GxP Compliance Knowledge Base Management
Build a Compliant Knowledge Base for GxP Excellence
Create a centralized, searchable repository for SOPs, validation protocols, audit trails, and regulatory updates—ensuring your quality system stays reliable and audit-ready.
Trusted by the best
ClickUp vs Legacy Systems
Why Choose ClickUp for Your GxP Compliance Knowledge Base
Consolidate compliance knowledge and streamline quality processes.
Using Traditional Compliance Tools
- Documentation scattered across multiple platforms risking version control issues
- Manual updates increase the chance of outdated procedures
- Compliance documents disconnected from execution and audit evidence
- Complex permission settings cause duplication and confusion
- Slow manual workflows delay critical compliance tasks
Using ClickUp for GxP Compliance
- Unified platform integrating documents, tasks, and communication
- Assign corrective actions directly from knowledge gaps
- Link SOPs and protocols to real-time compliance activities and audits
- Granular access controls for internal teams, partners, and auditors
- ClickUp Brain and AI accelerate content creation and continuous updates
Steps to Build Your Knowledge Base
How to Develop a GxP Compliance Knowledge Base with ClickUp
Follow this 6-step strategy to maintain a compliant, organized, and accessible knowledge repository.
1. Identify stakeholders and compliance objectives
- Define internal users, quality teams, and regulatory bodies
- Outline key compliance areas: SOPs, validation, CAPAs, audits
- Assign ownership to ensure accountability and consistency
2. Design a structured knowledge base framework
- Centralize documents with clear navigation: SOPs, protocols, training, audits
- Establish standardized templates for document types
- Ensure easy access to regulatory updates and change logs
3. Standardize document formats for regulatory consistency
