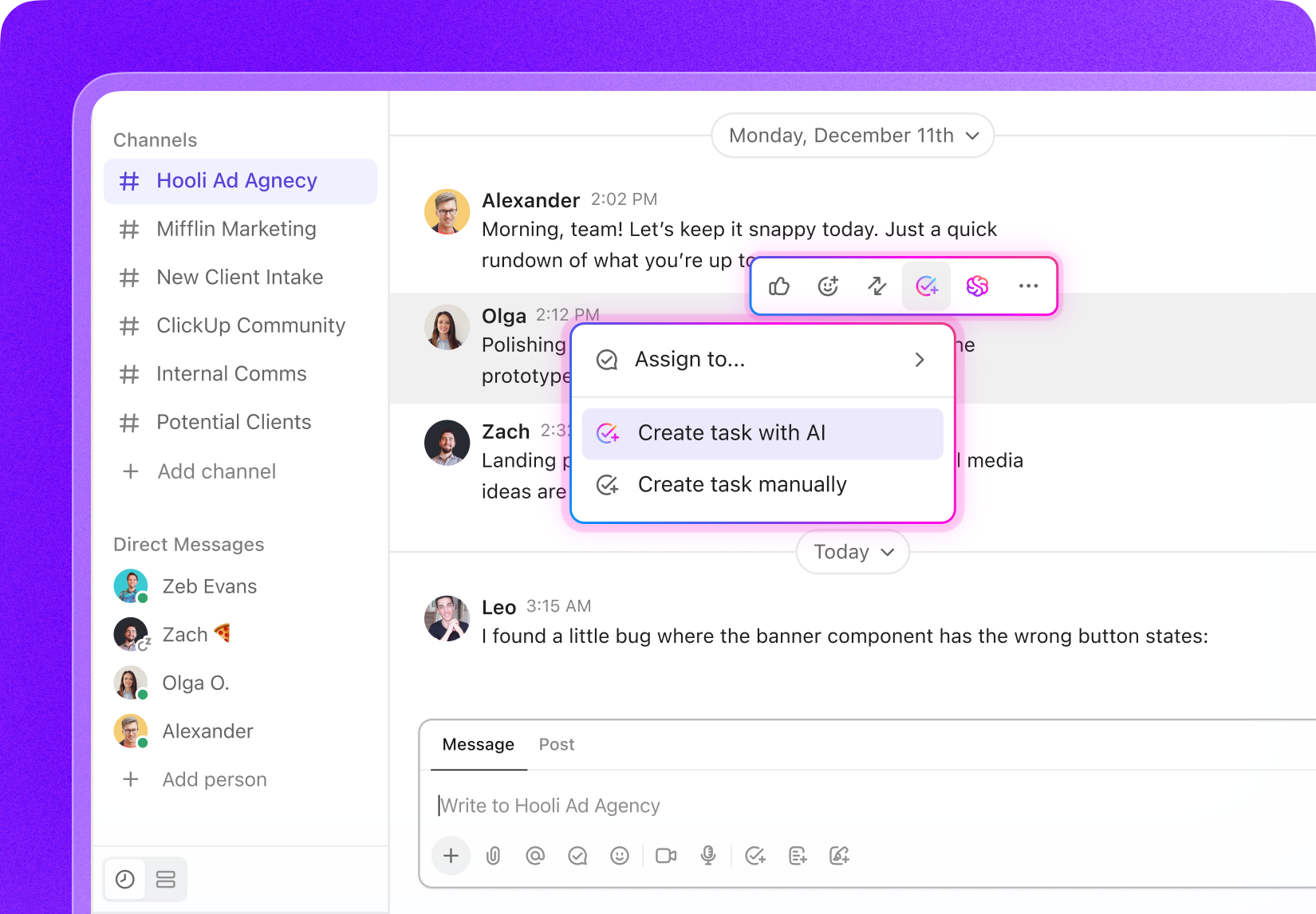
Task Management for Clinical Operations
Tailored Task Management Software for Clinical Operations Managers
Organize clinical workflows, monitor trial milestones, collaborate across teams, and maintain full transparency at every phase of your clinical studies.
Trusted by the best
The Challenge of Managing Clinical Tasks
Why Clinical Operations Managers Need Dedicated Task Management Software
Handling clinical operations without a unified system leads to missed deadlines, fragmented data, and operational inefficiencies — making complex trials more challenging than they need to be.
- Trial timelines become unclear — leading to delayed patient enrollment and study milestones.
- Documentation gets scattered — critical regulatory and patient data spread across platforms.
- Compliance risks increase — inconsistencies in protocol adherence and reporting.
- Team collaboration falters — unclear task ownership and communication gaps slow progress.
- Deadlines for submissions and audits slip — risking penalties and study delays.
- Progress tracking lacks visibility — making it tough to identify bottlenecks or delays.
- Resource scheduling conflicts arise — impacting site availability and staffing.
- Manual coordination leads to errors — increasing operational overhead and risk.
Traditional Methods vs ClickUp
Why Conventional Approaches Falter in Clinical Operations
Discover how ClickUp clarifies workflows where traditional tools fall short.
