Build a Centralized Knowledge Base for Biosafety Levels
Create a comprehensive, searchable repository for biosafety protocols, containment requirements, risk assessments, and compliance guidelines—ensuring your team accesses the most current and reliable biosafety standards.
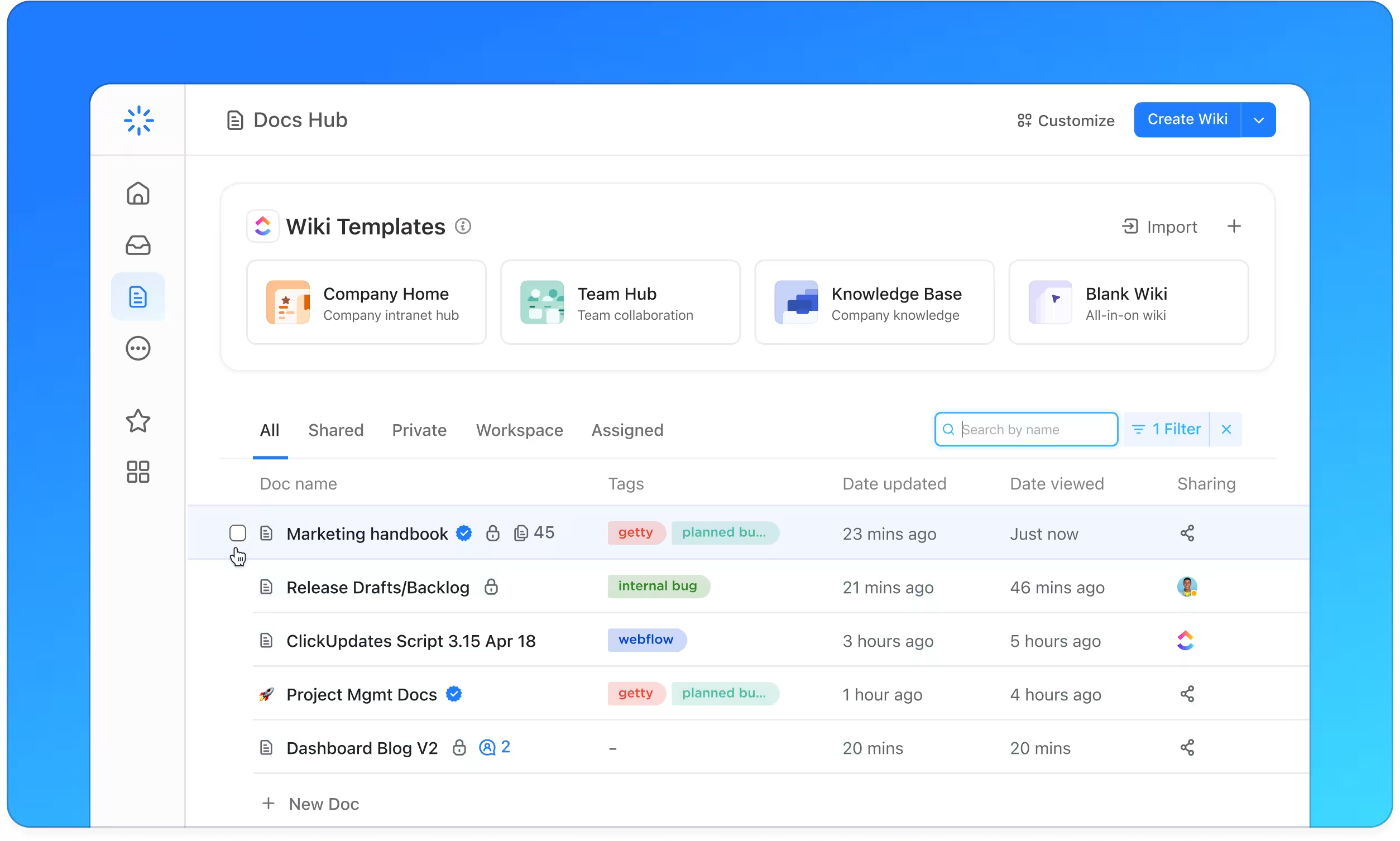
Why ClickUp Elevates Biosafety Level Knowledge Management
Consolidate knowledge and execution in a unified workspace tailored for biosafety compliance.
