ClickUp CRM Tailored for Clinical Trials
Empower Clinical Trial Managers with Advanced CRM Software
Coordinate study sites, monitor participant data, streamline communications, and track trial milestones effortlessly — all within one unified platform, not scattered spreadsheets.

Trusted by the best
Why Choose CRM
The Clinical Trial Manager’s Challenge Without a CRM
Managing clinical trials without a dedicated CRM is like navigating complex protocols relying solely on memory and emails. Here’s where manual processes typically fail clinical trial managers:
- Site and investigator details become disorganized — losing track of contacts, responsibilities, and communications.
- Participant enrollment lacks transparency — inconsistent tracking of recruitment status and consent documentation.
- Regulatory deadlines slip through cracks — missing critical submissions and audits.
- Vendor and CRO coordination gets complicated — fragmented follow-ups and unclear accountability.
- Data queries and protocol deviations are hard to monitor — no centralized log or audit trail.
- Communication spans multiple disconnected channels — hindering real-time collaboration and documentation.
- Onboarding new team members is inefficient — absence of consolidated project history and contact details.
Traditional vs ClickUp CRM
Why Legacy Systems Fall Short for Clinical Trial Management
Discover how ClickUp CRM offers complete oversight that spreadsheets and emails simply can’t provide.
Conventional Methods
- Contacts scattered across emails, notes, and spreadsheets
- No unified tracking of site progress or participant recruitment
- Regulatory and submission deadlines managed manually
- Limited visibility into vendor and CRO activities
- Communication fragmented across platforms
- No automation for follow-ups or alerts
- Difficulty maintaining audit trails
- Time-consuming onboarding processes
ClickUp CRM
- Centralized database for sites, investigators, vendors, and participants
- Visual pipelines for recruitment, regulatory submissions, and milestones
- Automated reminders for compliance deadlines and follow-ups
- Integrated communication logs with attachments and notes
- Real-time dashboards for monitoring trial progress
- Task automation to assign next steps and owners
- Comprehensive audit trails and documentation storage
- Streamlined onboarding with full project histories
Use cases
How CRM Software Streamlines Clinical Trial Management
ClickUp CRM keeps your clinical trial workflow transparent, compliant, and on schedule.
