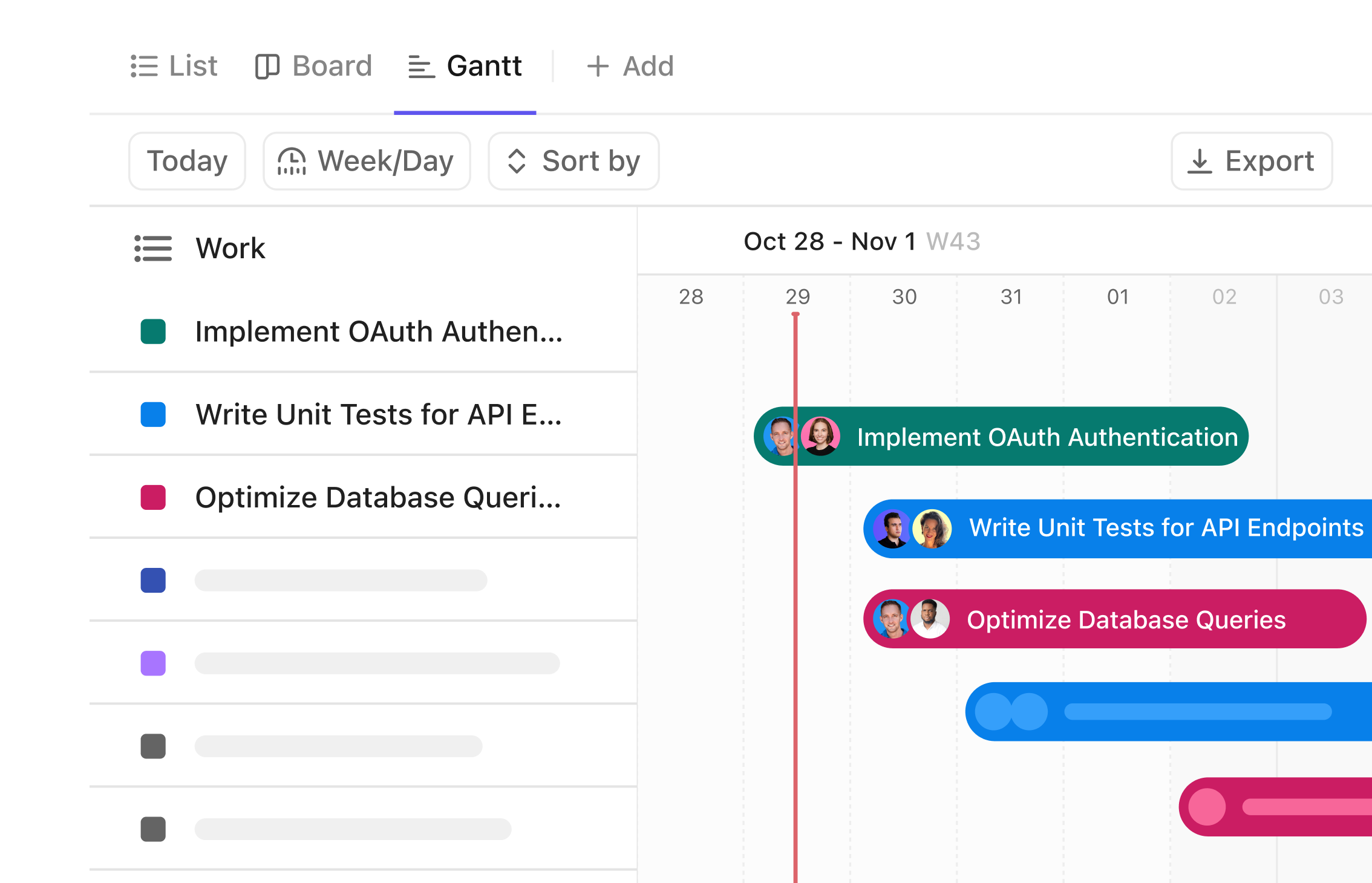
Gantt Chart Solutions for Biomedical Engineers
Streamline Biomedical Projects with Precision Gantt Charts
Visualize complex medical device timelines, coordinate cross-functional teams, and manage every development phase with clarity—letting you focus on innovation, not chaos.