- Use uniform templates covering purpose, scope, responsibilities, and procedures
- Include validation and audit trail requirements
- Detail deviations, corrective actions, and preventive measures
4. Integrate practical guides and compliance workflows
- Develop step-by-step instructions for common compliance tasks
- Embed risk assessments, CAPA tracking, and audit preparation
- Centralize training materials and competency checklists
5. Link documents to quality events and regulatory changes
- Connect SOP updates to audits, inspections, and CAPA cycles
- Automate notifications for document reviews and regulatory revisions
- Keep the knowledge base dynamic and reflective of current compliance status
6. Manage permissions and maintain ongoing reviews
- Control access by role: internal staff, partners, auditors
- Schedule periodic reviews and update cycles
- Collect feedback to continuously improve documentation quality
Leverage ClickUp for Robust Knowledge Management
Unlock GxP Compliance Success with ClickUp
Ensure your compliance documentation is well-structured, controlled, and aligned with every regulatory update.
Framework
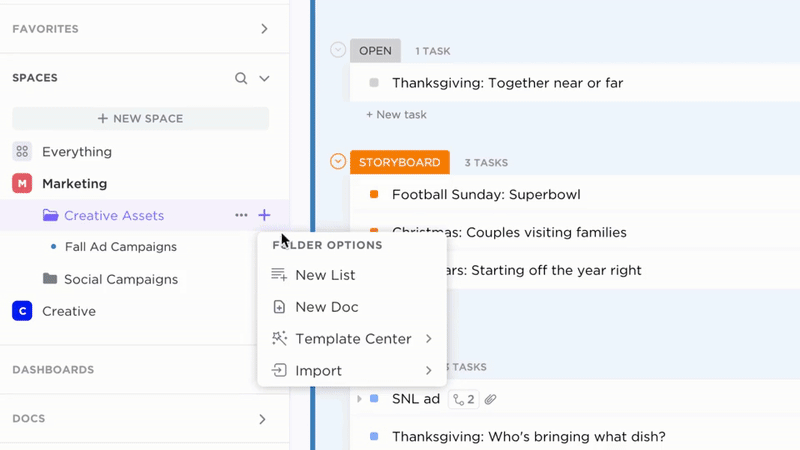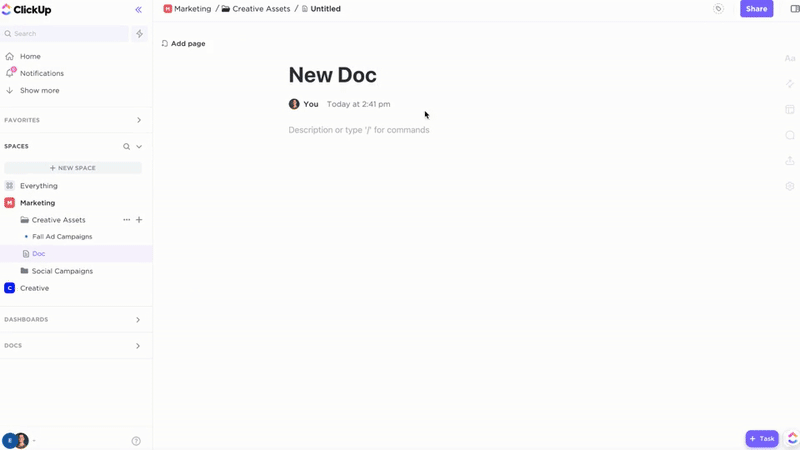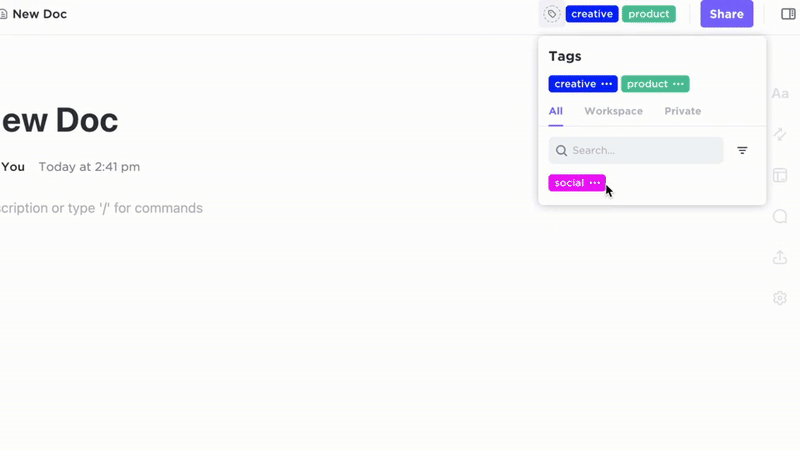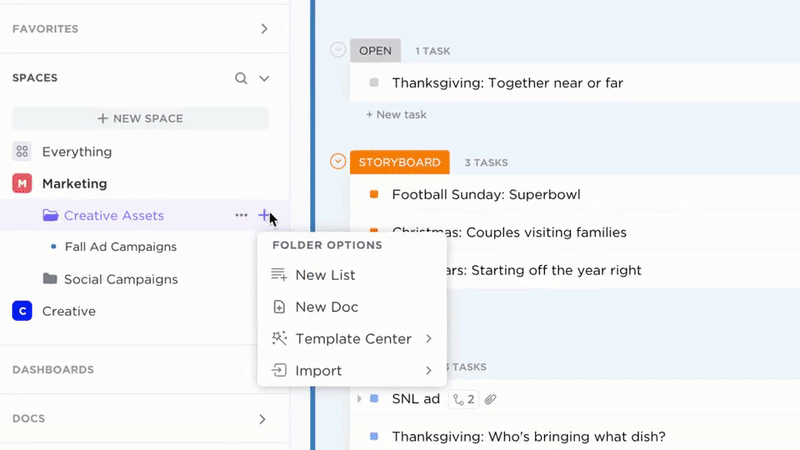
Comprehensive Documentation with ClickUp Docs
- SOPs, Validation Protocols, Training Records, Audit Reports
- Clear table of contents and nested subpages for easy navigation
- Consistent document layouts tailored for GxP standards
Why it matters: Teams access accurate procedures quickly, reducing compliance risks.
Governance
Accountable Documentation Ownership
- Identify document owners and set clear deadlines
- Assign review cycles and automate reminders
- Manage compliance tasks alongside quality operations
Why it matters: Documentation remains current and audit-ready through built-in accountability.