#UseCase1
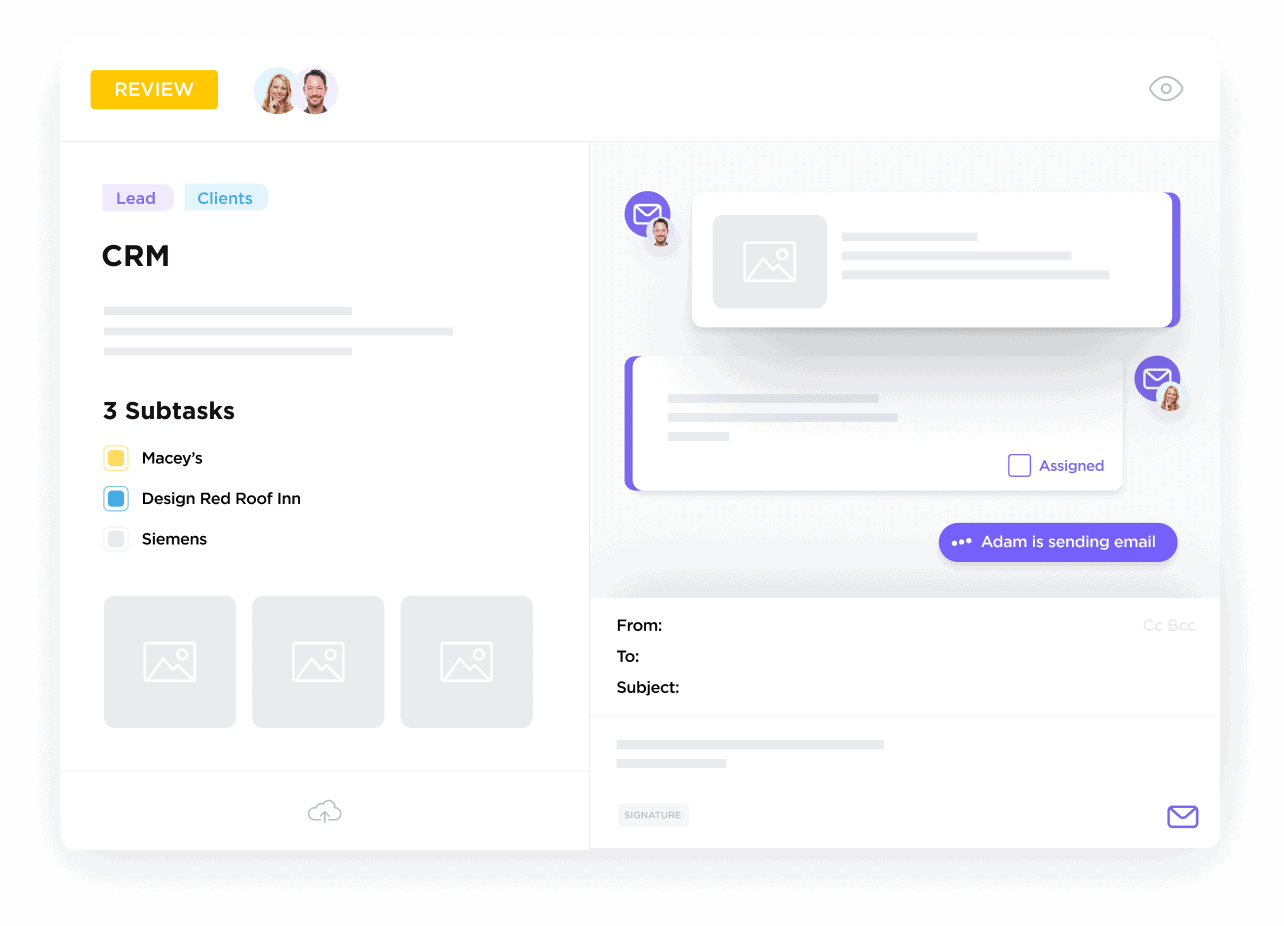
Centralizing Study Sites and Investigators
Organize every site contact, investigator credential, and activity log, so you always know who’s responsible and what’s next — reducing communication gaps.
#UseCase2
Managing Participant Recruitment & Retention
Track recruitment status, consent forms, and follow-up schedules with automation to keep enrollment steady and compliant.
#UseCase3
Overseeing Regulatory Submissions
Monitor every submission deadline, document version, and approval status to maintain compliance and avoid costly delays.
#UseCase4
Coordinating Vendors and CROs Seamlessly
Centralize all vendor contracts, orders, and communications to ensure timely deliveries and clear accountability.
#UseCase5
Tracking Protocol Deviations and Queries
Log and manage data queries, deviations, and corrective actions with full traceability and audit-ready records.
#UseCase6
Facilitating Team Collaboration Across Locations
Keep cross-functional teams aligned with shared tasks, comments, and real-time updates regardless of geography.
#UseCase7
Automating Follow-Ups and Alerts
Leverage ClickUp Brain to trigger timely reminders and automate next steps based on trial milestones and communications.
#UseCase8
Integrating Protocol Documents and Data
Attach study protocols, consent forms, and monitoring reports directly to CRM records for easy access and version control.
#UseCase9
Converting Meetings Into Action Items
Turn discussions into tasks with owners and deadlines, ensuring nothing from site visits or team calls gets overlooked.
Key Beneficiaries
Clinical Trial Professionals Who Benefit Most from ClickUp CRM
Ideal for managers juggling multiple sites, complex protocols, and extensive communication.
If You’re Managing Multinational Trials
ClickUp CRM provides a centralized hub, harmonizing communications, timelines, and documentation across countries and institutions.
If You’re Overseeing Participant Recruitment
Keep recruitment pipelines transparent, automate follow-ups, and maintain audit-ready records effortlessly.
If You’re Coordinating CROs and Vendors
Monitor contracts, deliverables, and communications in one place to ensure quality and timelines are met.

Benefits
How ClickUp CRM Transforms Clinical Trial Management
Bring all trial contacts, tasks, and data into one cohesive system.
Create a Centralized Contact Repository
Consolidate investigators, sites, vendors, and participants with detailed profiles and activity logs.
