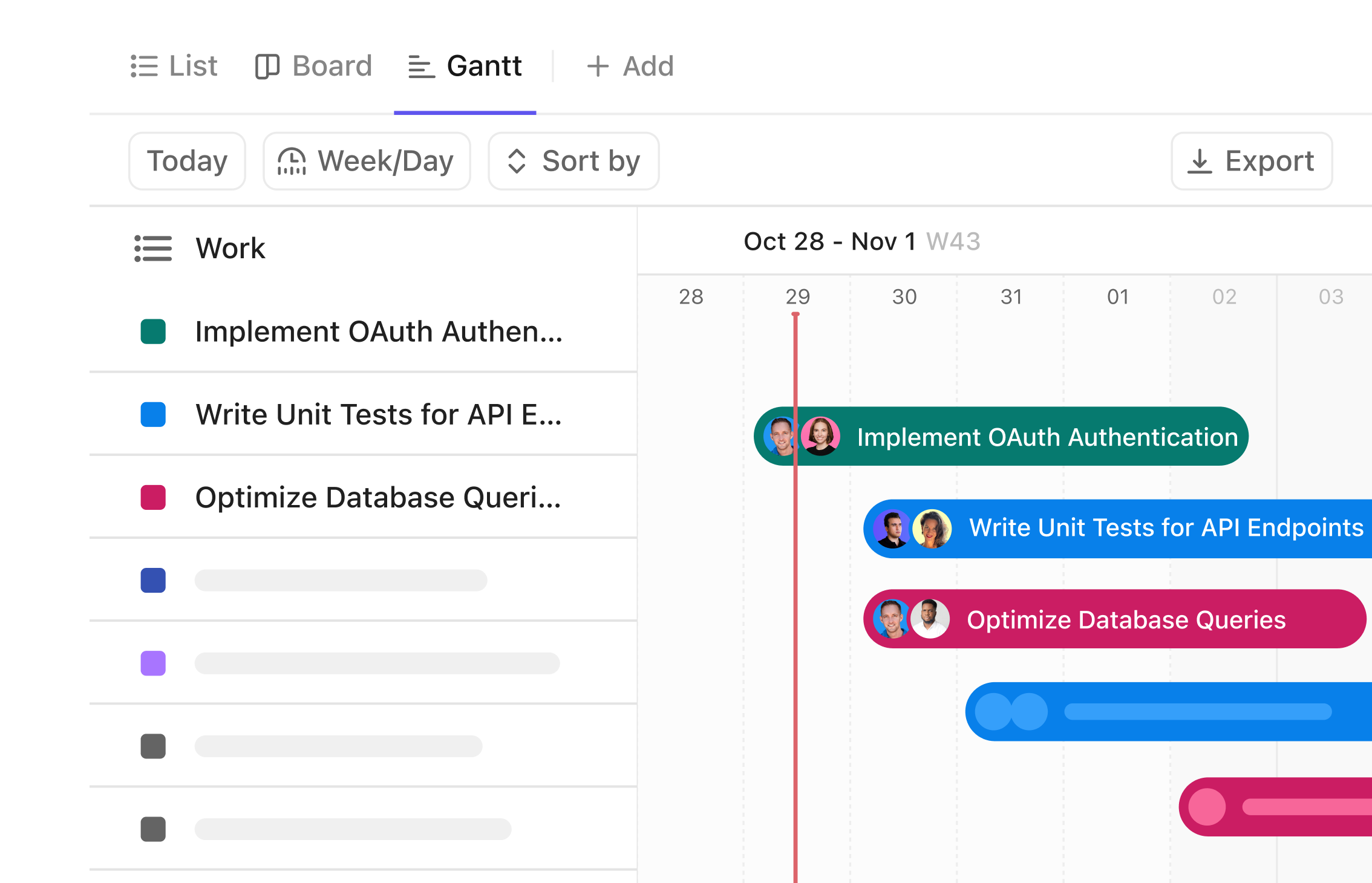
Gantt Chart Software for Supplier Compliance
Streamline Supplier Compliance Validation with Visual Timelines
Organize your supplier validation process from start to finish, track deadlines across months, coordinate teams effortlessly, and visualize every step clearly — no more scattered notes or missed milestones.
