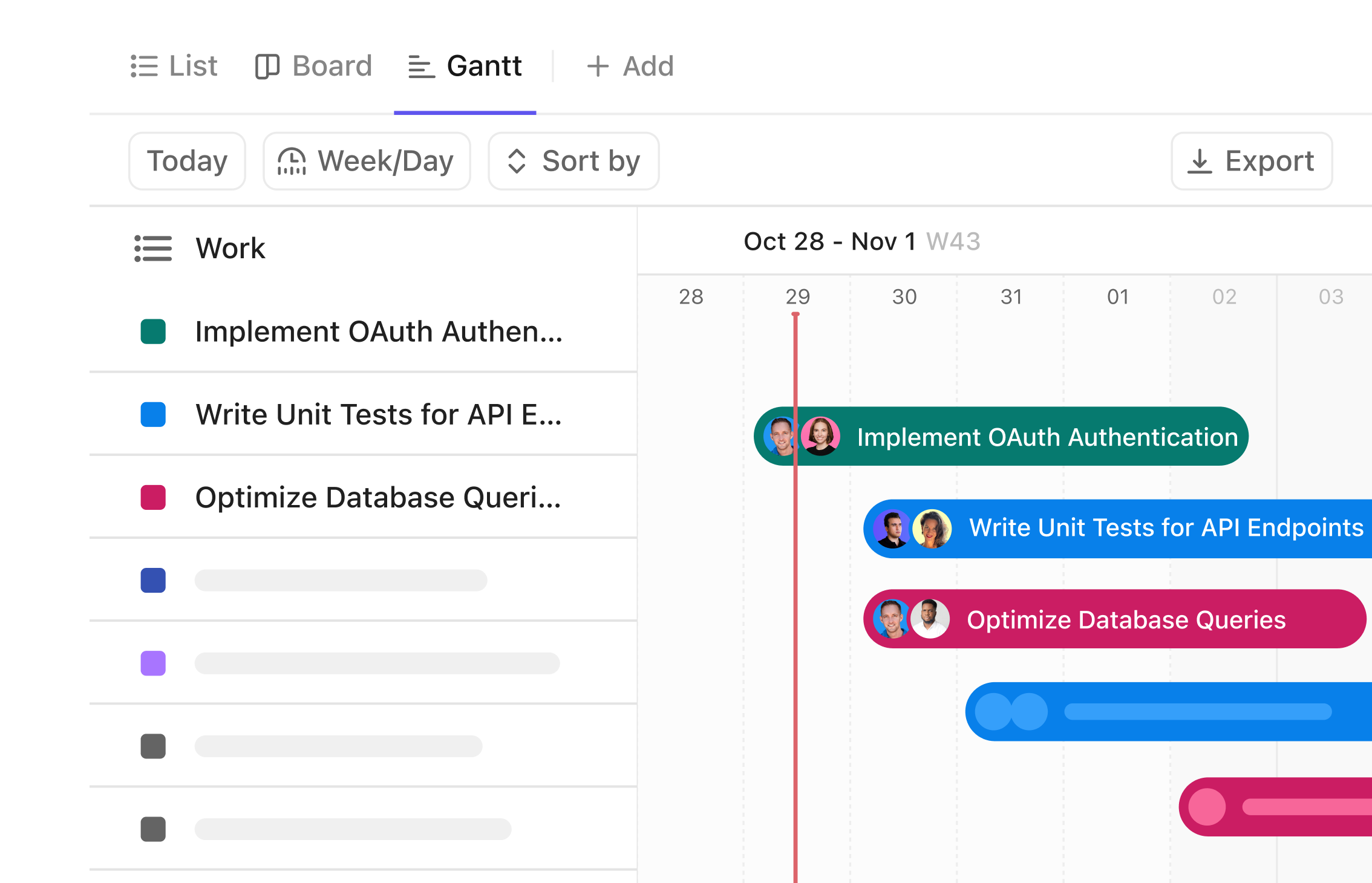
Gantt Chart Software Tailored for Clinical Researchers
Visualize Your Clinical Study Journey with Precision
Plan every phase from protocol design to patient follow-up, monitor timelines spanning months or years, coordinate multidisciplinary teams, and access a clear visual workflow beyond your mental notes.
