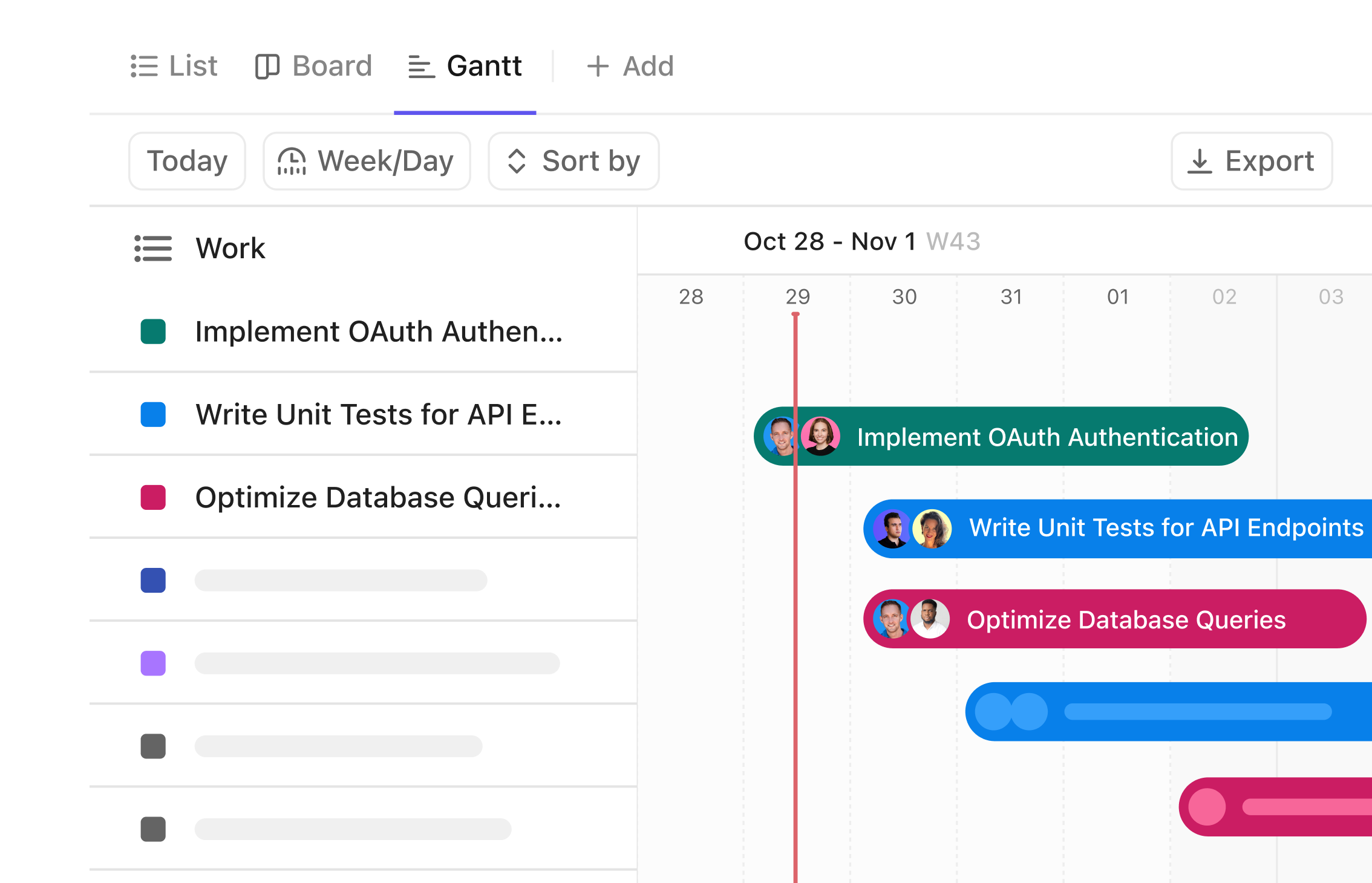
Gantt Chart Software Tailored for Biostatisticians
Transform Your Biostatistics Projects with Clear Visual Timelines
Visualize study phases from data collection to publication, coordinate multidisciplinary teams, and manage complex timelines effortlessly—all within a single, intuitive platform.