Trusted by the best
The Compliance Challenge
Why Streamlined Supplier Compliance Validation Demands Gantt Charts
Managing supplier compliance without a clear timeline invites confusion and risk. Without a structured view, critical deadlines and validation steps slip through the cracks, causing costly delays and regulatory issues.
Common pitfalls in manual tracking include:
- Unclear validation status — no quick way to see which suppliers have completed required checks.
- Missed certification renewals — leading to non-compliance and supply interruptions.
- Disjointed communication — scattered emails and documents create inefficiencies.
- Overlapping audit schedules — causing resource conflicts and audit fatigue.
- Inconsistent process adherence — lack of visibility into task dependencies and sequencing.
- Difficulty coordinating teams across departments — slowing down validation timelines.
- Hidden bottlenecks — delays remain invisible until they become urgent.
- Manual reporting errors — risking inaccurate compliance documentation.
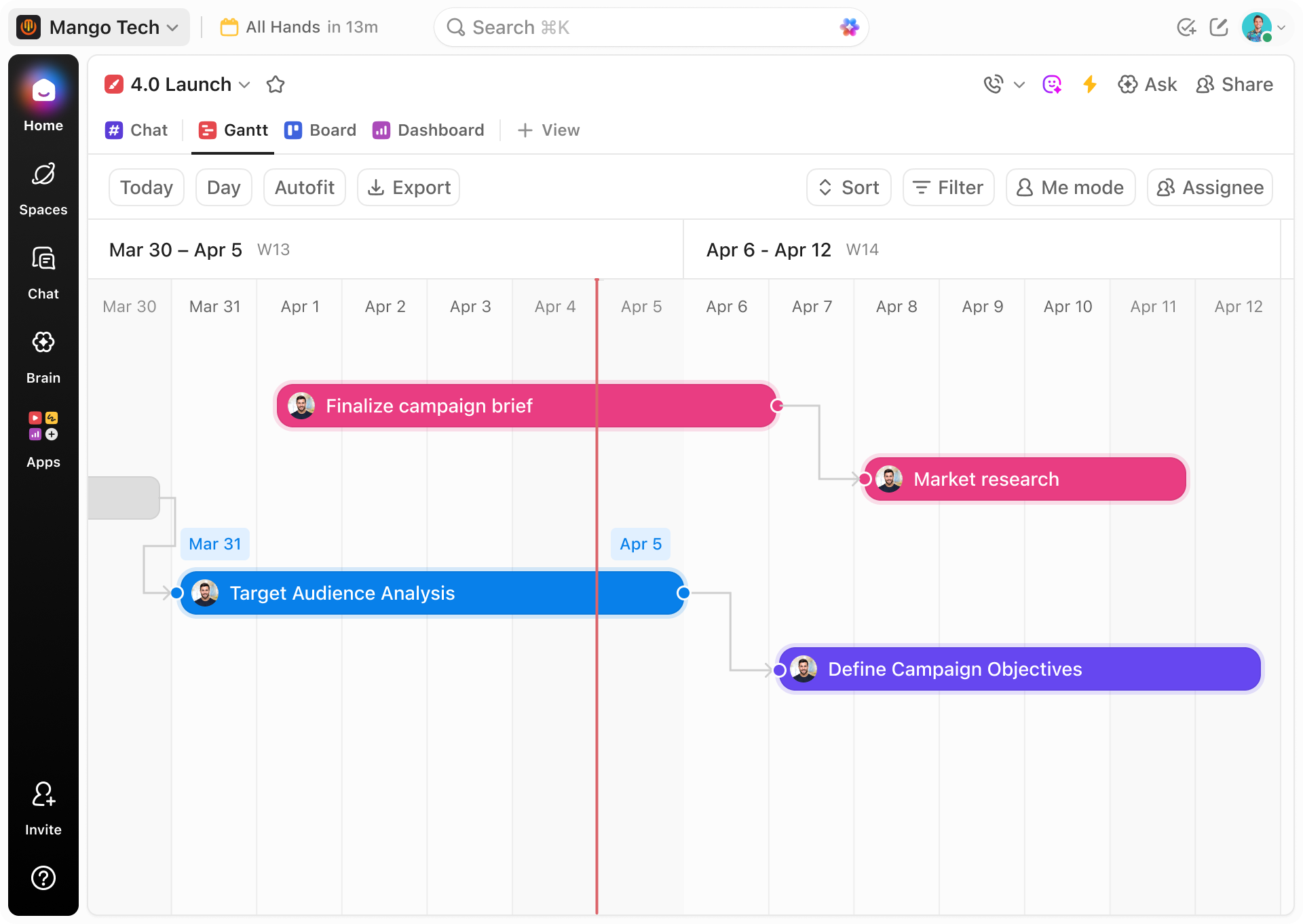
Traditional Methods vs ClickUp Gantt
Elevate Compliance Tracking Beyond Spreadsheets and Emails
Discover how ClickUp's Gantt Chart software centralizes your supplier validation process for transparency and control.
