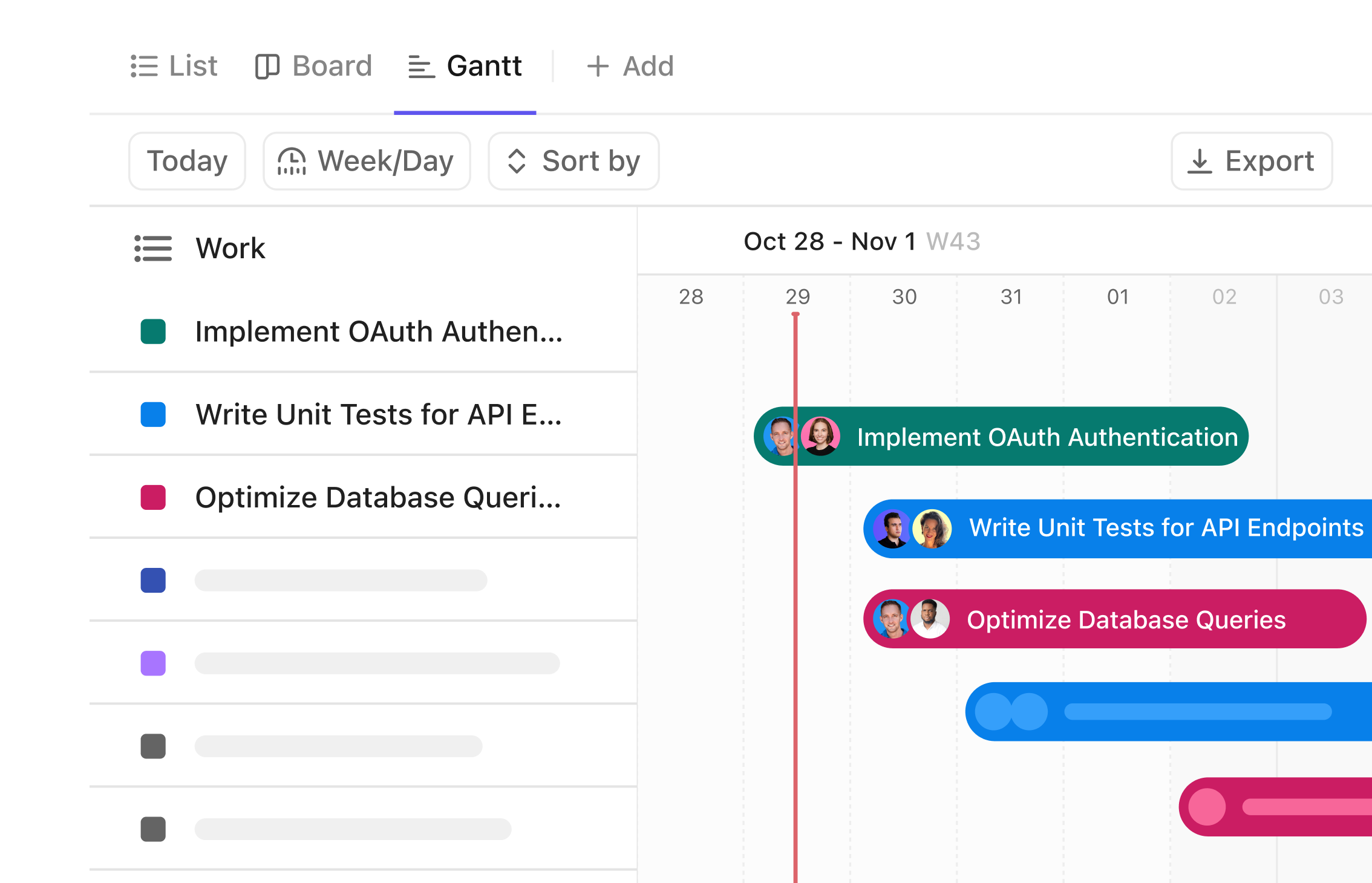
Gantt Chart Solutions for Biotech Innovators
Visualize Your Biotech Projects with Precision Gantt Charts
Navigate complex biotech development stages from discovery to product launch, synchronize teams across labs, and manage timelines with clarity using ClickUp’s Gantt Chart software—where your project’s DNA comes to life visually.
Trusted by the best
The Biotech Challenge
Why Biotech Professionals Rely on Gantt Chart Software
Biotech projects involve intricate processes and strict regulatory timelines. Without a dynamic timeline, progress stalls and critical steps slip through the cracks.