Trusted by the best
The Importance of Timeline Management
Why Clinical Researchers Benefit from Gantt Chart Software
Conducting clinical trials without a structured timeline invites confusion and risk. Without it, priorities blur, organization falters, and critical deadlines emerge unexpectedly.
Here’s what clinical research struggles with when lacking a timeline perspective:
- Complex trial phases lose clarity — it’s difficult to track recruitment, intervention, and follow-up stages.
- Regulatory submissions get overlooked — missed FDA, IRB, or ethics board deadlines delay approvals.
- Patient visit scheduling becomes chaotic — inconsistent timing leads to data gaps and protocol deviations.
- Data collection and analysis fall behind — unclear sequencing causes bottlenecks and errors.
- Collaboration across sites is disorganized — misaligned tasks and unclear responsibilities slow progress.
- Resource allocation is inefficient — equipment and staff scheduling conflicts go unnoticed.
- Progress tracking is opaque — months of effort feel stagnant without visual updates.
- Communication scatters across channels — emails, spreadsheets, and calls lack centralized coordination.
Traditional vs ClickUp
Why Traditional Processes Fall Short Compared to ClickUp Gantt for Clinical Research
Discover how ClickUp’s Gantt Charts bring clarity and control where conventional tools leave gaps.
Conventional Methods
- Trial steps tracked in spreadsheets and emails
- Recruitment and follow-up schedules managed manually
- Regulatory timelines often missed
- Site communication relies on fragmented channels
- Data management lacks integration
- Resource conflicts unmanaged
ClickUp Gantt Charts
- Unified view of tasks, milestones, and timelines
- Visualize patient visits and protocol phases with dependencies
- Automated reminders for regulatory submissions
- Centralized collaboration across all sites
- Link documents, patient data, and reports directly
- Monitor resource availability and allocation
Use Cases
How Gantt Chart Software Empowers Clinical Researchers
Explore how visual timelines eliminate bottlenecks and drive trial success.
#UseCase1
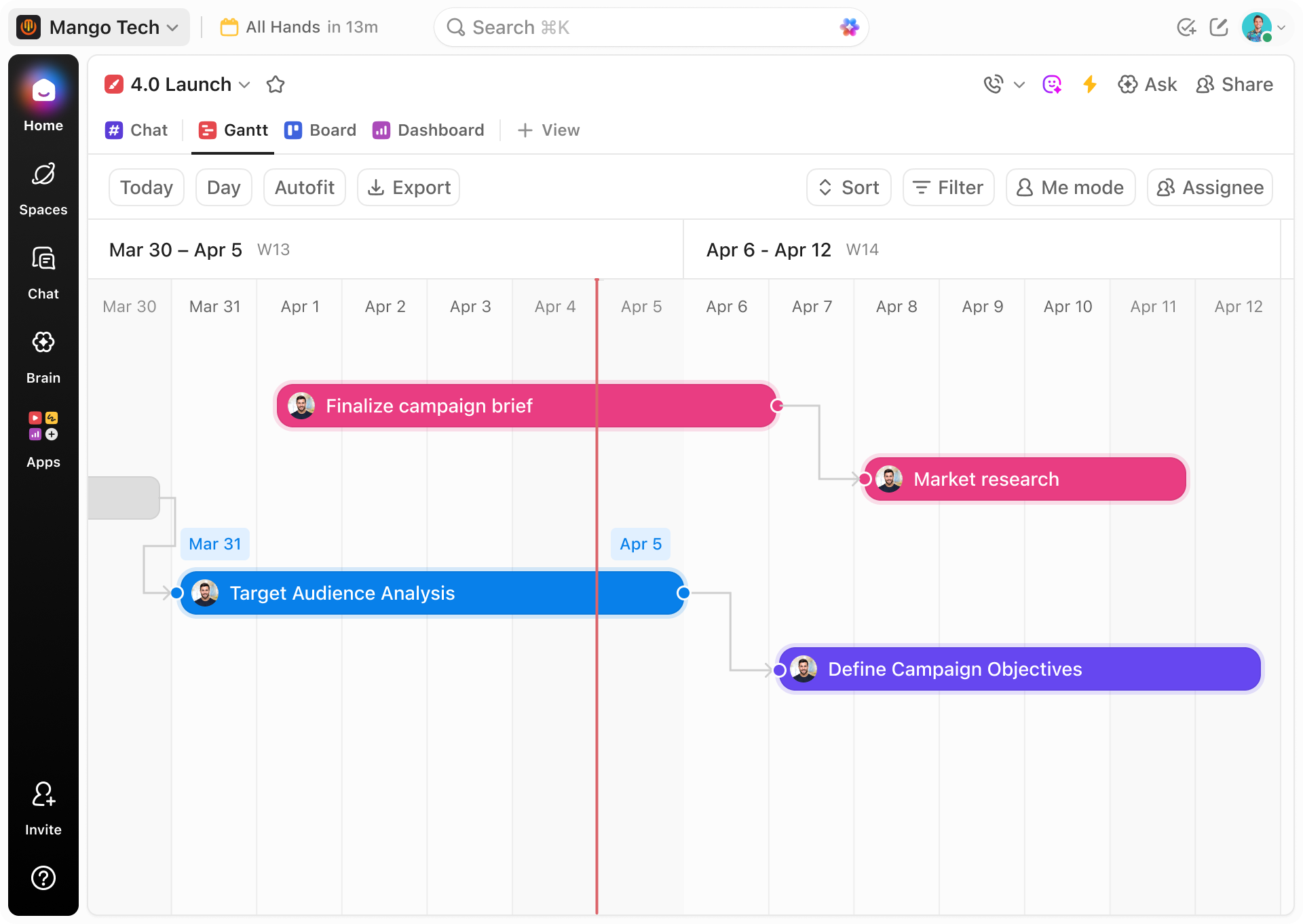
Clarifying Multiphase Trial Schedules
ClickUp’s Gantt charts transform complex clinical timelines into clear visuals, helping you identify and resolve scheduling bottlenecks across phases.
#UseCase2
Ensuring Compliance with Regulatory Deadlines
Map every submission date and approval milestone to keep your trial compliant and on track with IRB, FDA, and ethics requirements.
#UseCase3
Coordinating Multi-Site Patient Recruitment
