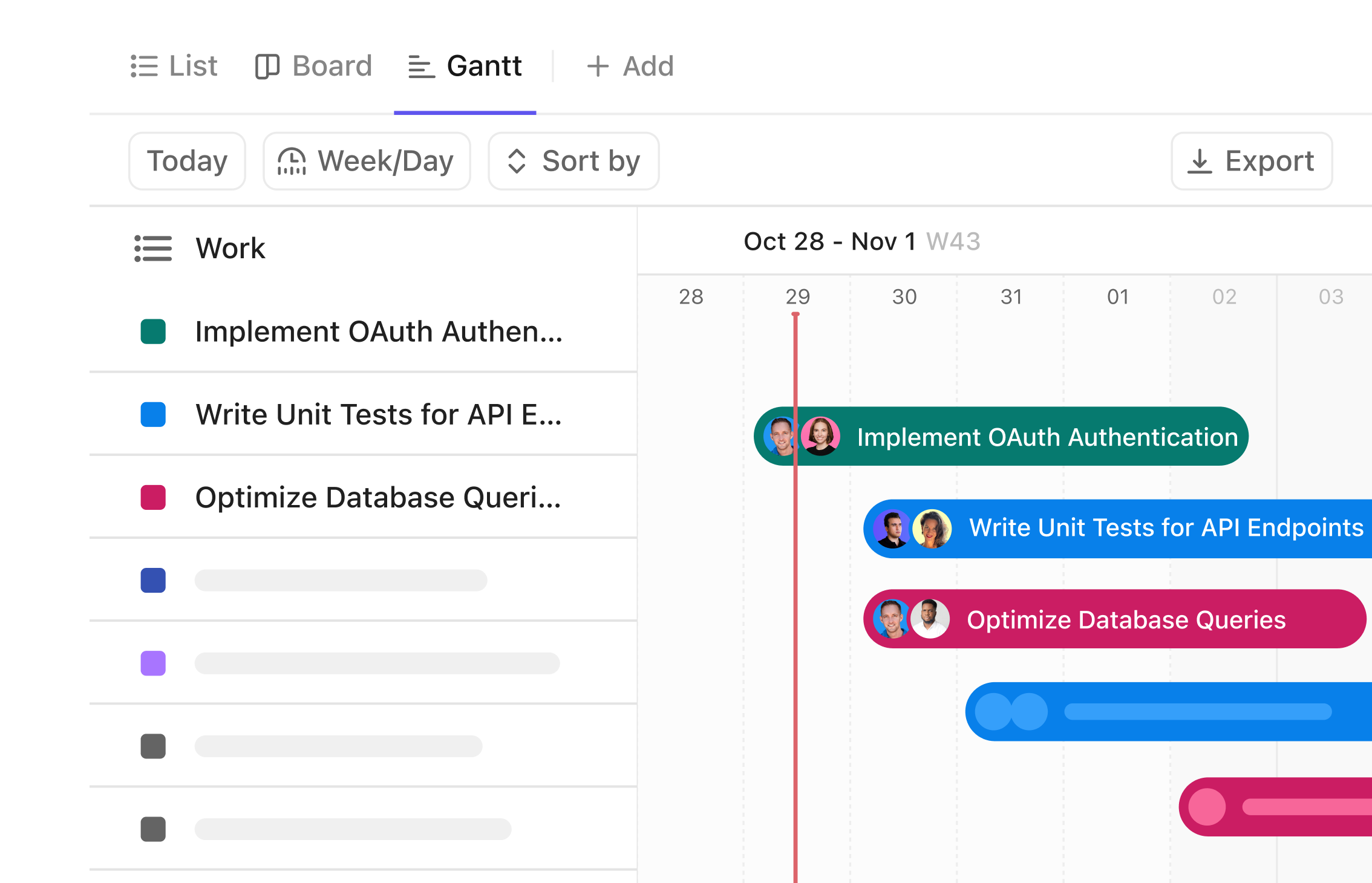
Gantt Chart for Clinical Data Managers
Visualize Clinical Trials with Precision Using Gantt Chart Software
Plan every phase of your clinical studies, coordinate cross-functional teams, and track critical milestones with a dynamic timeline that adapts to your workflow.
