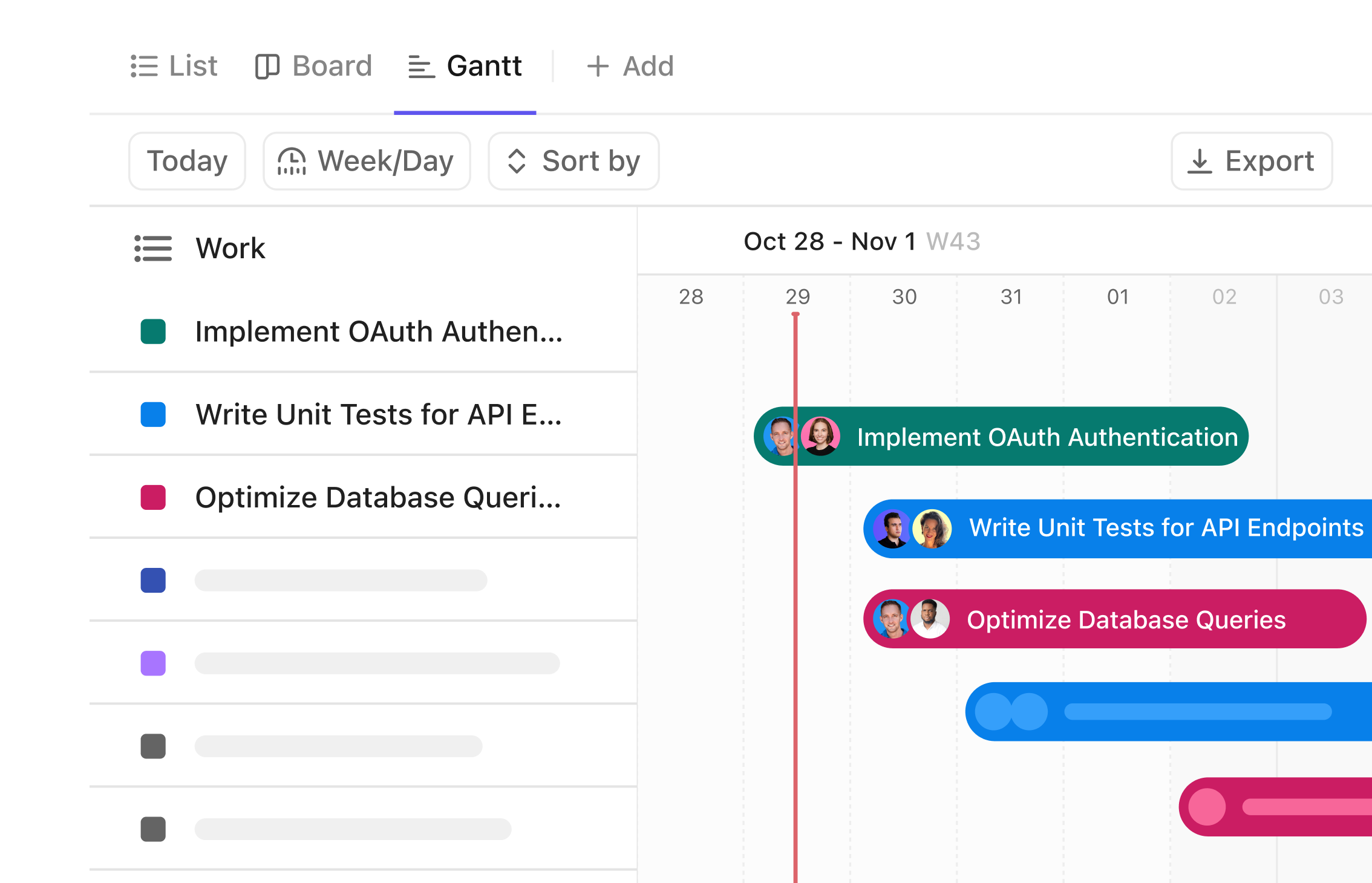
Gantt Chart Software for Lab Sample Management
Visualize Your Lab Sample Workflow with Precision
Track every sample from collection to analysis with a clear timeline. Coordinate your team and monitor progress with a Gantt chart designed for lab sample tracking.
