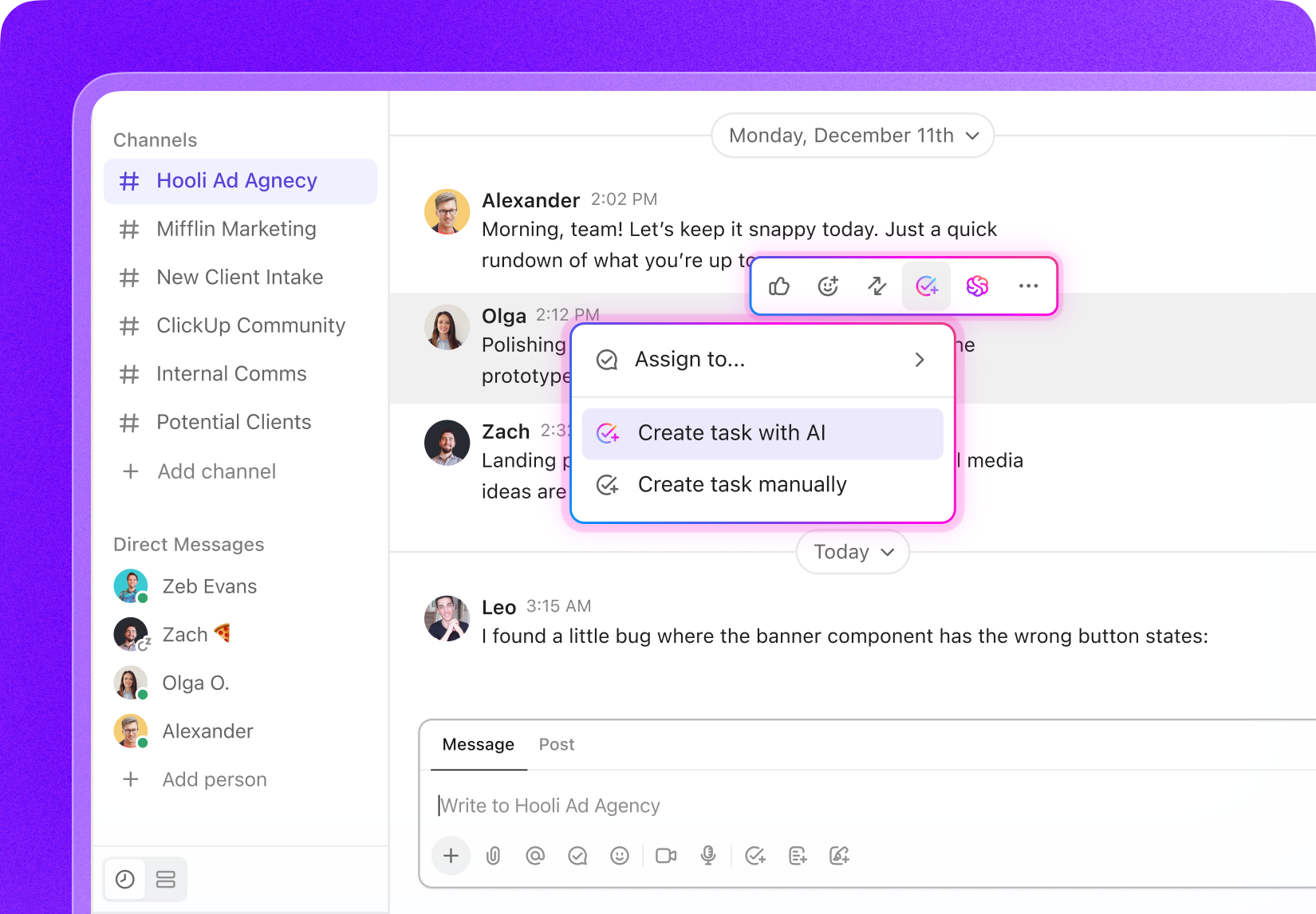
Task Management for Clinical Data Specialists
Tailored Task Management Software for Clinical Data Managers
Organize clinical trials, monitor data milestones, collaborate effortlessly with your team, and maintain full oversight of every phase of your clinical data projects.
Trusted by the best
The Need for Structure
Why Clinical Data Managers Benefit from Dedicated Task Management
Handling clinical data without a streamlined system creates risks of errors, scattered information, and workflow bottlenecks — making compliance and accuracy challenging.
- Trial timelines become unclear — difficult to pinpoint completed and pending data tasks, risking delays.
- Data discrepancies increase — uncoordinated datasets lead to mismatches and audit challenges.
- Documentation gets fragmented — critical files and notes are lost across platforms.
- Team communication falters — unclear responsibilities and updates cause confusion.
- Regulatory deadlines are missed — submissions and reporting fall behind without proper tracking.
- Monitoring progress feels uncertain — lack of visibility into data cleaning and validation stages.
- Resource allocation suffers — overlaps in personnel and tool usage slow trial progress.
- Quality assurance becomes inconsistent — tracking errors and corrective actions is cumbersome.
Challenges with Traditional Approaches
Why Conventional Clinical Data Management Falls Short
Discover how ClickUp’s task management bridges gaps traditional methods overlook.
