Build a Centralized Knowledge Base for CAPA Management Processes
Create a unified, searchable repository for corrective and preventive actions, root cause analyses, compliance records, and audit trails—so your quality team always accesses the latest, reliable information.
Why ClickUp Transforms CAPA Knowledge Management
Consolidate your CAPA documentation and execution in one dynamic platform.
Using Traditional Tools
- CAPA documents stored separately from action items, leading to outdated info
- Manual updates dependent on memory and slow communication
- Disconnected from real-time incident and audit data
- Access controls often cause redundant copies for different teams
- Entirely manual workflow slows down response and resolution times
With ClickUp for CAPA Management
- Integrate CAPA documentation with tasks, comments, and audits
- Automatically convert findings into actionable tasks with owners and deadlines
- Link knowledge base entries directly to incidents and compliance events
- Flexible permissions safeguard sensitive quality data across teams
- AI-powered drafting, summarization, and root cause analysis assistance accelerate updates
How to Develop a Robust CAPA Management Knowledge Base
Follow this 6-step approach to ensure your CAPA knowledge stays structured, actionable, and audit-ready.
1. Identify stakeholders and CAPA objectives
- Define who uses the knowledge base: quality, compliance, operations
- Clarify goals: reduce repeat incidents, accelerate investigations
- Assign ownership for ongoing content accuracy
2. Design a clear, navigable CAPA knowledge structure
- Organize by corrective actions, preventive measures, root cause analyses, audit findings
- Include sections for regulatory references and compliance requirements
- Ensure easy access to historical CAPA records and status
3. Standardize CAPA documentation templates