Trusted by the best
Why Choose Gantt Charts
Why Clinical Data Managers Rely on Gantt Chart Software
Managing clinical trials without a clear timeline risks costly delays and data inconsistencies. Without a visual plan, tasks overlap, responsibilities blur, and compliance deadlines sneak up unexpectedly.
Here’s where traditional approaches fall short for clinical data managers:
- Extended trial phases lose clarity — difficulty tracking recruitment, data collection, and analysis timelines.
- Data entry and validation become chaotic — lack of sequencing leads to errors and repeated work.
- Regulatory submission deadlines are missed — no centralized timeline to monitor key milestones.
- Team coordination suffers — unclear task ownership slows data processing.
- Resource allocation conflicts arise — equipment and personnel scheduling overlap without a visual plan.
- Progress tracking is manual and error-prone — making audits and reporting more complicated.
- Communication fragmented across platforms — increasing risk of misalignment.
- Risk management lacks foresight — no early alerts for potential bottlenecks or delays.
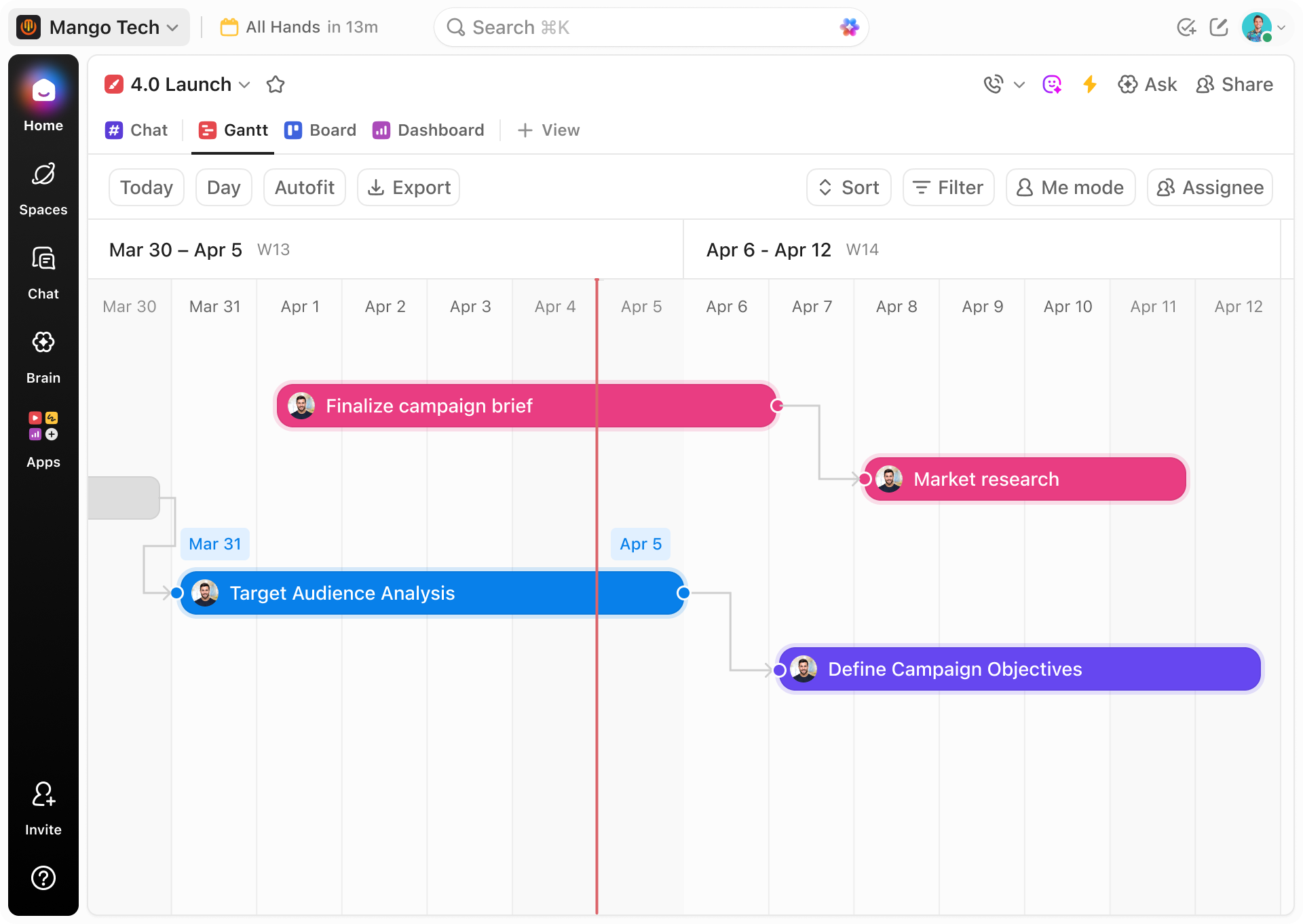
Traditional Methods vs ClickUp
Why Traditional Tracking Can't Match ClickUp Gantt for Clinical Data
Discover how ClickUp's Gantt charts bring clarity and control that spreadsheets and emails simply can’t provide.