Traditional Approaches
- Biosafety information scattered across multiple platforms, leading to outdated protocols
- Manual updates relying on memory and informal communication
- Documentation disconnected from laboratory incidents and operational changes
- Rigid permissions causing redundant copies for different teams
- Time-intensive creation and revision processes without automation
ClickUp Advantage
- Integrate biosafety documentation with tasks and team collaboration
- Identify and assign documentation gaps as actionable tasks
- Link knowledge base entries to real-time lab activities and compliance events
- Granular access control for researchers, safety officers, and external auditors
- Leverage AI-driven content generation and summarization for rapid updates
Step-by-Step Guide to Structuring Biosafety Level Documentation
Follow these 6 essential steps to create an organized, accessible, and maintainable biosafety knowledge base.
1. Identify Stakeholders and Biosafety Objectives
- Determine users such as lab personnel, safety officers, and compliance teams
- Outline goals including protocol adherence, incident reporting, and training support
- Assign ownership to ensure accountability and ongoing updates
2. Design a Logical Knowledge Base Framework
- Establish sections like BSL-1 to BSL-4 protocols, risk assessments, and emergency procedures
- Implement intuitive navigation with tables of contents and subpages
- Include regulatory references and change logs for transparency
3. Standardize Documentation Templates for Consistency
- Develop uniform pages for each biosafety level covering containment, PPE, and waste management
- Incorporate checklist items, procedural steps, and compliance criteria
- Address common hazards and mitigation strategies to reduce errors