Trusted by the best
The Need for Gantt Charts
Why Lab Sample Tracking Demands Gantt Chart Software
Managing lab samples without a clear timeline leads to confusion and errors. Samples get misplaced, tests overlap, and deadlines slip away.
Here’s where traditional tracking falls short:
- Sample processing steps lack coordination — tasks happen out of order or get missed.
- Data collection timelines are unclear — making it hard to monitor sample status.
- Resource scheduling conflicts arise — equipment and personnel get double-booked.
- Communication gaps cause delays — lab members lose track of sample progress.
- Compliance deadlines get overlooked — risking regulatory issues.
- Progress tracking is manual and error-prone — leading to bottlenecks and rework.
- Documentation is scattered — important notes and files live in different places.
- Urgent samples get lost in the shuffle — delaying critical results.
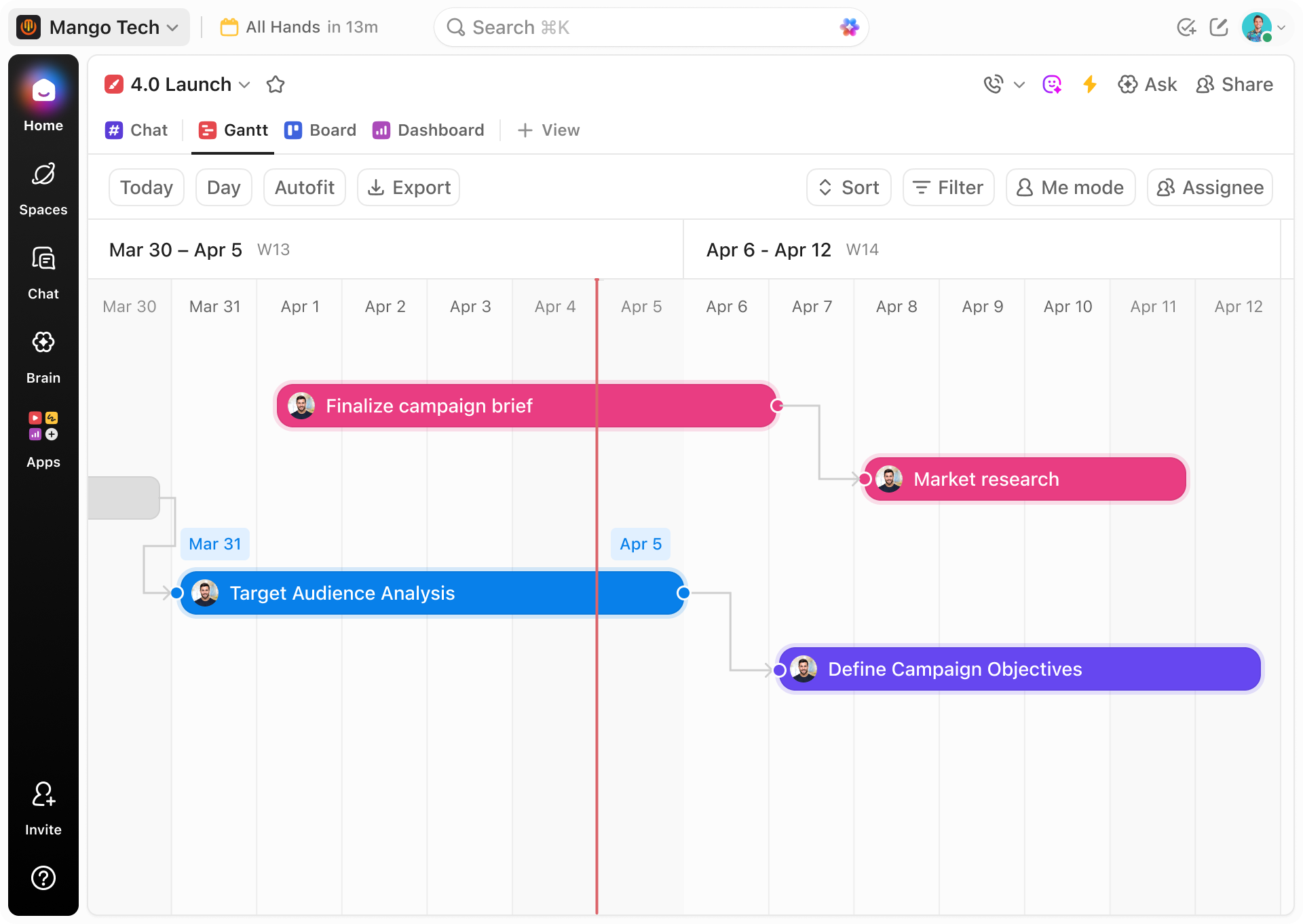
Lab Sample Tracking Methods
Why Traditional Tracking Falls Short Compared to ClickUp Gantt
Discover how ClickUp's Gantt charts bring clarity and control to your lab sample workflows.