Trusted by the best
The Case for Gantt Charts
Why Biostatisticians Need Specialized Gantt Chart Software
Managing biostatistical studies without a clear timeline leads to missed nuances and delayed results. Without a centralized view, it’s easy to lose track of critical analyses and deadlines.
Common challenges faced without timeline management include:
- Complex study phases overlap — making it difficult to monitor progress and dependencies.
- Data analysis tasks become disjointed — unclear sequencing causes rework and errors.
- Collaborations across clinical and research teams falter — lack of clarity in roles and timelines.
- Regulatory submission dates creep up unexpectedly — risking compliance and funding.
- Tracking revisions and peer reviews is cumbersome — slowing publication timelines.
- Resource allocation for software, datasets, and personnel is inefficient — leading to bottlenecks.
- Communication scattered across emails and spreadsheets — causing misalignment.
- Integrating statistical programming with project milestones is manual and error-prone.
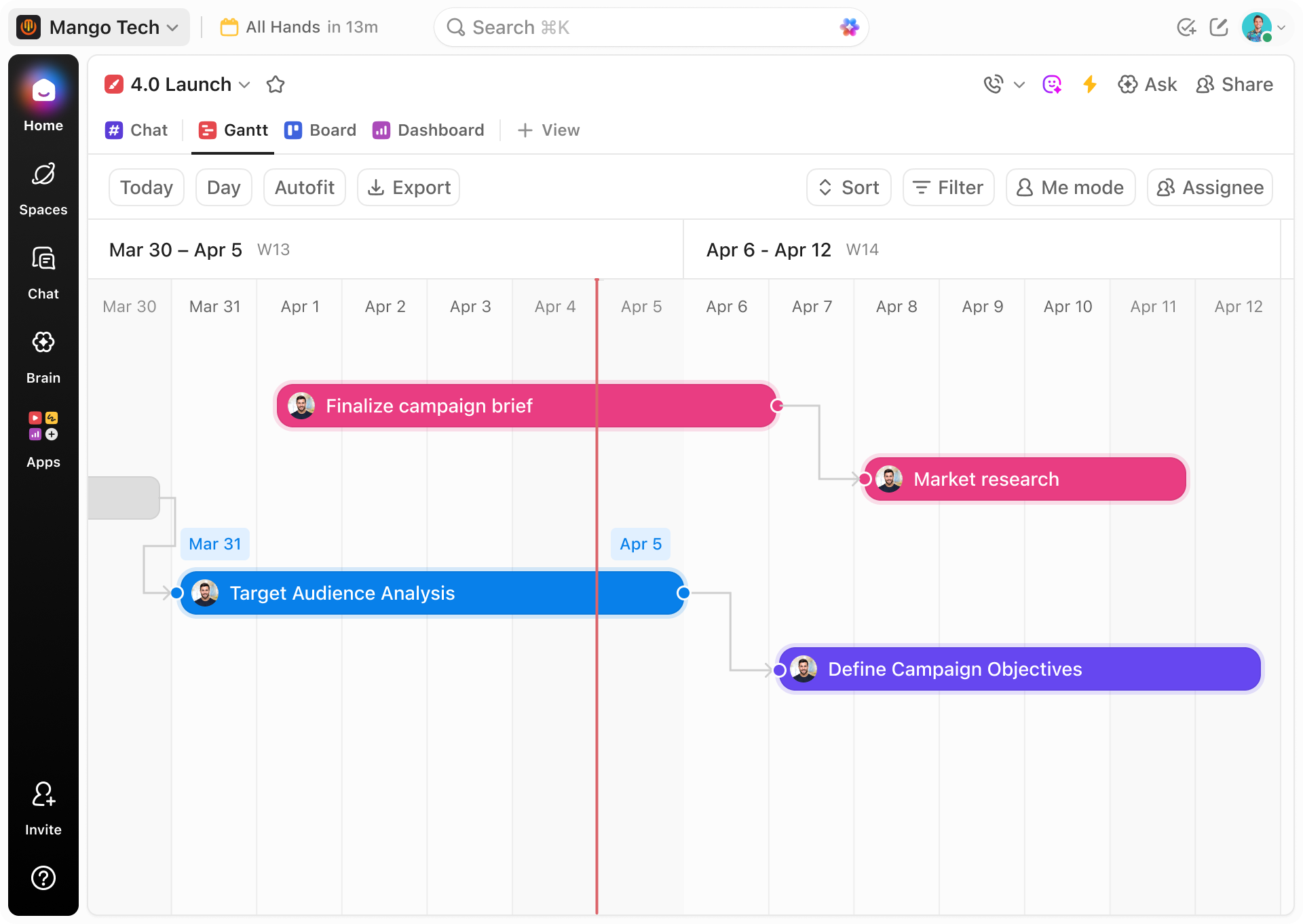
Traditional Approaches vs ClickUp Gantt
Why Conventional Methods Fall Short for Biostatisticians
Discover how ClickUp Gantt Charts consolidate your project complexities into a single source of truth, unlike fragmented traditional tools.