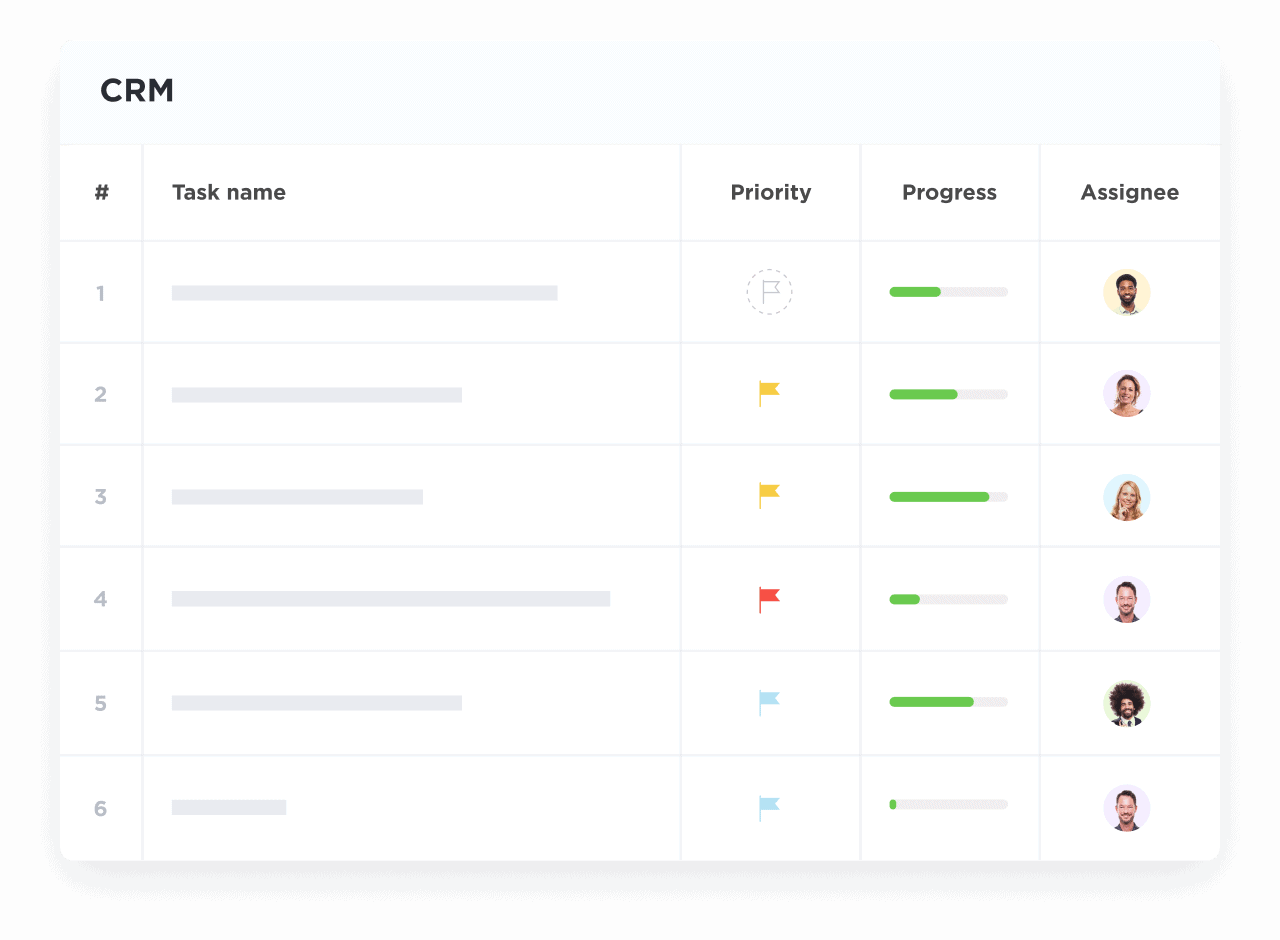
Visualize Trial Progress with Custom Pipelines
Track recruitment, regulatory milestones, and vendor stages with intuitive dashboards.
Capture Comprehensive Communication Histories
Log emails, calls, and meetings linked directly to trial items for full context.
Convert Conversations Into Actionable Tasks
Assign next steps with deadlines, owners, and automated reminders.
Attach Protocols and Regulatory Documents
Keep all essential files accessible and version-controlled within the CRM.
Stay Ahead with Alerts and Dashboards
Monitor trial status and deadlines proactively, reducing compliance risks.