Streamline Clinical Research with the Leading CRM Software

Elevate Clinical Research Through Effective CRM Management
Conducting clinical studies without CRM software is like navigating a complex trial without a protocol — confusion and errors multiply quickly.
Common pitfalls when managing clinical research manually include:
- Unclear participant tracking — losing sight of recruitment status, consent forms, and follow-up schedules.
- Grant and funding deadlines missed — critical submissions slip through without centralized reminders.
- Collaborator communication breakdowns — inconsistent updates between research sites and teams.
- Regulatory documentation scattered — difficulty locating IRB approvals, amendments, and audits.
- Data collection and analysis delays — fragmented records stall study progress.
- Stakeholder engagement falters — sponsors, vendors, and oversight committees remain out of sync.
- Onboarding new researchers inefficient — lack of accessible project history and contacts.
- Multiple platforms cause confusion — emails, spreadsheets, and chat apps lead to lost context and duplicated work.
Transform Your Clinical Research with ClickUp CRM
Discover how ClickUp CRM empowers clinical researchers far beyond conventional tools.
Traditional Methods
- Participant info scattered across spreadsheets and emails
- No centralized tracking of funding proposals
- Limited oversight on recruitment and retention
- Regulatory documents stored in multiple places
- Communication gaps across research teams
- No automated reminders for critical tasks
- Difficulty monitoring vendor and sponsor interactions
- Manual follow-ups prone to errors
ClickUp CRM Advantages
- Consolidate participant, collaborator, and stakeholder data in one CRM
- Visual grant and protocol pipelines with deadline alerts
- Automated participant outreach and status tracking
- Centralized regulatory documentation with audit trails
- Real-time team communication and task assignments
- Integrated reminders minimize missed deadlines
- Track vendor and sponsor communications comprehensively
- Turn meetings into actionable tasks with owners and timelines
Unlock Clinical Research Potential with CRM Use Cases
Centralize Clinical Team and Participant Management
Manage Grant Applications and Study Milestones
Streamline Participant Enrollment and Follow-Up
Capture Regulatory and Ethics Committee Feedback
Coordinate Multi-Site Clinical Trials
Manage Vendor and Equipment Logistics
Monitor Journal Submissions and Peer Reviews
Enhance Industry and Sponsor Collaboration
Convert Meetings Into Actionable Clinical Tasks
Who Benefits Most from ClickUp CRM in Clinical Research?
For Global Clinical Trial Coordinators
For Laboratory-Based Clinical Researchers
For Clinical Research Students and Fellows

Optimize Clinical Research Workflows with ClickUp CRM
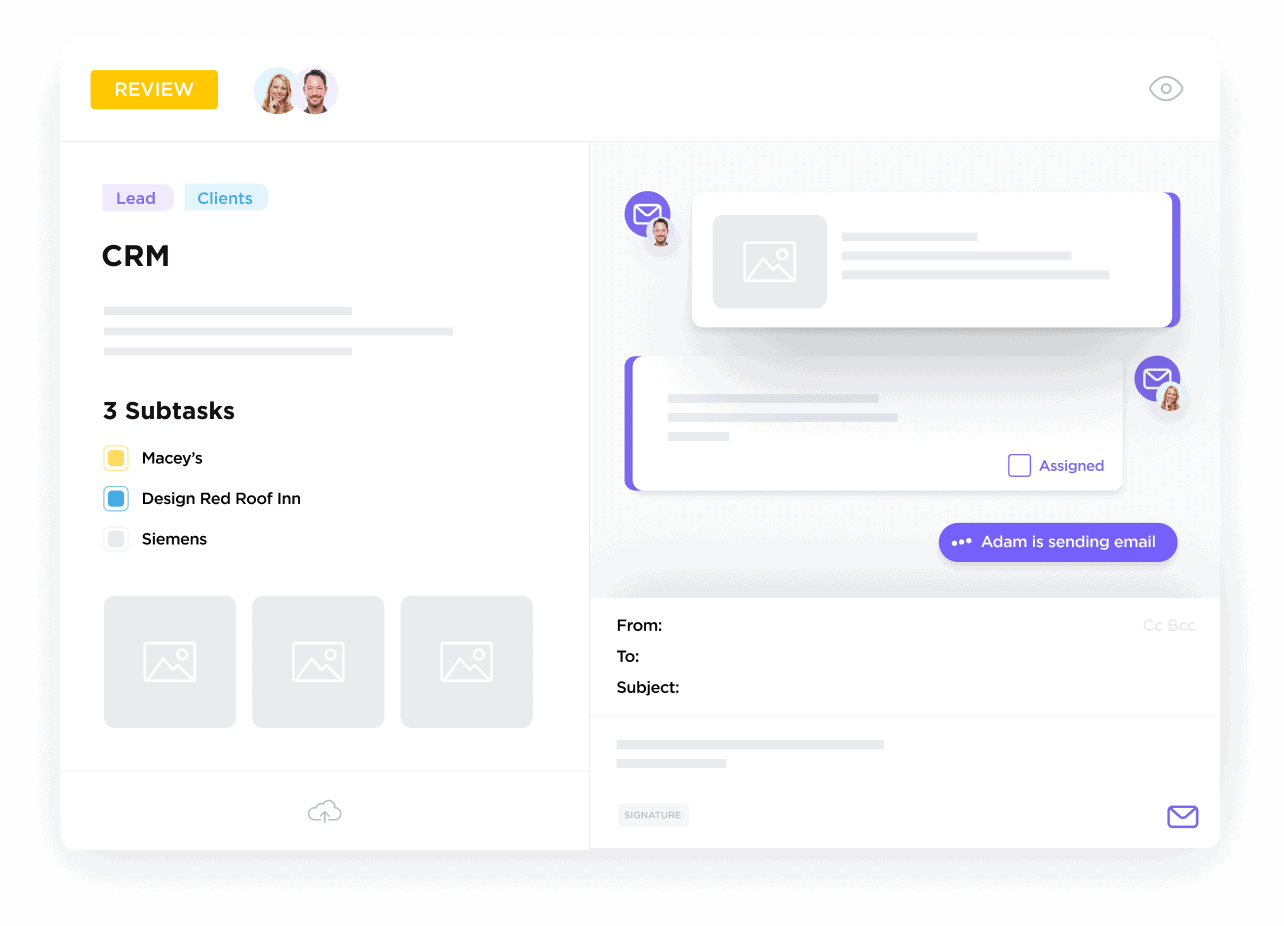
Create a Comprehensive Clinical Contact Database
Organize investigators, participants, sponsors, vendors, and ethics boards with detailed custom fields and activity logs.