Traditional Methods
- Tasks tracked across disparate spreadsheets and emails
- Manual scheduling leads to overlapping analyses
- No clear view of data cleaning, modeling, and validation steps
- Collaboration challenges with unclear task ownership
- Missed regulatory and publication deadlines
- Disorganized storage of protocols and datasets
ClickUp Gantt Charts
- Unified timeline integrating all study phases and analyses
- Visual dependencies prevent sequencing errors
- Assign roles and deadlines transparently
- Automated reminders for regulatory and submission milestones
- Centralized documents linked directly to tasks
- Real-time collaboration with Brain and Brain Max AI-powered insights
Use Cases
Unlock the Power of Gantt Charts for Biostatistical Research
Explore how a dynamic timeline brings clarity and precision to your biostatistics workflow.
#UseCase1
Eliminate Blind Spots in Clinical Trial Timelines
Visualize every trial phase and milestone clearly, so delays and bottlenecks are identified early and addressed proactively.
#UseCase2
Build Transparent Data Analysis Pipelines
Map out data cleaning, transformation, modeling, and validation steps with dependencies that maintain analytic integrity.
#UseCase3
Track Regulatory Submissions and Compliance Dates
Stay ahead of IRB approvals, FDA submissions, and reporting deadlines with automated alerts and timeline visibility.
#UseCase4
Coordinate Multidisciplinary Teamwork Seamlessly
Assign ownership and deadlines across statisticians, clinicians, and data managers to keep everyone aligned.
#UseCase5
Manage Resource Allocation Efficiently
Plan software licenses, compute time, and personnel availability to optimize study throughput.
#UseCase6
Streamline Manuscript Preparation and Peer Review
Track drafting, revisions, and submission processes to accelerate publication timelines.
#UseCase7
Visualize Longitudinal Study Progression
Use Gantt charts to monitor ongoing cohort studies and longitudinal data collection phases.
#UseCase8
Integrate Statistical Programming Workflows
Link R, SAS, or Python script milestones directly to project timelines for seamless progress tracking.
#UseCase9
Leverage AI Insights with ClickUp Brain and Brain Max
Gain predictive analytics on task durations and potential risks to make data-driven project adjustments.
Key Beneficiaries
Who Benefits Most from ClickUp Gantt Charts in Biostatistics
Ideal for professionals managing complex studies, regulatory timelines, and cross-functional teams.
If You’re a Clinical Trial Statistician
ClickUp synchronizes study phases across sites and teams, ensuring compliance and timely data analysis.

If You’re a Data Scientist in Biostatistics
Sequence data preprocessing, modeling, and validation tasks with clear dependencies and milestones.

If You’re a Biostatistics Project Manager
Manage resources, coordinate collaborators, and monitor regulatory deadlines effortlessly on one timeline.

How ClickUp Empowers You
Step-by-Step Guide to Mastering ClickUp Gantt for Biostatistics
From setup to execution, streamline your biostatistics projects in six practical steps.
Centralize All Study Documentation
Attach protocols, datasets, analysis scripts, and reports directly to tasks for immediate access.
Define Project Phases Clearly
Outline study design, data collection, analysis, and reporting with start and end dates.
Establish Task Dependencies
Set up sequences so each analytic step triggers the next, preventing workflow errors.
Assign Roles and Responsibilities
Clarify ownership for every task, from data cleaning to manuscript drafting.
Leverage AI-Powered Risk Predictions
Use Brain and Brain Max to identify potential delays and optimize project pacing.
Monitor Progress with Automated Alerts
Receive notifications on approaching deadlines, regulatory milestones, and task completions.