Visualize recruitment progress and align site activities with dependencies to optimize enrollment timelines.
#UseCase4
Sequencing Protocol Steps to Avoid Data Errors
Link tasks with dependencies to ensure protocol adherence and reduce risks of procedural deviations.
#UseCase5
Managing Resource Allocation Across Teams
Monitor and adjust equipment, staff, and lab availability visually to prevent conflicts and downtime.
#UseCase6
Tracking Data Collection and Analysis Milestones
Set clear timelines for data capture and statistical review phases, ensuring timely insights and reporting.
#UseCase7
Preparing for Audits with Documented Timelines
Maintain a transparent timeline trail that auditors and sponsors can review instantly for compliance verification.
#UseCase8
Facilitating Effective Team Communication
Centralize updates, assign responsibilities, and keep all stakeholders informed within one interactive timeline.
#UseCase9
Adapting to Protocol Amendments Swiftly
Easily adjust timelines and dependencies to reflect changes, keeping your trial agile and compliant.
Key Beneficiaries
Who Gains the Most from ClickUp Gantt in Clinical Research
Ideal for anyone managing complex trial timelines, multidisciplinary teams, or regulatory milestones.
If You Lead Multicenter Clinical Trials
ClickUp provides a synchronized timeline across sites and time zones, aligning research teams globally.

If You're a Clinical Research Coordinator
Sequence patient visits, manage resource bookings, and avoid protocol deviations effortlessly with ClickUp.

If You're a Principal Investigator or Clinical Scientist
Plan study milestones, oversee data collection, and monitor progress through a centralized visual timeline.

How ClickUp Supports Clinical Study Success
Step Into Seamless Clinical Trial Management
Harness ClickUp Gantt Charts to master every aspect of your clinical research.
Centralize Protocols and Data
Keep study documents, patient data, and regulatory files linked directly to tasks in your timeline.
Plan Trial Phases Clearly
Visualize recruitment, intervention, follow-ups, and analysis phases for comprehensive oversight.
Standardize Study Procedures
Use templates, dependencies, and checklists to maintain protocol fidelity and reduce errors.
Enhance Team Collaboration
Assign roles with deadlines, track progress, and coordinate cross-functional teams effortlessly.
Convert Meetings into Actionable Plans
Turn discussion points into sequenced tasks on your Gantt chart ensuring follow-through.
Stay Ahead of Regulatory Deadlines
Receive automated reminders for IRB submissions, audits, and reporting milestones.