Conventional Tracking
- Relies on spreadsheets, paper logs, and memory
- Manual updates lead to inaccuracies
- No visual overview of sample status
- Difficult to coordinate team tasks
- Overlapping equipment bookings
- Compliance deadlines tracked separately
Use Cases
How Gantt Chart Software Enhances Lab Sample Tracking
Transform your lab workflows with visual timelines that keep every sample on track.
#UseCase1
Eliminating Sample Processing Delays
Visualize each processing step so you can identify bottlenecks and keep samples moving smoothly through the pipeline.
#UseCase2
Ensuring Compliance with Regulatory Timelines
Map critical deadlines and documentation requirements to stay audit-ready and avoid compliance risks.
#UseCase3
Coordinating Multi-Step Assays Effectively
Track dependencies between assay stages to prevent errors and ensure accurate results.
#UseCase4
Optimizing Equipment and Resource Allocation
Schedule lab instruments and personnel with clear visibility to avoid conflicts and downtime.
#UseCase5
Tracking Sample Quality Control Processes
Monitor QC steps with milestones and alerts to maintain high standards throughout your workflow.
#UseCase6
Facilitating Cross-Department Collaboration
Keep everyone aligned on sample status and next steps, whether in the lab or supporting teams.
#UseCase7
Managing Urgent Sample Prioritization
Adjust timelines dynamically to prioritize critical samples without disrupting overall workflow.
#UseCase8
Documenting Sample Histories Clearly
Link all related data, notes, and files directly to sample tasks for easy access and traceability.
#UseCase9
Automating Notifications for Time-Sensitive Tasks
Set up alerts to ensure important steps happen on time, reducing risk of delays.
Key Beneficiaries
Who Gains the Most from ClickUp Gantt for Lab Sample Tracking
Designed for lab managers, technicians, and researchers handling complex sample workflows.
If You Manage a High-Volume Laboratory
ClickUp helps you visualize workflows across multiple projects and teams, keeping sample tracking streamlined and error-free.

If You’re a Lab Technician
Sequence sample preparation, testing, and data entry with clear timelines to minimize errors and maximize throughput.

If You’re a Research Scientist
Plan experiments, track sample batches, and coordinate with collaborators on a transparent visual timeline.

How ClickUp Supports Lab Sample Tracking
Step Up Your Lab’s Sample Management Game
Harness the power of ClickUp Gantt charts to streamline every phase of sample tracking.
Centralize Sample Data and Protocols
Keep all sample information, testing protocols, and results linked to specific timeline tasks for easy reference.
Visualize Sample Processing From Start to Finish
Map every step from receipt to analysis and reporting to ensure nothing falls through the cracks.
Standardize Procedures with Dependencies
Use task dependencies to enforce correct order of operations and prevent costly mistakes.
Coordinate Lab Team Activities
Assign responsibilities with deadlines and track progress to keep everyone in sync.
Turn Lab Meetings into Action Plans
Convert discussions into actionable tasks on the timeline, so next steps are clear and assigned.
Never Miss Critical Sample Deadlines
Set automated alerts for regulatory submissions, quality checks, and reporting milestones.