4. Incorporate Practical Guidance and Troubleshooting
- Provide how-to guides for equipment sterilization and incident response
- Document frequently encountered challenges and resolutions
- Centralize contact information for biosafety officers and emergency services
5. Link Documentation to Operational Events and Training
- Connect updates to lab audits, incident reports, and protocol revisions
- Embed training schedules and certification tracking
- Ensure documentation evolves alongside laboratory practices
6. Manage Permissions and Continuous Improvement
- Control access levels for internal teams, collaborators, and regulatory bodies
- Maintain version control, review cycles, and feedback mechanisms
- Use AI-assisted insights to identify knowledge gaps and streamline updates
Unlock the Power of ClickUp for Biosafety Knowledge
Ensure biosafety documentation is comprehensive, accountable, and synchronized with lab operations.
Structured Biosafety Documentation with ClickUp Docs
- Clear sections for BSL-1 through BSL-4 protocols, risk assessments, emergency plans
- Nested pages and tables of contents for effortless navigation
- Consistent formatting across all biosafety level pages
Why it matters: Safety officers and lab personnel find critical information quickly, reducing risk and improving compliance.
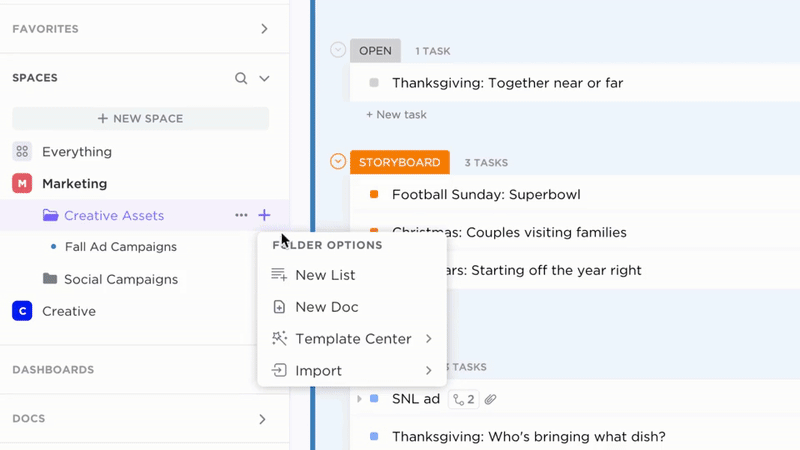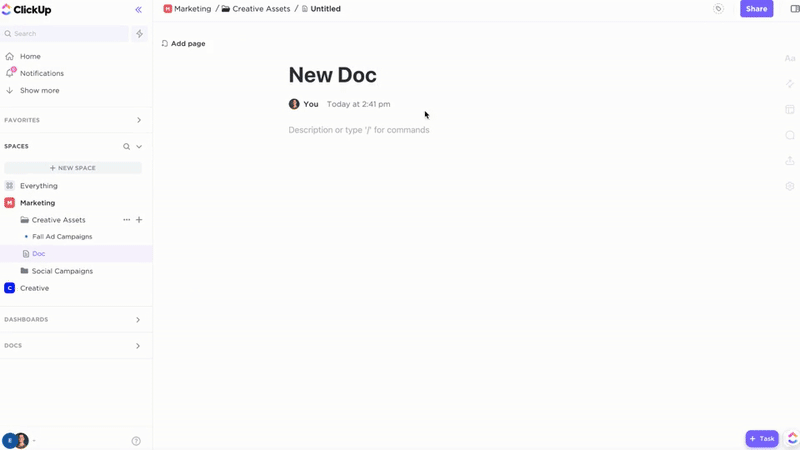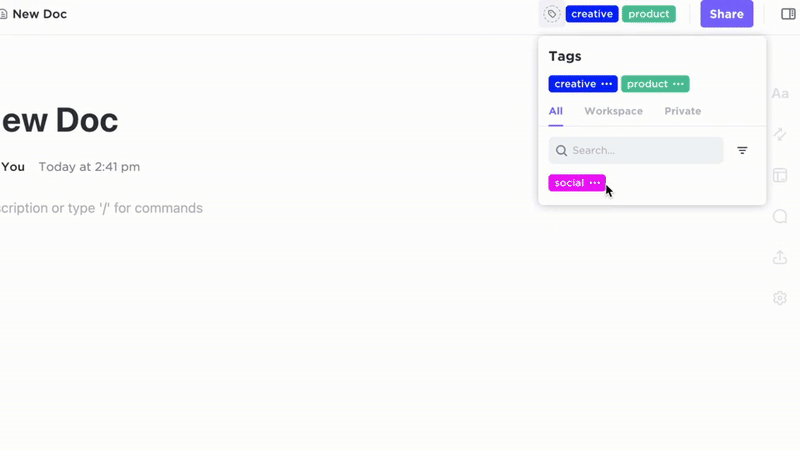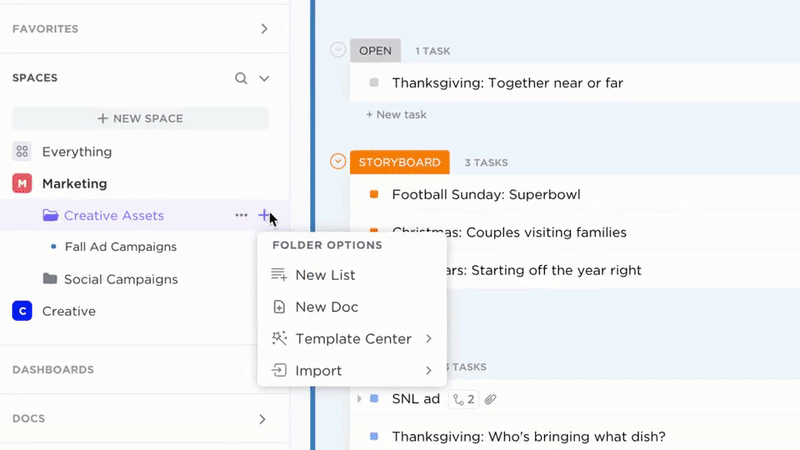
Trackable Ownership of Biosafety Content
- Identify gaps and convert them into assigned tasks
- Assign responsibility with due dates and scheduled reviews
- Monitor documentation updates like operational workflows
Why it matters: The knowledge base remains reliable and up-to-date through clear ownership, ensuring ongoing biosafety compliance.