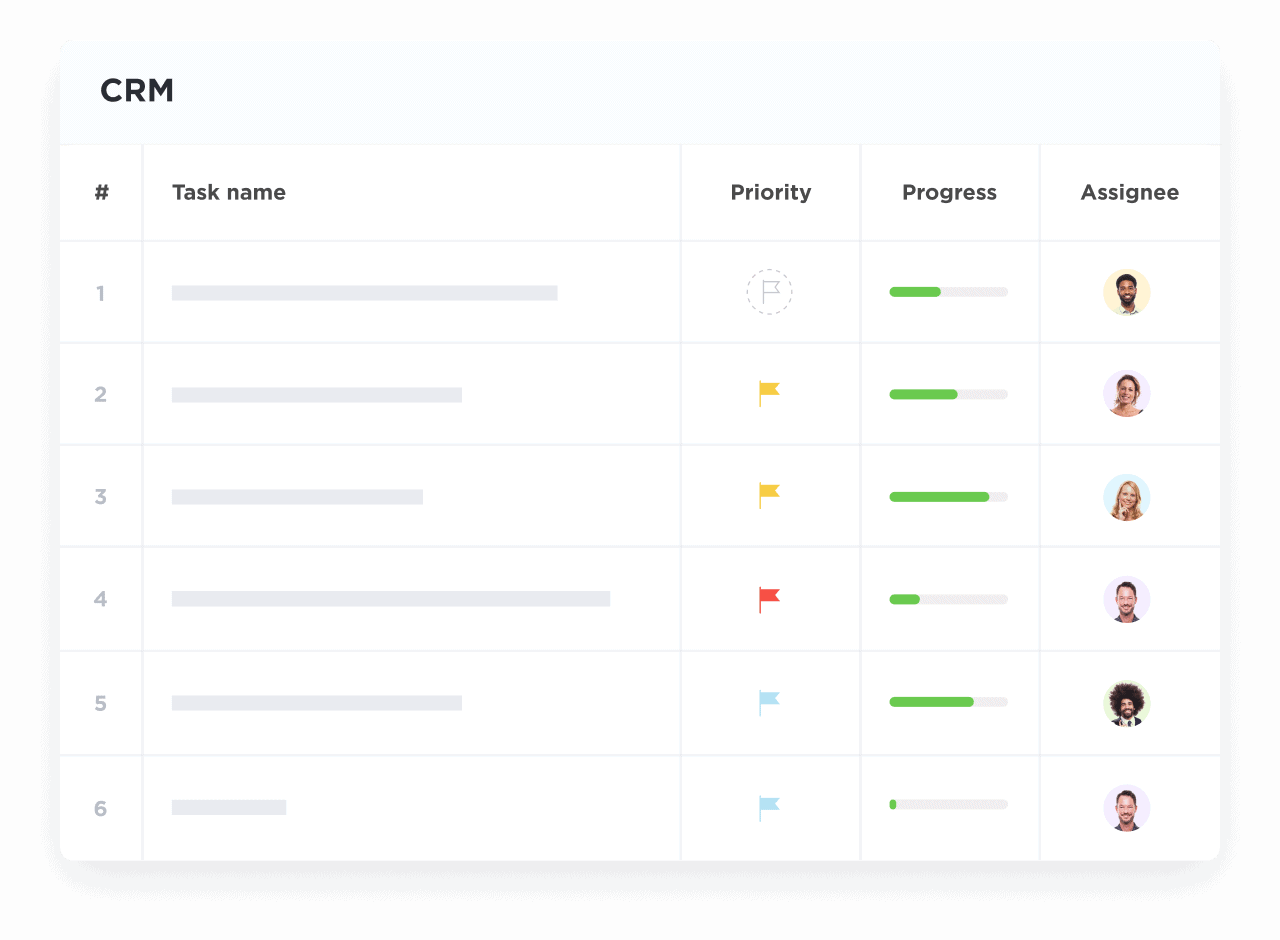
Visualize Every Research Process Stage
Track patient recruitment, grant progress, and regulatory approvals with intuitive pipelines.
Maintain Complete Communication History
Log emails, calls, decisions, and documents to preserve essential context across trials.
Transform Discussions into Manageable Tasks
Convert follow-ups into assigned tasks with deadlines, reminders, and automation.
Integrate Critical Documents and Notes
Attach protocols, consent forms, data sheets, and meeting minutes directly within the CRM.
Stay Ahead with Automated Alerts and Dashboards
Monitor upcoming deadlines, revisions, and funding milestones with real-time visibility.