Conventional Tools
- Tasks dispersed across emails, spreadsheets, and paper logs
- Regulatory documents managed manually with high error potential
- Protocol deviations tracked inconsistently
- Communication fragmented across teams
- Submission deadlines often overlooked
- Data and files stored in disconnected systems
ClickUp Task Management
- Centralized task management with clear priorities and statuses
- Structured documentation with attachments and version control
- Built-in protocol checklists and compliance tracking
- Real-time collaboration and task ownership clarity
- Automated reminders for key deadlines and audits
- Unified repository for all clinical trial data and communications
Use Cases
How Task Management Software Empowers Clinical Operations Managers
See how task organization reduces errors, improves compliance, and accelerates clinical trials.
#UseCase1
Centralizing Trial Documentation and Protocols
Keep all study documents, protocols, and regulatory files in one searchable, accessible location linked directly to relevant tasks.
#UseCase2
Ensuring Protocol Compliance and Audit Readiness
Track adherence to study protocols with checklists, dependencies, and automated alerts to prevent deviations and maintain audit readiness.
#UseCase3
Coordinating Multisite Clinical Trial Teams
Manage tasks across sites with clear ownership, real-time updates, and collaborative communication to ensure alignment and timely execution.
#UseCase4
Streamlining Patient Enrollment and Visit Scheduling
Use task templates and reminders to keep patient recruitment, screening, and visits on track and well-documented.
#UseCase5
Tracking Regulatory Submissions and Approvals
Automate tracking of IRB submissions, amendments, and approvals with customized workflows and deadline notifications.
#UseCase6
Managing Resource Allocation and Equipment Availability
Schedule clinical staff, equipment, and site resources efficiently to avoid conflicts and ensure smooth operations.
#UseCase7
Facilitating Data Collection and Quality Control
Centralize data entry tasks and quality checks with status tracking and collaboration tools to enhance data integrity.
#UseCase8
Reducing Manual Errors with Standardized Procedures
Leverage templates and checklists to standardize trial processes, minimizing human error and ensuring consistency.
#UseCase9
Converting Meetings into Clear Action Plans
Turn team discussions and sponsor meetings into actionable tasks with assigned owners and deadlines to maintain momentum.
Key Beneficiaries
Who Benefits Most from ClickUp in Clinical Operations
For clinical operations managers and teams seeking a single source of truth for their trials.
If you’re a Clinical Operations Manager
ClickUp helps you oversee trial progress, coordinate teams, and ensure compliance without juggling multiple systems.
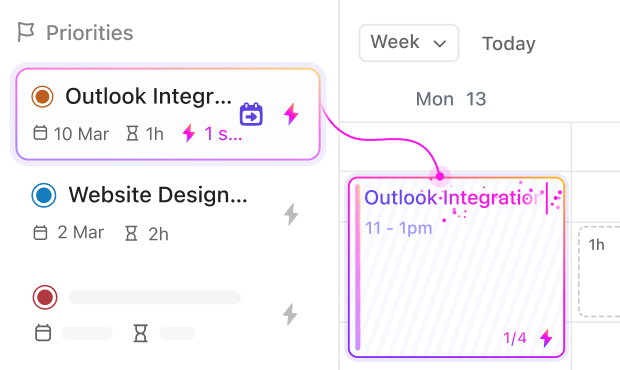
If you’re a Clinical Research Associate
Track monitoring visits, document findings, and manage follow-ups efficiently with structured task workflows.
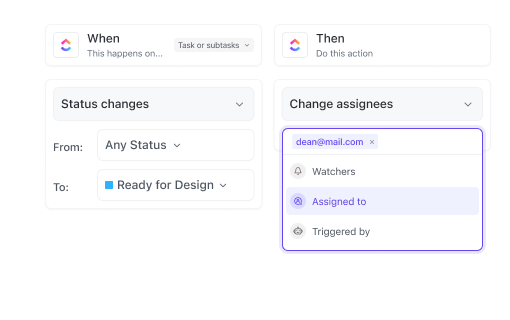
If you’re a Regulatory Affairs Specialist
Organize submission deadlines, track approvals, and maintain audit-ready documentation effortlessly.
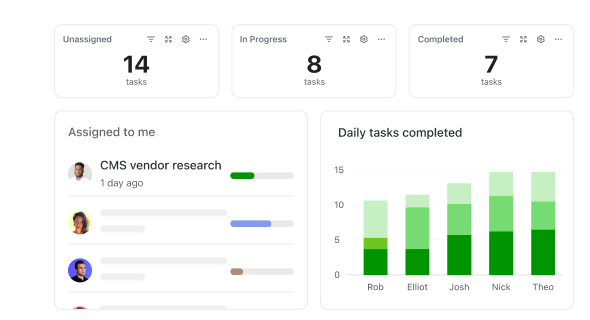
How ClickUp Supports Clinical Operations
How ClickUp Simplifies Clinical Trial Management
Manage protocols, teams, and deadlines without switching tools.
Centralize Everything
Store literature, datasets, protocols, drafts, and grant docs in one workspace — no more scattered files.
Plan Research in Phases
Break projects into proposal, literature review, experiments, analysis, and writing with task lists and Gantt timelines.
Standardize Experiments & Fieldwork
Use templates and checklists for repeatable, error-free lab or field procedures.
Collaborate Across Teams
Assign tasks to co-authors, lab members, or collaborators. Shared boards and dashboards keep everyone aligned.
Turn Meetings Into Actionable Tasks
Convert supervisor or lab meetings into tasks with owners, checklists, and deadlines.
Stay on Top of Deadlines & Funding
Track grants, conferences, and submissions with automated reminders and calendars.