Trusted by the best
Why Choose Gantt Charts
Why Biomedical Engineers Benefit from Gantt Chart Software
Biomedical projects involve intricate processes, strict regulations, and tight deadlines. Without a clear timeline, tasks overlap, compliance risks grow, and progress stalls.
Here’s where traditional approaches fall short in biomedical engineering:
- Project phases blur together — design, testing, and validation timelines become confusing.
- Regulatory milestones get missed — FDA and ISO deadlines slip through the cracks.
- Prototype iterations become disorganized — unclear sequencing causes delays and rework.
- Team communication fragments — engineers, clinicians, and manufacturers lose sync.
- Resource scheduling conflicts arise — lab equipment and testing facilities overbooked.
- Progress tracking lacks transparency — stakeholders can’t easily see project status.
- Data management is scattered — test results, documentation, and reports spread across platforms.
- Risk management becomes reactive — potential issues surface too late to mitigate effectively.
Comparing Methods
Why Traditional Project Tracking Falls Short in Biomedical Engineering
Discover how ClickUp’s Gantt Charts provide biomedical teams the structure and visibility that manual methods can’t match.
Traditional Approaches
- Reliance on spreadsheets and emails causes scattered information
- Manual tracking leads to missed dependencies and errors
- Regulatory timelines tracked in separate systems
- Prototype development stages lack clear ownership
- Communication gaps between cross-disciplinary teams
- Resource allocation handled informally without visualization
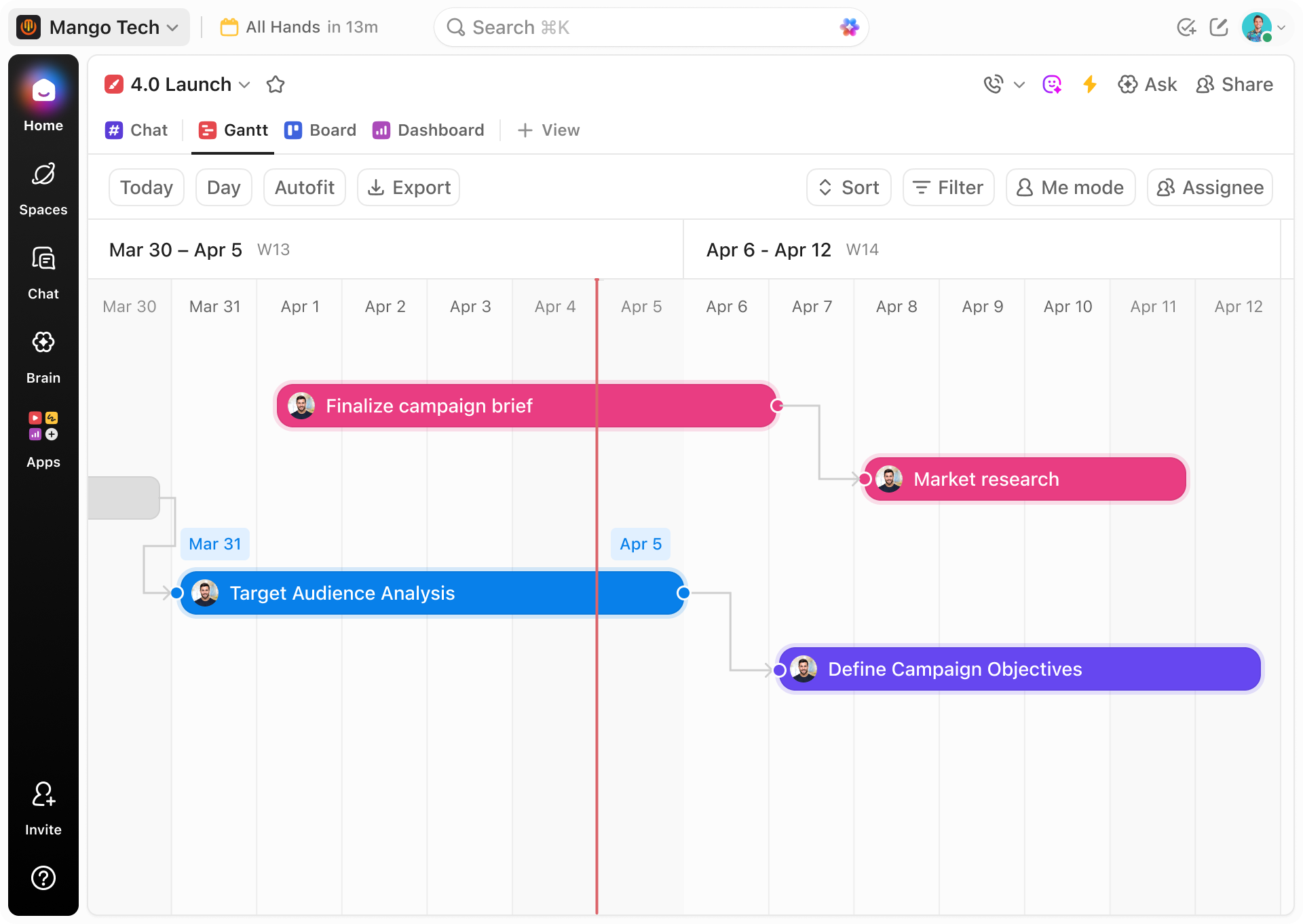
ClickUp Gantt Charts
- Integrate timelines, tasks, and documentation in a single view
- Visualize dependencies, milestones, and critical paths
- Automate alerts for regulatory and compliance deadlines
- Assign responsibilities with clear deadlines and progress tracking
- Facilitate collaboration across engineering, clinical, and manufacturing teams
- Manage lab and testing resources with real-time scheduling
Use Cases
How Gantt Chart Software Empowers Biomedical Engineers
Harness timeline visibility to reduce errors, meet compliance, and accelerate biomedical innovation.
#UseCase1
Clarifying Complex Device Development Timelines
Use ClickUp’s Gantt charts to map design, prototyping, testing, and manufacturing phases—removing ambiguity and highlighting critical deadlines.
#UseCase2
Ensuring Regulatory Compliance with Timely Milestones
Track FDA submissions, ISO certifications, and clinical trial phases with automated reminders and clear approval pathways.
#UseCase3
