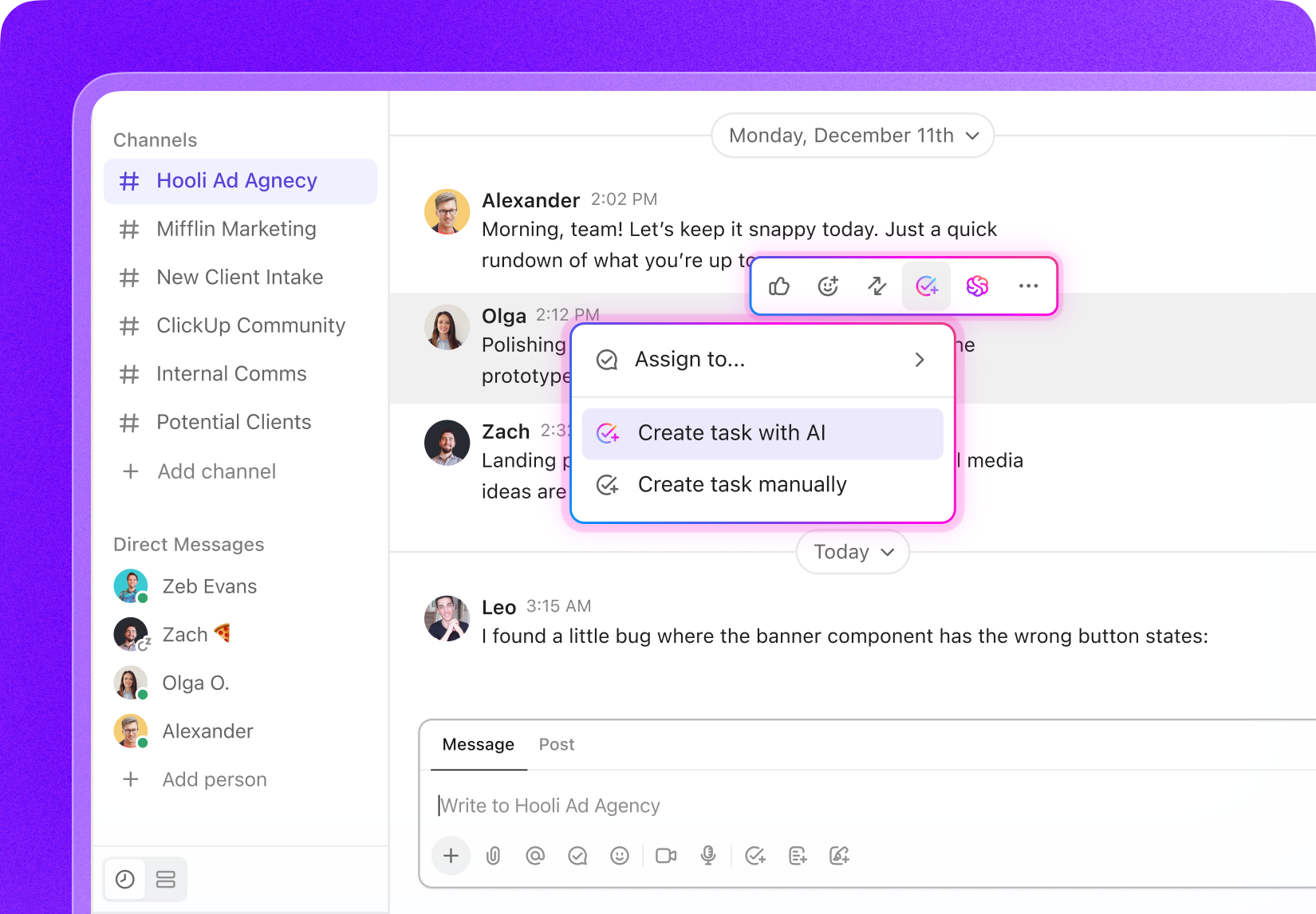
Task Management for Clinical Trial Managers
Task Management Software Tailored for Clinical Trial Management
Organize your trial tasks, monitor milestone progress, collaborate effortlessly with your team, and maintain full transparency throughout each phase of your clinical trials.
Trusted by the best
The Challenge of Managing Trials
Why Clinical Trial Managers Benefit from Dedicated Task Management
Handling complex clinical trials without an organized system results in missed deadlines, fragmented data, and operational confusion — turning essential tasks into overwhelming challenges.
- Multi-site coordination complicates communication — making it tough to track progress and align teams.
- Regulatory documentation piles up quickly — increasing the risk of errors and non-compliance.
- Patient recruitment and retention become unpredictable — without clear tracking, timelines slip.
- Protocol deviations go unnoticed — inconsistent monitoring leads to compliance issues.
- Data collection suffers from inconsistency — scattered notes and reports delay insights.
- Timelines blur across phases — making it difficult to see what’s completed or pending.
- Communication breakdowns cause delays — emails and informal messages scatter crucial information.
- Resource allocation is inefficient — equipment and staff scheduling conflicts slow progress.
Traditional Approaches vs ClickUp
Why Conventional Trial Management Falls Short
Discover how ClickUp delivers clarity and control where traditional methods falter.