Conventional Approaches
- Tracking data scattered across spreadsheets and emails
- Manual deadline monitoring prone to errors
- No clear visualization of task dependencies
- Difficult collaboration across teams and departments
- Reacting to compliance issues rather than preventing them
- Time-consuming status reporting and documentation
ClickUp Gantt Chart Software
- Unified timelines showing all validation tasks and milestones
- Automated reminders and deadline alerts
- Visual mapping of dependencies and sequencing
- Real-time collaboration with clear task ownership
- Proactive identification of potential delays
- Easy access to documentation linked directly to tasks
Use Cases
How Gantt Chart Software Advances Supplier Compliance Validation
Leverage visual project management to ensure your supplier validation stays on track and audit-ready.
#UseCase1
Visualizing Complex Validation Timelines
ClickUp’s Gantt charts bring every validation step into clear view—so you can spot bottlenecks and keep processes moving smoothly.
#UseCase2
Coordinating Cross-Functional Teams
Assign tasks, set deadlines, and track progress across procurement, quality, and compliance teams seamlessly.
#UseCase3
Tracking Certification and Documentation Status
Keep all supplier certificates and compliance documents linked to timeline tasks for instant access and audit readiness.
#UseCase4
Managing Audit Schedules and Follow-ups
Map audit dates and required follow-up actions with dependencies to ensure nothing falls through the cracks.
#UseCase5
Automating Reminders for Renewal Deadlines
Set automated alerts for certification expirations and validation renewals to maintain continuous compliance.
#UseCase6
Streamlining Risk Assessment and Approval Processes
Visualize each risk evaluation step and approval milestone to guarantee thorough supplier vetting.
#UseCase7
Integrating Supplier Feedback and Issue Resolution
Turn supplier communication and corrective actions into trackable tasks within your Gantt timeline.
#UseCase8
Providing Transparent Reports to Stakeholders
Generate clear visual reports from your Gantt charts to keep leadership and auditors informed.
#UseCase9
Ensuring Consistent Compliance Across Global Suppliers
Coordinate validation activities internationally with timelines that adapt to time zones and regional requirements.
Key Beneficiaries
Who Benefits Most from ClickUp Gantt for Supplier Compliance?
Ideal for professionals managing complex supplier networks and compliance demands.
If you’re a Compliance Manager
Visualize all supplier validation tasks and deadlines in one place to maintain oversight and accountability.

If you’re a Procurement Specialist
Coordinate supplier onboarding and validation steps seamlessly across departments and regions.

If you’re a Quality Assurance Lead
Track audits, certifications, and corrective actions with clear timelines and task dependencies.

How ClickUp Facilitates Compliance
Transform Supplier Validation with ClickUp Gantt Charts
Gain full visibility and control over your compliance processes
Centralize Supplier Data and Documents
Link certificates, contracts, and audit reports directly to tasks for easy reference and audits.
Plan Validation Steps End-to-End
Map every stage from initial assessment to final approval for comprehensive oversight.
Standardize Processes with Templates
Use reusable templates to ensure consistent compliance validation across all suppliers.
Coordinate Teams Across Departments
Assign responsibilities, track progress, and communicate within the timeline for efficiency.
Turn Compliance Meetings into Action Items
Convert discussions into actionable tasks with deadlines and dependencies on your Gantt chart.
Never Miss Critical Deadlines
Automated reminders and timeline views keep you ahead of renewals and audits.