Traditional Methods
- Data tasks dispersed across spreadsheets, emails, and notes
- Inconsistent tracking of data queries and cleaning
- Manual recording increases risk of errors
- Collaboration hindered by unclear task ownership
- Submission deadlines often overlooked
- Documents scattered in multiple storage locations
ClickUp Task Management
- Unified task lists with clear priorities and statuses
- Structured data query tracking and resolution workflows
- Automated reminders and audit trails for compliance
- Transparent ownership with real-time team updates
- Integrated calendars syncing all critical deadlines
- Centralized, searchable data and documentation attachments
Use Cases
Unlocking Efficiency: Task Management in Clinical Data Operations
See how task management reduces errors, enhances collaboration, and accelerates trial timelines.
#UseCase1
Consolidating Clinical Data Across Sites & Systems
ClickUp unites data entry, validation, and queries from multiple sources into one accessible place, improving accuracy and traceability.
#UseCase2
Maintaining a Clear Audit Trail for Regulatory Compliance
Every data correction and query is tracked with timestamps and comments, ensuring full transparency and inspection readiness.
#UseCase3
Managing Data Queries with Real-Time Updates
Track, assign, and resolve data queries efficiently with status updates and notifications that keep the whole team informed.
#UseCase4
Enforcing Protocol Adherence to Prevent Data Drift
Templates and checklists standardize data entry and monitoring, minimizing deviations and ensuring consistency throughout the trial.
#UseCase5
Tracking Ethics Approvals and Regulatory Submissions
Custom workflows and reminders help you stay ahead of IRB approvals, amendments, and submission deadlines without last-minute rushes.
#UseCase6
Coordinating Complex Data Cleaning and Validation Pipelines
Visual task dependencies and statuses map each step of data cleaning, preventing errors and ensuring timely completion.
#UseCase7
Meeting Submission Deadlines for Reports and Publications
Manage timelines for clinical study reports, journal submissions, and conference abstracts with Gantt views and centralized document storage.
#UseCase8
Avoiding Duplicate Data Reviews and Overlaps
Assign reviews and track progress with tagging and filters, so no dataset is reviewed twice and nothing is overlooked.
#UseCase9
Transforming Team Meetings Into Actionable Tasks
Capture meeting decisions and follow-ups as tasks with owners and deadlines to maintain momentum and accountability.
Key Beneficiaries
Who Gains Most from ClickUp in Clinical Data Management
For clinical data managers and teams seeking centralized control over complex trial data workflows.
If you’re a Clinical Data Manager
ClickUp helps you track data milestones, manage queries, and ensure regulatory compliance without juggling multiple tools.
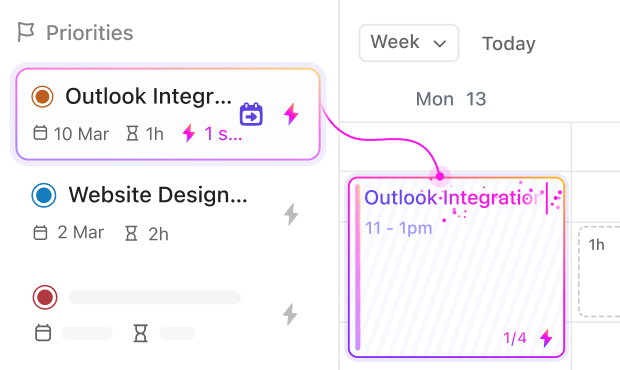
If you’re a Data Coordinator
Coordinate site data submissions, monitor progress, and communicate updates efficiently across all trial locations.
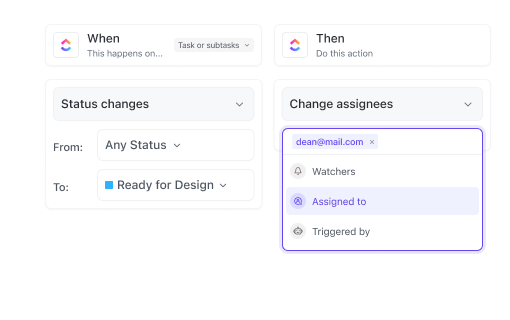
If you’re a Clinical Trial Lead
Oversee data timelines, resource allocation, and team collaboration to keep your trial on track and within scope.
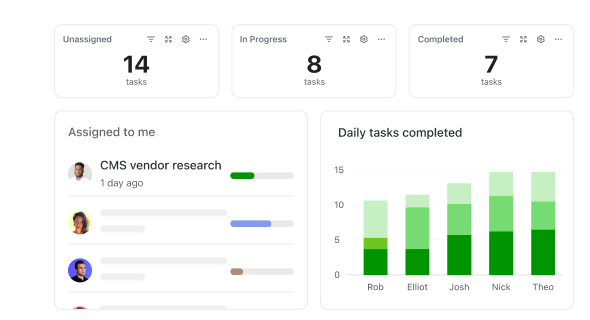
How ClickUp Enhances Clinical Data Workflows
Step-by-Step Support for Clinical Data Managers
Simplify complexity from site data capture to database lock with integrated tools.
Centralize Trial Documentation
Keep protocols, CRFs, audit logs, and data files all in one searchable space — reducing risk of lost information.
Plan and Track Data Milestones
Break down trials into phases with clear task lists and timeline views to monitor enrollment, data cleaning, and analysis.
Standardize Data Validation Processes
Use templates and automated checklists to ensure consistent data queries and validation across studies.
Facilitate Team Collaboration
Assign tasks, add comments, and share updates in real time to keep data managers, coordinators, and monitors aligned.
Convert Meetings Into Clear Action Items
Turn protocol discussions and team meetings into tasks with owners and deadlines to maintain accountability.
Stay Ahead of Regulatory Deadlines
Automate reminders and visualize submission timelines to avoid missing critical compliance dates.