elevates your pharmacovigilance workflow beyond traditional approaches.
Conventional Tools
- Case updates scattered in emails and spreadsheets
- Safety data fragmented across systems
- Manual tracking prone to errors and delays
- Limited collaboration with unclear role assignments
- Regulatory deadlines managed with calendar alerts only
- Documentation dispersed in multiple folders
ClickUp Task Management
- Centralized case tracking with real-time status updates
- Integrated safety data and reports linked to tasks
- Automated workflows reduce manual errors
- Defined ownership with transparent collaboration
- Smart reminders and synchronized calendars for deadlines
- Searchable, attached documents accessible to the team
Use Cases
Unlock How Task Management Software Empowers Drug Safety Associates
Explore how ClickUp simplifies pharmacovigilance workflows and enhances compliance accuracy.
#UseCase1
Centralizing Safety Case Data Across Teams
All adverse event reports, follow-ups, and source documents remain in one searchable location linked to respective cases for easy access and audit readiness.
#UseCase2
Maintaining a Complete Audit Trail for Regulatory Compliance
ClickUp logs every update, comment, and file attachment, ensuring full traceability from case intake through closure for inspections.
#UseCase3
Capturing Evolving Regulatory Feedback Seamlessly
Comments and change requests from regulatory reviewers are tracked with version history and clear action items to ensure nothing is missed.
#UseCase4
Preventing Deviations in Case Processing Protocols
Built-in templates and checklists guide associates through standardized workflows, reducing errors and ensuring consistency.
#UseCase5
Tracking Safety Reporting Deadlines and Submission Milestones
Automated reminders and visual timelines help keep all reporting obligations on schedule, avoiding regulatory penalties.
#UseCase6
Managing Complex Signal Detection and Risk Assessment Tasks
Custom fields and dependencies allow systematic tracking of signal validation steps, risk evaluation, and mitigation planning.
#UseCase7
Coordinating Cross-Functional Pharmacovigilance Teams
Assign tasks clearly among medical reviewers, data managers, and compliance officers to optimize collaboration and workflow transparency.
#UseCase8
Avoiding Duplicate Case Entries and Data Overload
ClickUp’s tagging and filtering prevent redundant work by highlighting case ownership and status at a glance.
#UseCase9
Transforming Regulatory Meetings into Clear Action Plans
Meeting notes convert directly into assigned tasks with deadlines, ensuring follow-ups are tracked and completed efficiently.
Key Beneficiaries
Who Benefits the Most from ClickUp Task Management
Ideal for drug safety associates and pharmacovigilance professionals seeking streamlined operations and compliance assurance.
If You’re a Drug Safety Associate
ClickUp helps you monitor case progress, manage adverse event reports, and meet submission deadlines without juggling multiple tools.
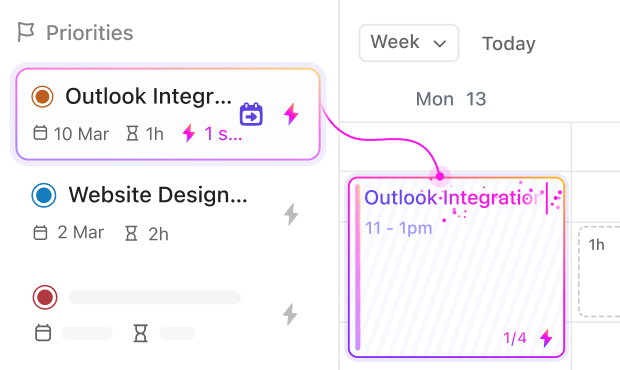
If You’re a Pharmacovigilance Manager
Oversee team workflows, standardize procedures, and ensure regulatory compliance effortlessly with centralized task tracking.
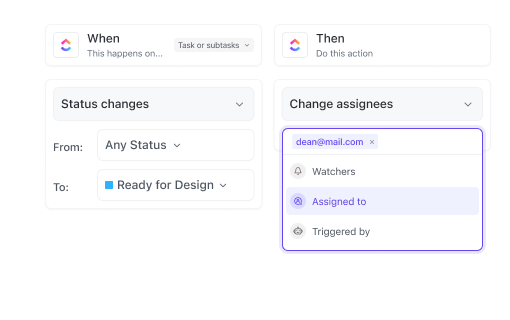
If You Coordinate Global Safety Operations
Coordinate cross-border case processing, share updates in real-time, and maintain consistent standards across regions using ClickUp.
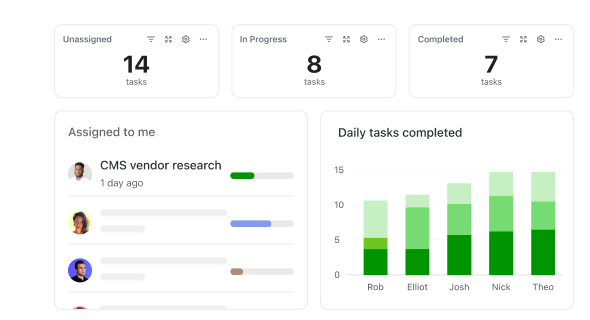
How ClickUp Supports Drug Safety
How ClickUp Streamlines Every Stage of Pharmacovigilance
Manage safety cases, compliance tasks, and team collaboration all in one platform.
Centralize Everything
Store literature, datasets, protocols, drafts, and grant docs in one workspace — no more scattered files.
Plan Research in Phases
Break projects into proposal, literature review, experiments, analysis, and writing with task lists and Gantt timelines.
Standardize Experiments & Fieldwork
Use templates and checklists for repeatable, error-free lab or field procedures.
Collaborate Across Teams
Assign tasks to co-authors, lab members, or collaborators. Shared boards and dashboards keep everyone aligned.
Turn Meetings Into Actionable Tasks
Convert supervisor or lab meetings into tasks with owners, checklists, and deadlines.
Stay on Top of Deadlines & Funding
Track grants, conferences, and submissions with automated reminders and calendars.