Coordinating Cross-Disciplinary Teams Seamlessly
Align engineers, clinicians, and quality assurance teams on shared timelines to enhance collaboration and reduce miscommunication.
#UseCase4
Managing Prototype Iterations Efficiently
Visualize iteration cycles with dependencies to prevent costly rework and ensure each version meets quality standards.
#UseCase5
Scheduling Lab Resources and Testing Slots
Avoid conflicts by tracking equipment availability and test schedules within your project timeline.
#UseCase6
Documenting Validation and Verification Processes
Attach test results, approvals, and reports directly to Gantt tasks to maintain a comprehensive audit trail.
#UseCase7
Tracking Risk Mitigation Activities
Identify potential bottlenecks and address them proactively by visualizing risk management tasks and dependencies.
#UseCase8
Streamlining Clinical Trial Timelines
Plan patient enrollment, data collection, and analysis phases to keep trials on schedule and within budget.
#UseCase9
Facilitating Product Launch Coordination
Coordinate marketing, regulatory approval, and distribution tasks to ensure a smooth market entry.
Key Beneficiaries
Who Gains the Most From ClickUp Gantt Charts
Biomedical professionals managing complex projects, compliance, and collaboration.
If You Lead a Medical Device Development Team
Coordinate design, engineering, and testing tasks across departments and partners globally to maintain alignment and transparency.

If You’re a Quality Assurance Specialist
Track compliance checklists, validation protocols, and regulatory deadlines to ensure nothing is overlooked.

If You’re a Biomedical Research Engineer
Manage experiment schedules, prototype iterations, and data analysis milestones on a clear, adaptable timeline.

How ClickUp Supports Biomedical Innovation
Step-by-Step Guide to Using ClickUp Gantt Charts for Biomedical Engineering
From initial concept to market launch, organize your projects with confidence.
Centralize Project Documentation
Keep all design files, test data, and regulatory documents linked directly to tasks for easy access.
Phase Your Development Process
Break down projects into stages—design, prototyping, testing, validation—with clear timelines.
Set and Track Dependencies
Map how tasks impact each other to avoid bottlenecks and ensure smooth progression.
Assign Roles and Responsibilities
Clarify ownership for every task and milestone to enhance accountability.
Leverage Automation and Alerts
Use ClickUp Brain and Brain Max to predict delays and automate reminder notifications.
Review Progress with Visual Insights
Monitor timelines, completion rates, and resource allocation in real time to stay on track.