Common pitfalls in biotech project management include:
- Extended R&D phases blur critical milestones — losing sight of where each discovery phase stands.
- Regulatory submissions become bottlenecks — missing deadlines risks approval delays.
- Laboratory experiments lack sequencing — inconsistent protocols lead to costly errors.
- Cross-functional collaboration suffers — misaligned teams delay development.
- Resource allocation is inefficient — lab equipment and personnel overlap without clear scheduling.
- Data analysis pipelines lack transparency — making reproducibility and validation difficult.
- Progress tracking is fragmented — vital insights get lost across disconnected tools.
- Communication gaps cause delays — emails, spreadsheets, and meetings fail to synchronize efforts.
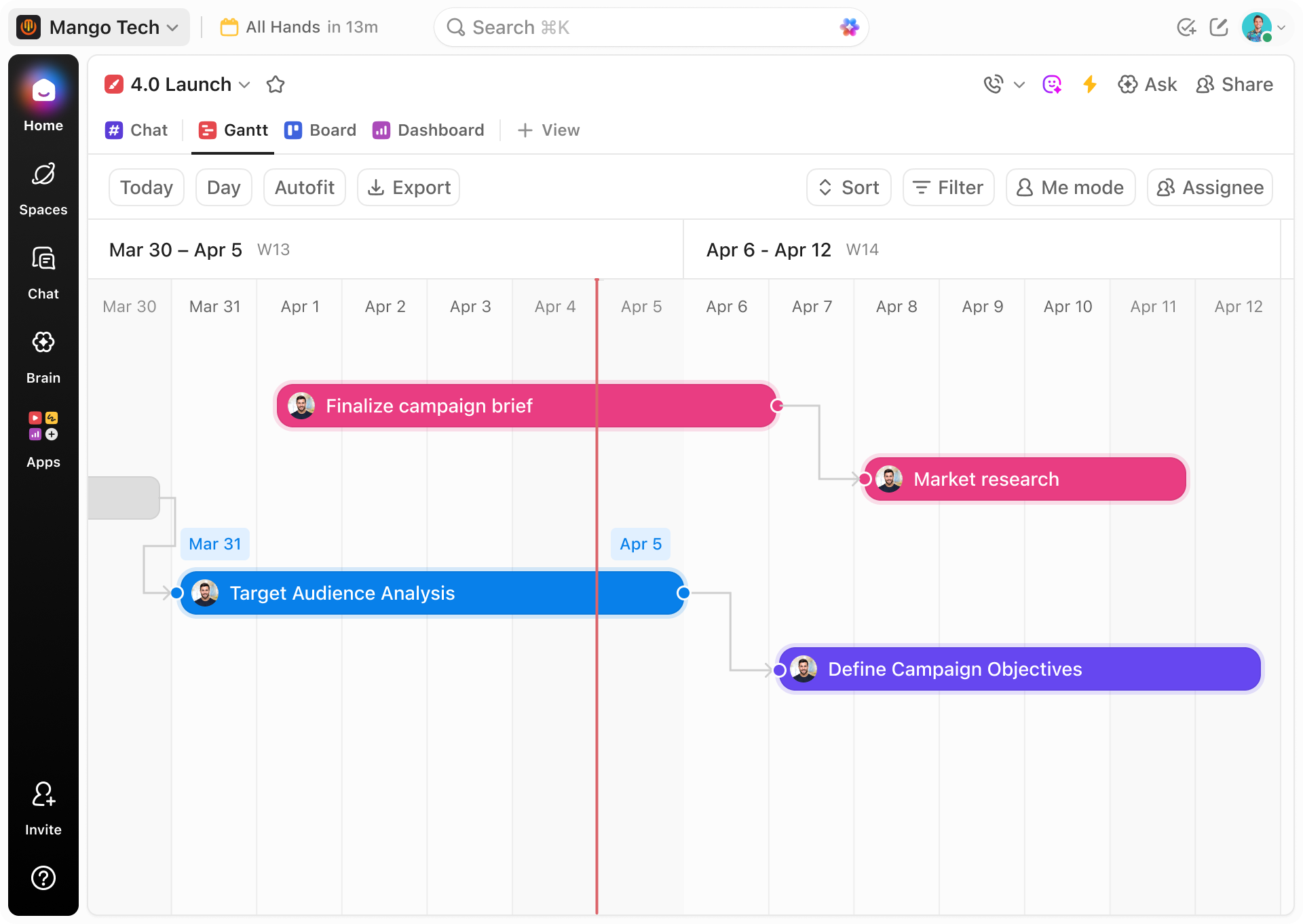
Conventional vs ClickUp Gantt
Why Traditional Biotech Planning Falls Short Compared to ClickUp Gantt
Discover how ClickUp’s Gantt Charts bring clarity and control to biotech project timelines where manual methods falter.