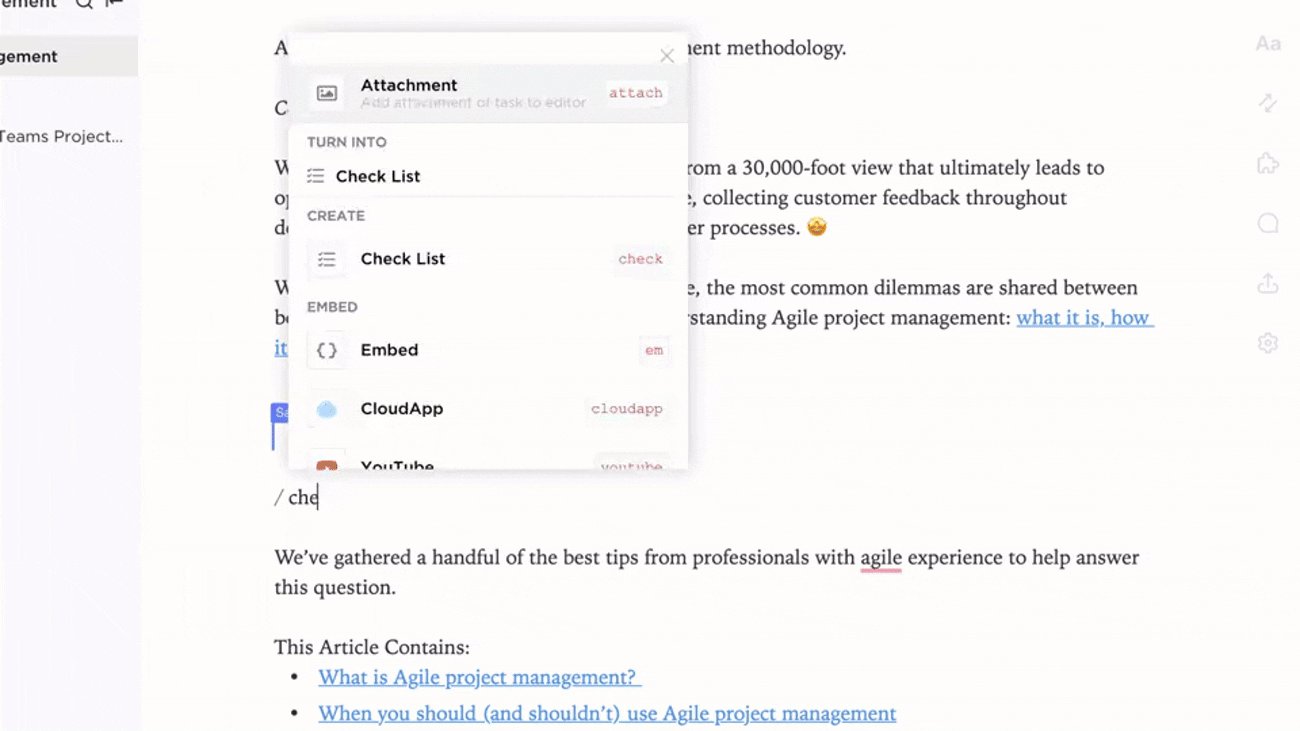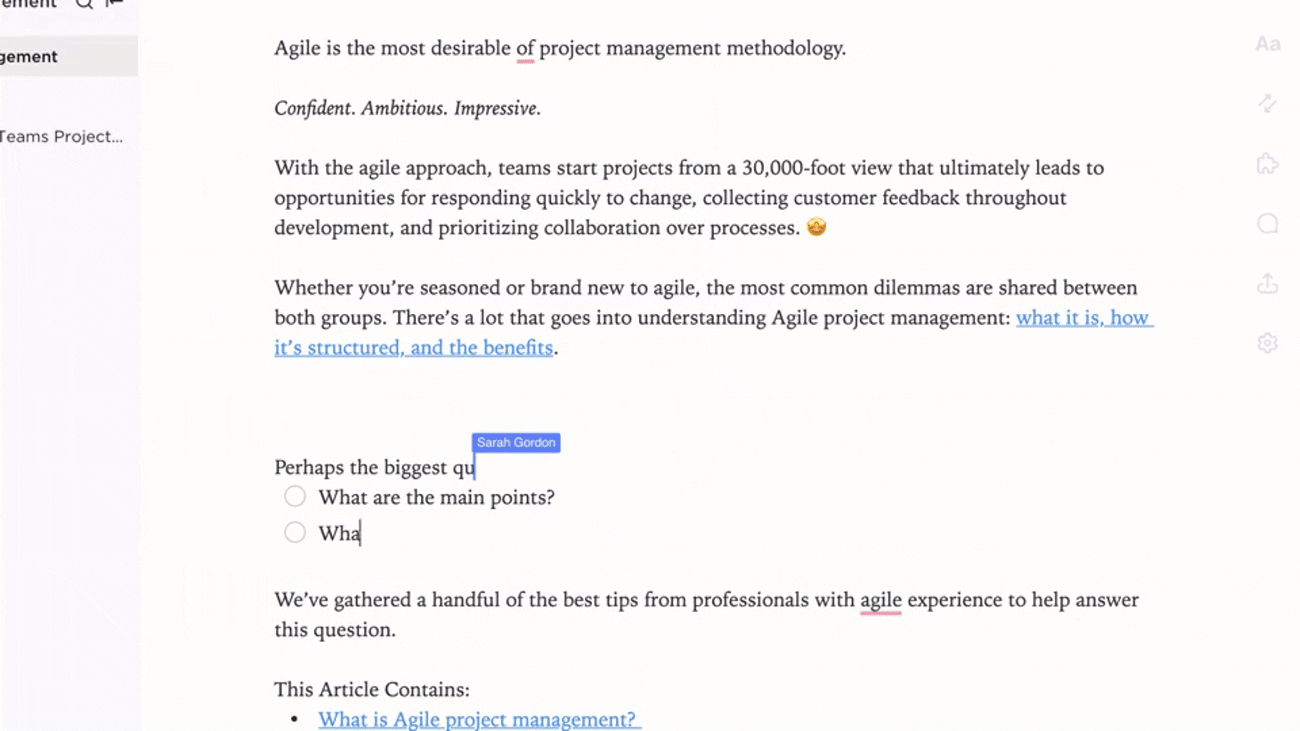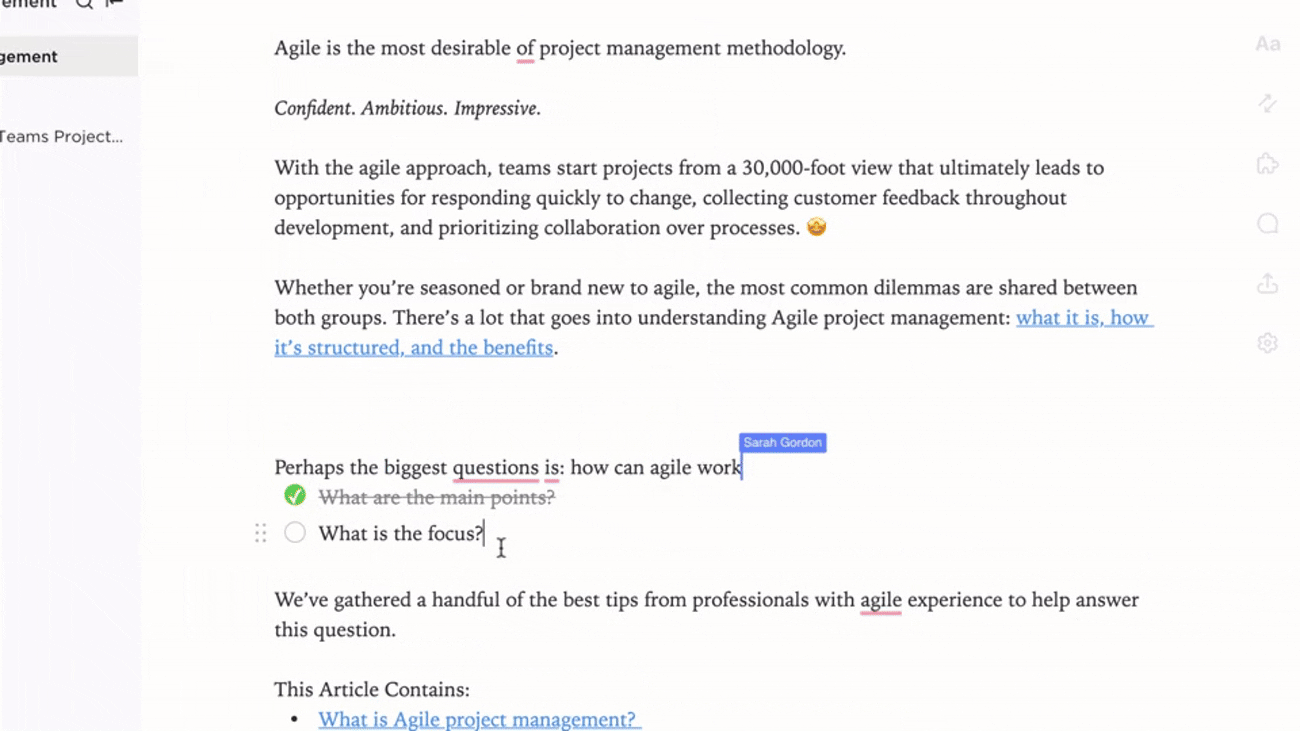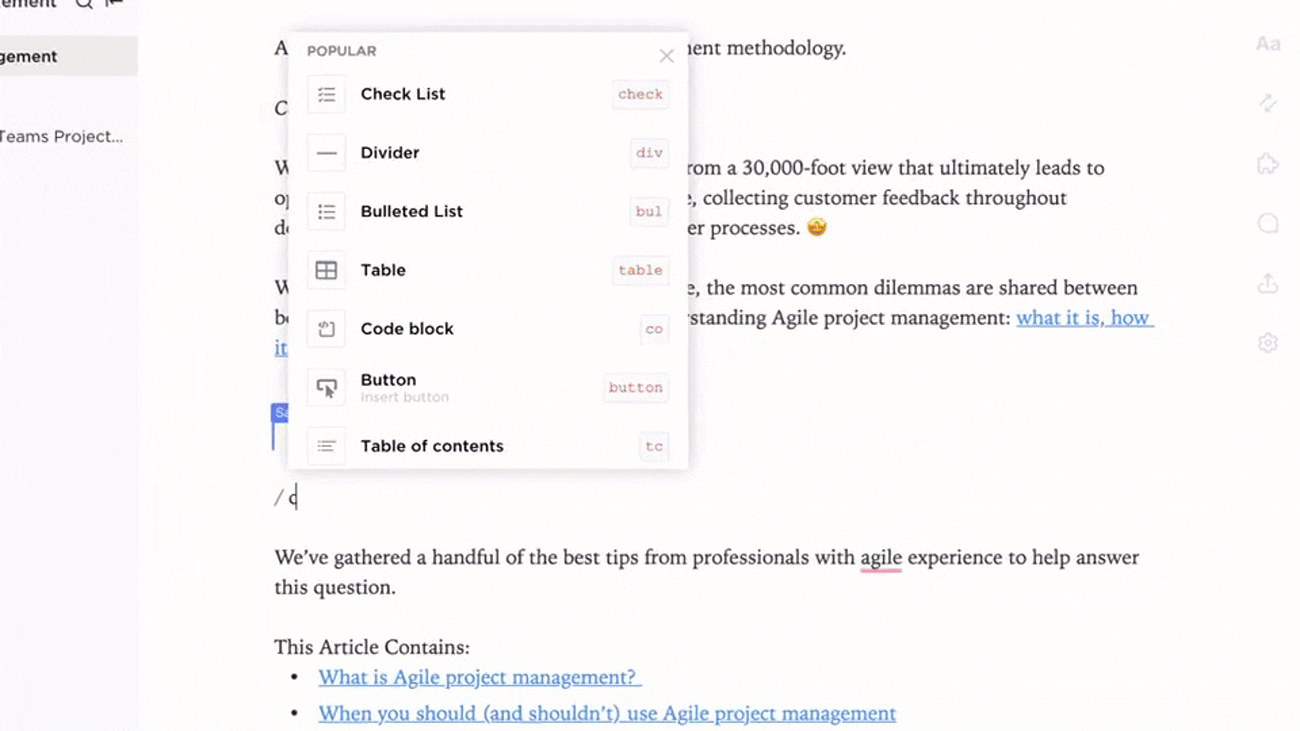
Connect Biosafety Docs to Lab Activities and Training
- Link documentation to incident reports, audits, and procedural updates
- Integrate training records and certification statuses
- Associate feedback and compliance findings directly with documentation
Why it matters: Documentation evolves with lab practices, supporting a safer and more informed environment.