Conventional Tracking Methods
- Data scattered across spreadsheets, emails, and notes
- No unified timeline causes missed dependencies
- Manual tracking increases risk of errors
- Team roles and responsibilities unclear
- Deadlines managed reactively
- Files and documents stored separately
ClickUp Gantt Chart Advantages
- Integrated tasks and timelines in one dashboard
- Visualize study phases with dependencies and milestones
- Automate alerts and reminders for key dates
- Assign clear responsibilities with deadlines
- Link protocols, CRFs, and reports directly to tasks
- Adapt timelines dynamically with real-time updates
Use Cases
How Gantt Chart Software Empowers Clinical Data Managers
Transform complex trial workflows into clear, manageable timelines that enhance data integrity and compliance.
#UseCase1
Eliminating Scheduling Conflicts in Multi-Site Trials
ClickUp’s Gantt charts visualize all site timelines, preventing overlaps and ensuring synchronized data collection.
#UseCase2
Maintaining Audit-Ready Documentation
Track every data validation step and protocol amendment with transparent timelines that satisfy regulatory requirements.
#UseCase3
Coordinating Cross-Functional Teams Efficiently
Assign tasks and deadlines clearly across data managers, statisticians, and monitors to keep everyone aligned.
#UseCase4
Monitoring Data Cleaning and Query Resolution
Visualize dependencies between data entry, cleaning, and query resolution to maintain study quality.
#UseCase5
Tracking Regulatory Submission Milestones
Automated reminders and timeline views ensure no FDA or EMA submission deadlines are missed.
#UseCase6
Optimizing Resource Allocation Across Trials
Map personnel and equipment availability to prevent conflicts and maximize efficiency.
#UseCase7
Managing Protocol Deviations with Clear Timelines
Document deviations and corrective actions in sequence to maintain compliance and audit trails.
#UseCase8
Streamlining Data Transfer and Integration
Coordinate data import/export phases visually to reduce errors and delays.
#UseCase9
Facilitating Real-Time Progress Reporting
Generate up-to-date visual reports to keep stakeholders informed and proactive.
Key Beneficiaries
Who Benefits Most From Gantt Chart Software in Clinical Data Management
Designed for professionals navigating the complexities of clinical trial timelines and data workflows.
Clinical Data Managers Coordinating Multi-Site Studies
Use ClickUp to align timelines, track progress across locations, and ensure consistent data quality globally.

Biostatisticians and Data Analysts
Visualize data processing pipelines and dependencies to streamline analysis and reporting phases.

Regulatory Affairs Specialists
Manage submission timelines and documentation workflows to meet all compliance deadlines effectively.

How ClickUp Supports Clinical Data Management
Step-by-Step: Streamline Your Clinical Data Workflow with ClickUp Gantt
Leverage ClickUp to bring clarity and control to every trial phase
Centralize Protocols and Data Collection Plans
Keep all trial documents, CRFs, and SOPs linked directly to tasks within your timeline for easy access.
Plan Trial Phases with Clear Milestones
Visualize recruitment, data cleaning, and analysis stages to monitor progress from start to finish.
Implement Task Dependencies and Sequencing
Ensure critical steps happen in the right order to maintain data integrity and trial compliance.
Assign Roles and Responsibilities Transparently
Define ownership for each task and phase to foster accountability across your clinical team.
Automate Alerts for Key Deadlines
Set reminders for data lock, monitoring visits, and submission dates to stay ahead of timelines.
Generate Real-Time Reports for Stakeholders
Provide sponsors and oversight committees with up-to-date visual progress reports to facilitate decision-making.