Conventional Methods
- Data scattered across spreadsheets, emails, and lab notebooks
- Milestones tracked manually → high risk of oversight
- Experiment steps unlinked → sequencing errors
- Collaboration fragmented across departments
- Regulatory deadlines tracked inconsistently
- Documentation dispersed in multiple systems
ClickUp Gantt Charts
- Unified tasks and timelines in a single visual interface
- Map experiments, regulatory steps, and milestones with start/end dates
- Visualize dependencies and critical paths
- Assign ownership with clear deadlines and notifications
- Automate alerts for upcoming regulatory submissions
- Attach protocols, data, and reports directly to tasks
Use Cases
How Gantt Chart Software Supports Biotech Industry Success
Explore how visual timelines transform biotech project management and accelerate innovation.
#UseCase1
Eliminating Visibility Gaps in Multi-Phase Development
ClickUp’s Gantt charts make complex R&D timelines transparent, enabling teams to identify bottlenecks and maintain steady progress.
#UseCase2
Ensuring Regulatory Compliance on Schedule
Map every regulatory step—clinical trials, submissions, approvals—with deadlines and reminders to avoid costly delays.
#UseCase3
Coordinating Cross-Disciplinary Teams Seamlessly
Align researchers, clinicians, and manufacturing teams with shared timelines and clear task ownership.
#UseCase4
Sequencing Lab Experiments to Prevent Errors
Define dependencies between protocols to ensure experiments follow precise order and maintain reproducibility.
#UseCase5
Managing Resource Allocation Efficiently
Schedule lab equipment, personnel, and materials to avoid conflicts and maximize utilization.
#UseCase6
Visualizing Data Analysis Pipelines End-to-End
Track bioinformatics workflows with linked tasks to guarantee validation and traceability.
#UseCase7
Meeting Product Launch Deadlines Reliably
Plan marketing, manufacturing, and distribution phases alongside development to ensure timely commercialization.
#UseCase8
Tracking Funding Milestones and Grant Deliverables
Keep grants and investor requirements front and center with automated alerts and progress visuals.
#UseCase9
Integrating ClickUp Brain for Predictive Project Insights
Leverage ClickUp Brain and Brain Max to anticipate delays, optimize schedules, and adapt plans proactively.
Key Beneficiaries
Who Gains the Most from ClickUp Gantt in Biotech
Ideal for professionals managing complex projects, cross-functional teams, and regulatory demands in biotech.
If You Lead Multi-Site Biotech Collaborations
ClickUp connects teams across labs and offices worldwide, synchronizing workflows despite differing time zones and protocols.

If You Manage Laboratory Operations
Coordinate experiments, manage equipment usage, and sequence protocols efficiently to minimize downtime and errors.

If You Are a Biotech Project Manager
Stay on top of deliverables, regulatory submissions, and stakeholder communication with a clear, updated timeline.

How ClickUp Supports Biotech Excellence
Step Into a New Era of Biotech Project Management
Harness ClickUp Gantt Charts to achieve clarity, compliance, and collaboration in every project phase.
Centralize Research & Development Data
Link protocols, datasets, reports, and regulatory documents to timeline tasks for immediate access and audit readiness.
Plan Clinical Trials with Confidence
Map trial phases, patient recruitment, and data collection with dependencies that ensure no step is overlooked.
Standardize Experimental Workflows
Use templates and dependencies to ensure consistency and reproducibility across experiments.
Visualize Cross-Team Collaboration
Assign tasks, monitor progress, and coordinate efforts between R&D, regulatory, and manufacturing teams.
Leverage ClickUp Brain for Proactive Planning
Utilize AI-driven insights from Brain and Brain Max to forecast risks and optimize project timelines.
Stay Ahead of Regulatory Deadlines
Automate reminders for submissions, audits, and compliance checks to maintain uninterrupted progress.