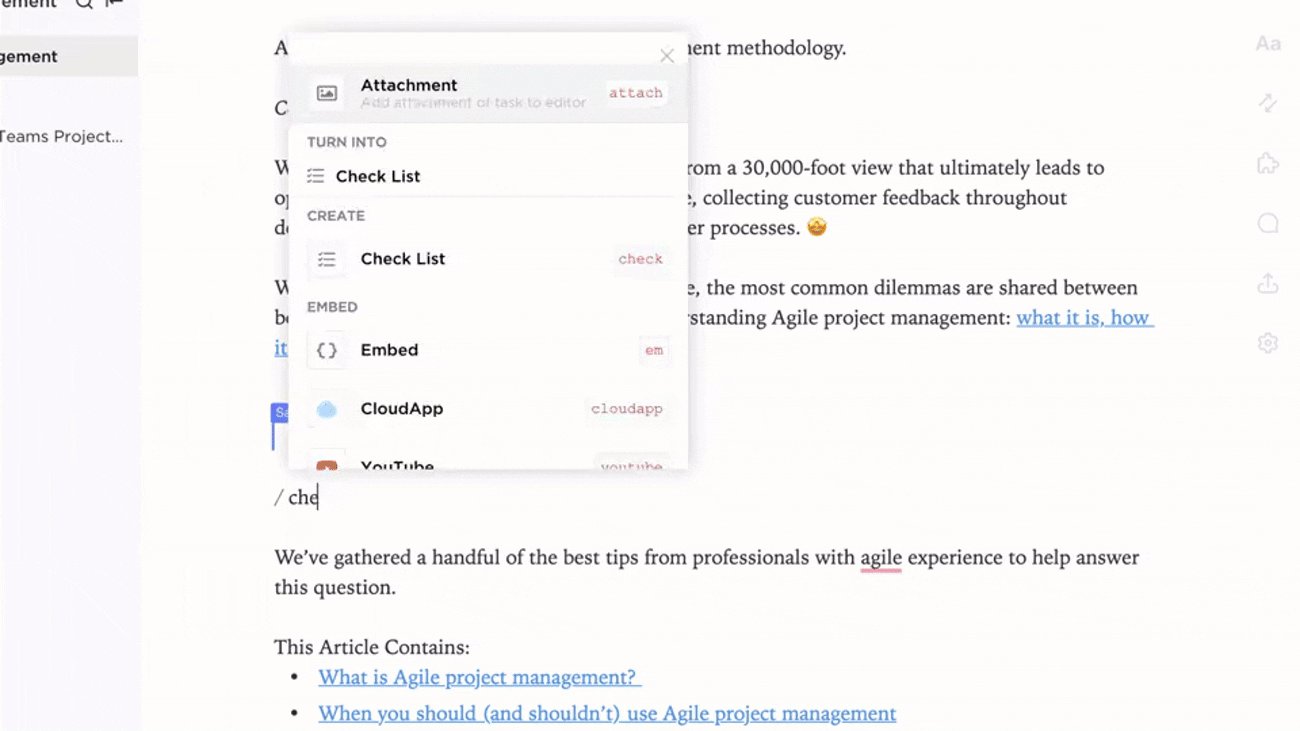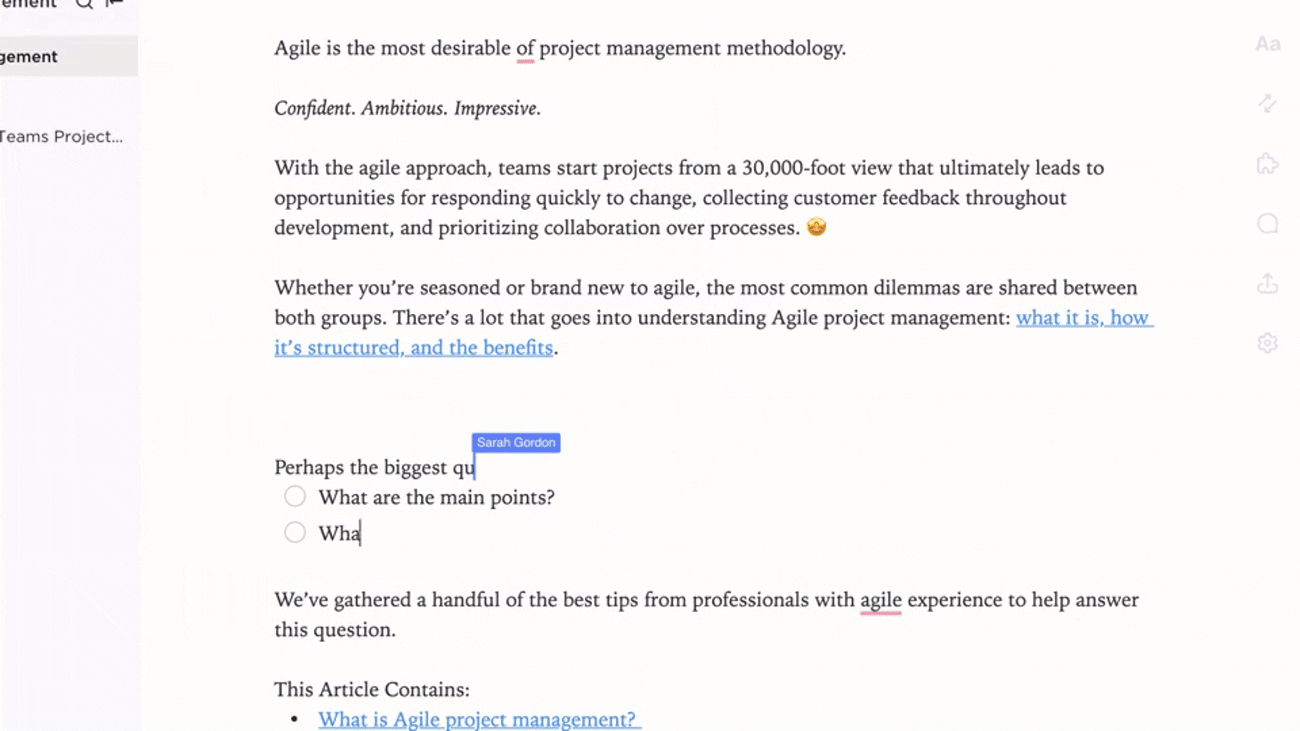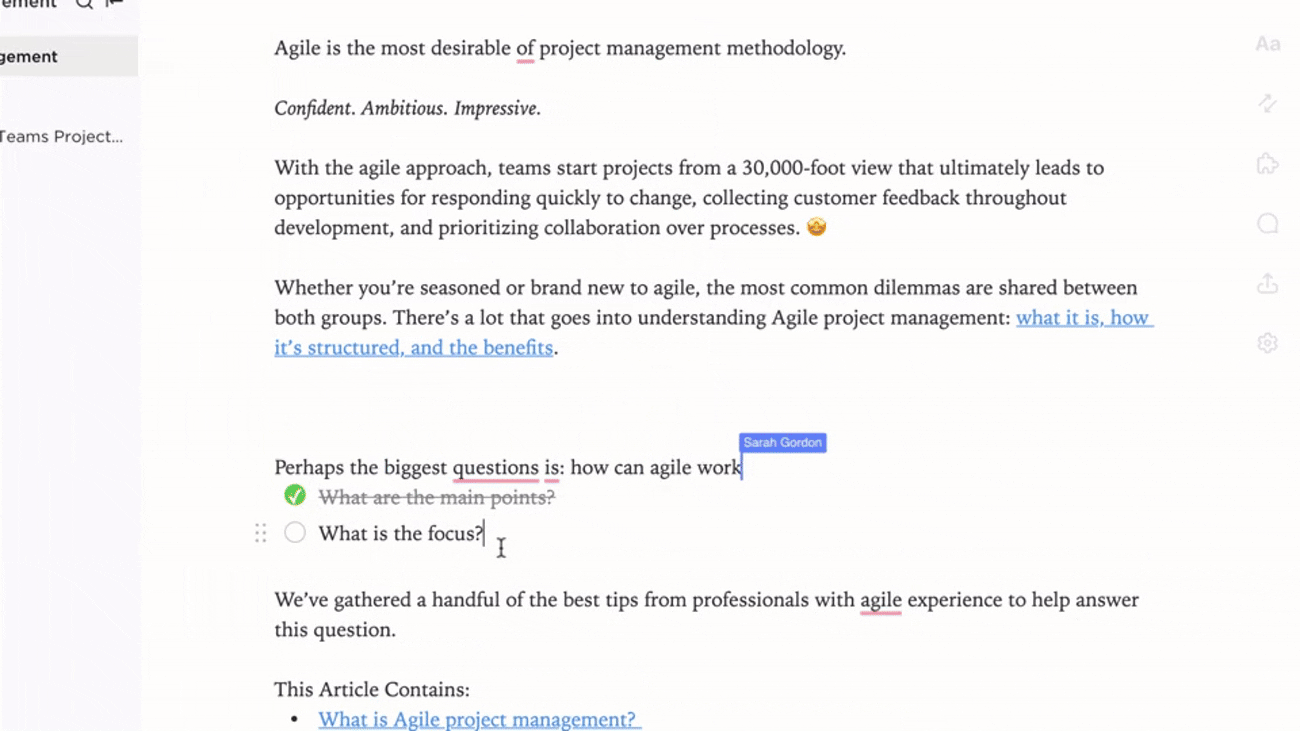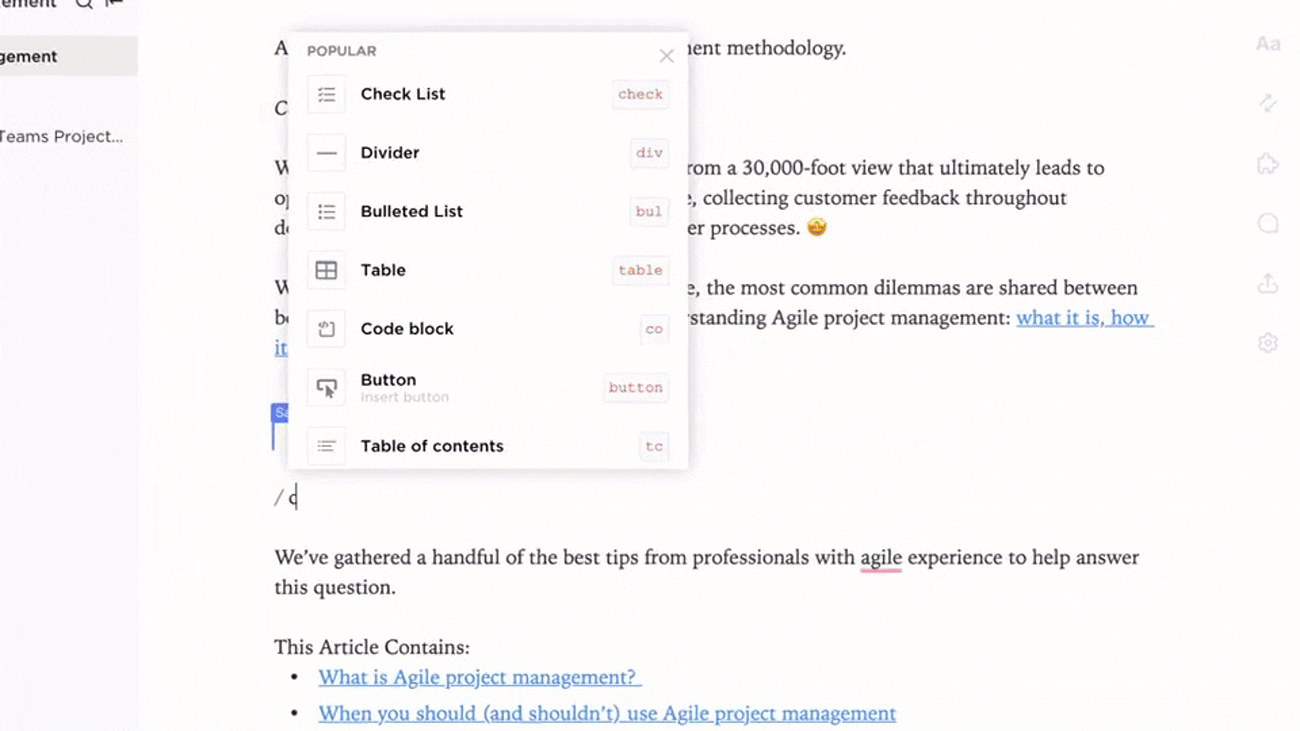
Integration
Connected Compliance Documentation
- Link documents to audits, CAPAs, and change control processes
- Connect deviations, incidents, and investigation reports
- Utilize feedback loops to refine procedures continuously
Why it matters: Documentation evolves with your quality system, ensuring regulatory alignment.
ClickUp for GxP Compliance Knowledge Base