- Use consistent formats for corrective action reports, investigation summaries, and verification steps
- Capture essential fields: issue description, root cause, corrective action, verification, and closure
- Promote uniformity across all quality teams
4. Incorporate practical guides and troubleshooting resources
- Develop how-to content for conducting investigations and audits
- Document common CAPA challenges and resolution strategies
- Centralize tools for risk assessment and impact analysis
5. Keep knowledge base aligned with quality events and audits
- Link updates to non-conformance reports, audit results, and change requests
- Embed CAPA progress tracking within workflows
- Ensure documentation evolves alongside process improvements
6. Manage access and review cycles effectively
- Control permissions for internal teams, auditors, and external partners
- Schedule periodic reviews to maintain compliance and relevance
- Collect feedback to continuously enhance knowledge quality
How ClickUp Supports CAPA Knowledge Base Excellence
Keep CAPA documentation organized, accountable, and linked to quality outcomes in real time.
Structured CAPA Documentation with ClickUp Docs
- Sections for Investigations, Actions, Verifications, and Audit Evidence
- Intuitive table of contents with nested subpages for easy navigation
- Consistent templates for CAPA reports and root cause analyses
Why it matters: You find critical quality information quickly, reducing delays in issue resolution.
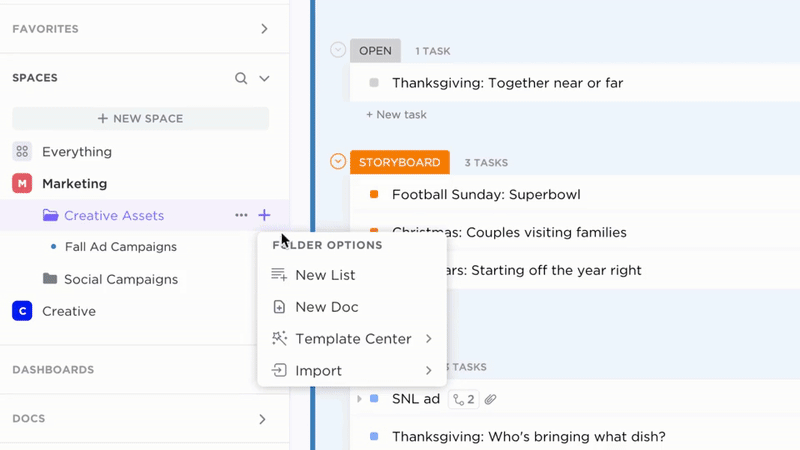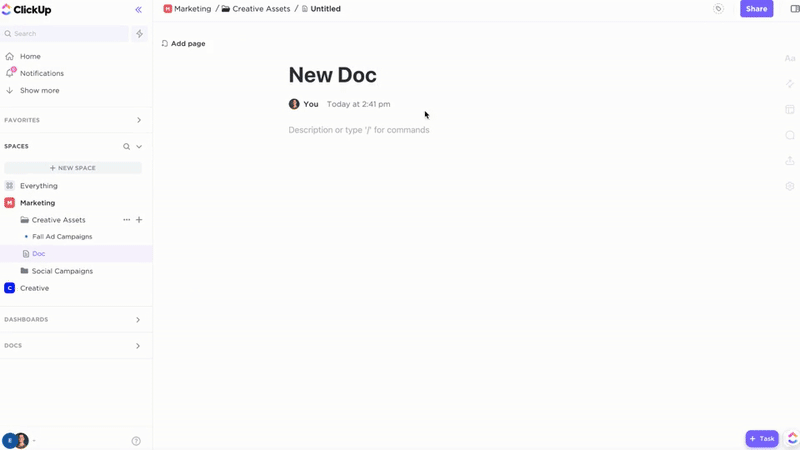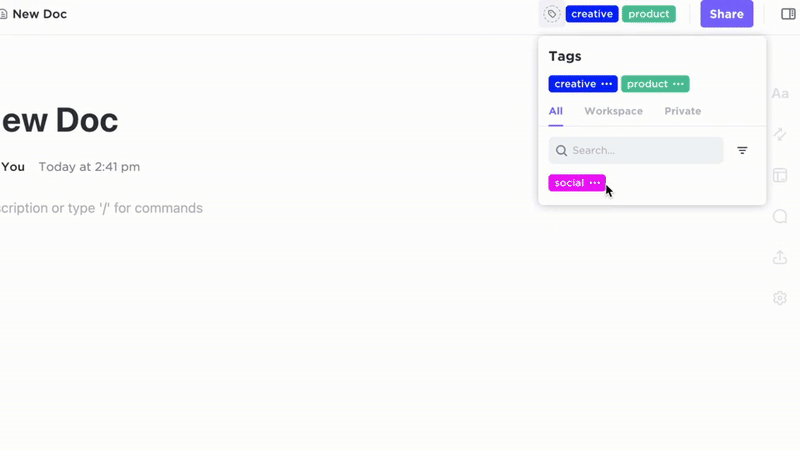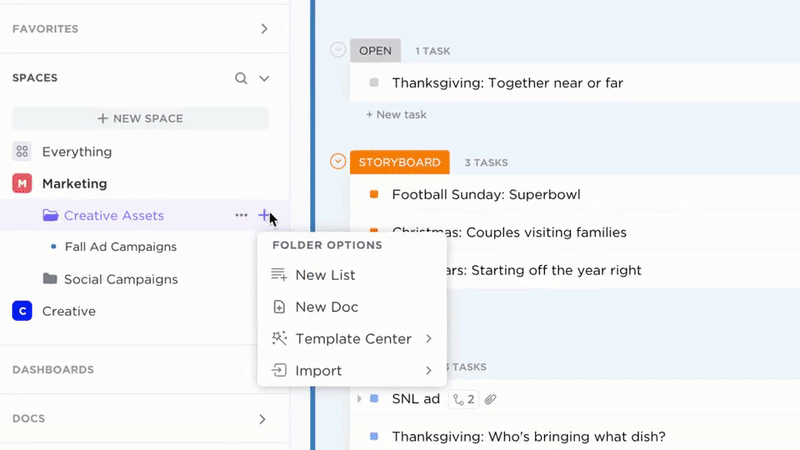
Clear Ownership and Progress Tracking
- Convert documentation gaps into assigned tasks with due dates
- Assign owners and set regular review cycles
- Monitor CAPA progress like any project work
Why it matters: Accountability ensures your CAPA knowledge stays accurate and actionable.