Conventional Methods
- Tasks dispersed across spreadsheets, emails, and memory
- Regulatory documents hard to track and update
- Manual monitoring prone to errors
- Communication scattered among multiple channels
- Deadlines and milestones easily overlooked
- Data files spread across disconnected platforms
ClickUp Task Management
- Unified task tracking with clear statuses and priorities
- Centralized document storage with version control
- Automated reminders for milestones and compliance
- Real-time collaboration with clear ownership
- Integrated timelines and calendar syncing
- Searchable records tied to specific trial tasks
Use Cases
Unlocking the Power of Task Management for Clinical Trial Success
See how streamlined task tracking reduces errors and keeps trials on schedule.
#UseCase1
Consolidating Trial Data and Communications
Keep patient data, regulatory documents, and team notes organized in one place — accessible to all stakeholders exactly when needed.
#UseCase2
Maintaining a Clear Audit Trail for Regulatory Compliance
Ensure every task, approval, and update is logged with timestamps and comments, simplifying audits and inspections.
#UseCase3
Managing Protocol Amendments Seamlessly
Track changes and communicate updates instantly with version control and real-time notifications.
#UseCase4
Preventing Recruitment and Retention Delays
Monitor patient enrollment and follow-ups with automated checklists and reminders to keep trials on track.
#UseCase5
Coordinating Multi-Site Trial Activities Efficiently
Assign and track tasks across sites with clear ownership and status updates to avoid overlaps and gaps.
#UseCase6
Streamlining Data Collection and Query Resolution
Use customizable workflows to manage data queries and ensure timely resolution and data integrity.
#UseCase7
Meeting Submission Deadlines with Confidence
Track regulatory submissions and ethics approvals with automated alerts and visual timelines.
#UseCase8
Avoiding Documentation Errors and Omissions
Attach all forms and reports directly to tasks to ensure completeness and easy access during reviews.
#UseCase9
Converting Meetings into Actionable Trial Milestones
Transform discussion points into assigned tasks with deadlines to accelerate decision-making and follow-up.
Key Beneficiaries
Who Gains the Most from ClickUp in Clinical Trials
Designed for clinical trial managers seeking full control over complex study workflows.
If you manage patient recruitment
ClickUp helps you track enrollment targets, follow-up schedules, and retention strategies without losing sight of individual participant status.
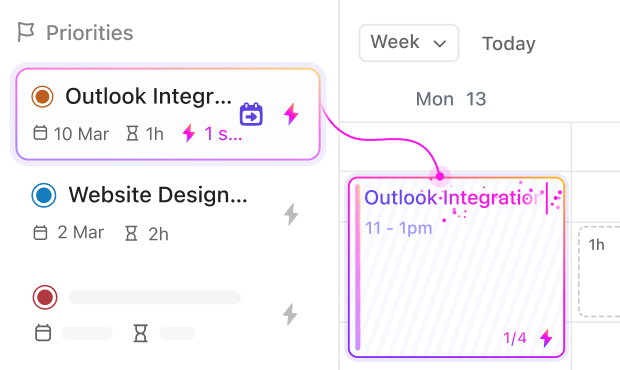
If you oversee multi-site coordination
ClickUp lets you assign tasks across locations, monitor site progress, and synchronize communication to prevent scheduling conflicts.
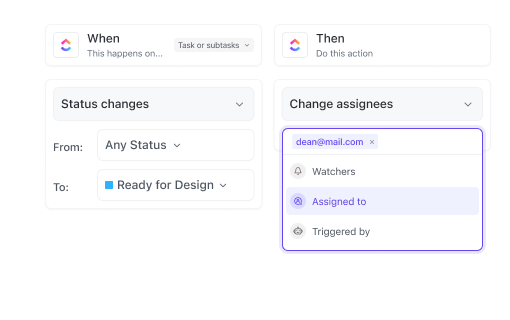
If you ensure regulatory compliance
ClickUp organizes protocols, amendments, and submissions with automated reminders and audit-ready documentation.
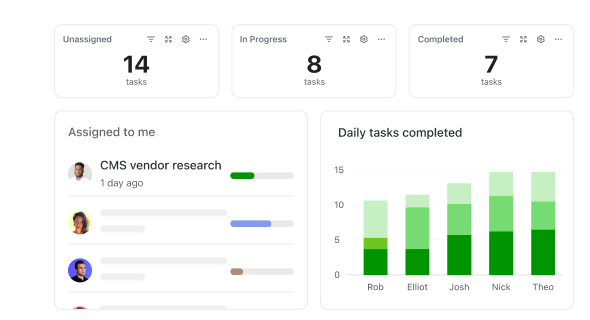
How ClickUp Supports Clinical Trial Management
Step-by-Step Guidance to Simplify Your Trial Workflow
Manage patient data, regulatory steps, and team tasks all within one platform.
Centralize Everything
Store literature, datasets, protocols, drafts, and grant docs in one workspace — no more scattered files.
Plan Research in Phases
Break projects into proposal, literature review, experiments, analysis, and writing with task lists and Gantt timelines.
Standardize Experiments & Fieldwork
Use templates and checklists for repeatable, error-free lab or field procedures.
Collaborate Across Teams
Assign tasks to co-authors, lab members, or collaborators. Shared boards and dashboards keep everyone aligned.
Turn Meetings Into Actionable Tasks
Convert supervisor or lab meetings into tasks with owners, checklists, and deadlines.
Stay on Top of Deadlines & Funding
Track grants, conferences, and submissions with automated reminders and calendars.