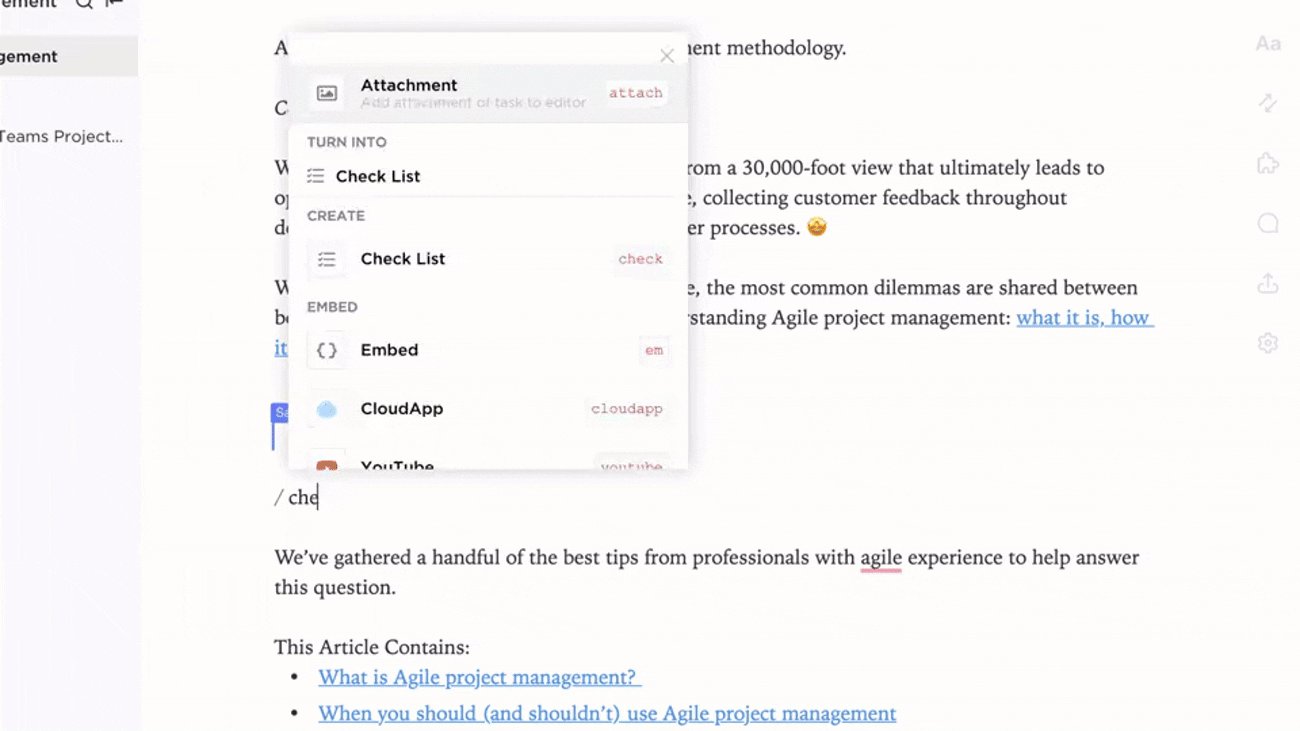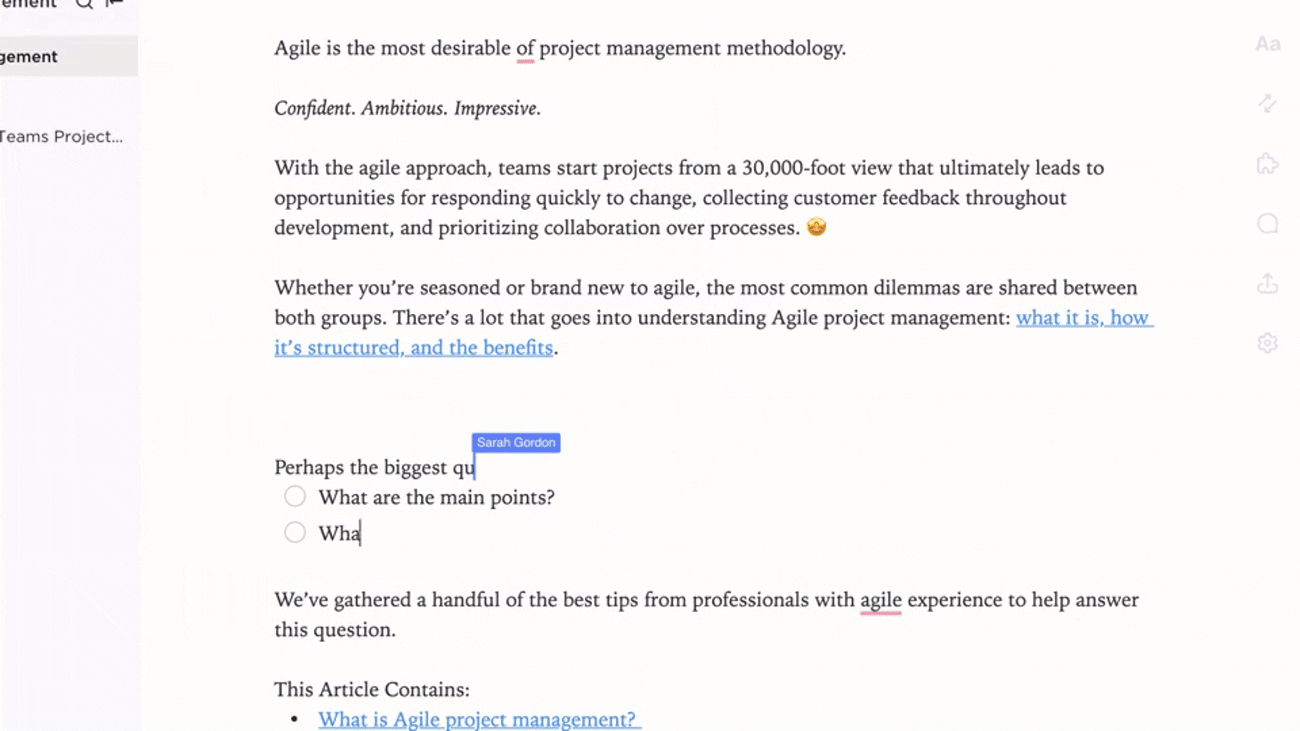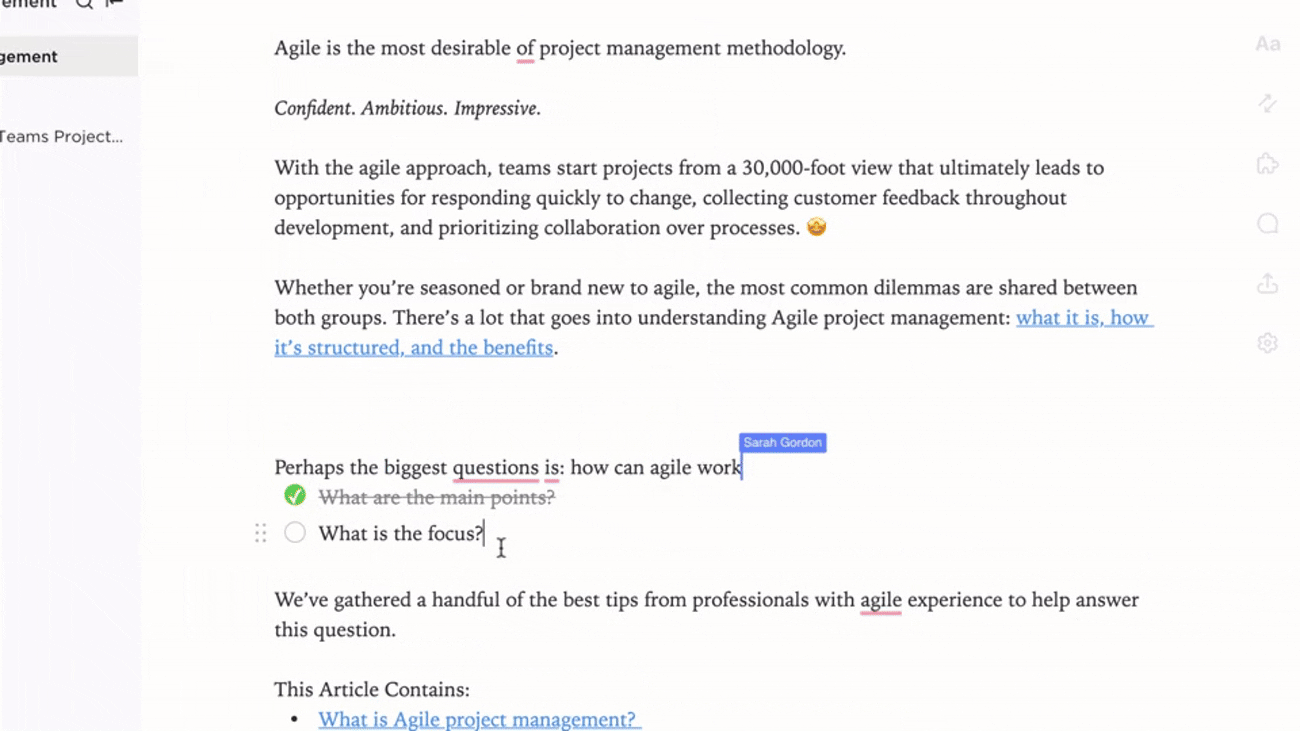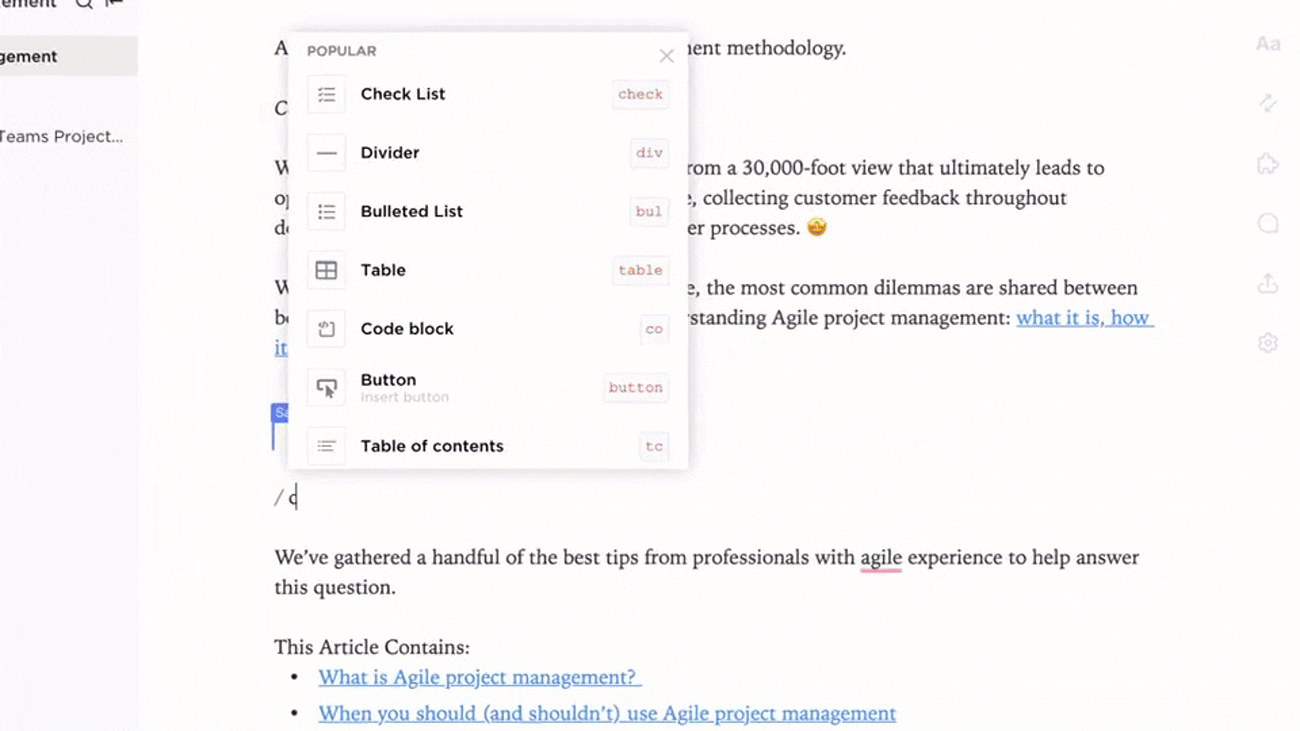
Link Documentation to Quality Events
- Attach docs to audit findings, incidents, and change requests
- Integrate CAPA knowledge with real-time compliance data
- Use feedback loops to refine procedures
Why it matters: Your knowledge base reflects the evolving quality landscape, supporting continuous improvement.