Elevate Your Biotech Startup with Tailored CRM Solutions

The Critical Need for CRM in Biotech Startups
Managing a biotech startup without a CRM is like handling complex experiments with no lab notebook — essential details slip through the cracks.
Challenges that arise without a dedicated CRM include:
- Disjointed stakeholder communication — losing track of investors, partners, and regulatory contacts.
- Funding opportunities overlooked — missing critical deadlines and documentation for grants and investments.
- Clinical trial participant coordination falters — scattered outreach and inconsistent data capture.
- Collaboration between R&D, regulatory, and business teams stalls — lack of centralized updates and task ownership.
- Regulatory compliance documentation risks — fragmented records increase audit vulnerabilities.
- Vendor and equipment management becomes chaotic — no clear tracking of orders, contracts, and maintenance.
- Onboarding new team members without context — knowledge silos slow down progress.
Traditional Tools Fall Short for Biotech Startups
Traditional Approaches
- Contacts scattered across emails, spreadsheets, and notes
- No centralized tracking of investor or regulatory communications
- Inconsistent clinical trial participant management
- Manual follow-ups prone to errors
- Fragmented document storage across platforms
- Lack of automation for reminders and compliance
- Limited visibility into project milestones
- Difficulty adapting to biotech-specific workflows
ClickUp CRM Tailored for Biotech
- Consolidate contacts: investors, partners, participants, vendors all in one place
- Log all communications, calls, and regulatory correspondence with timelines
- Visualize funding and clinical trial pipelines with customizable dashboards
- Automate reminders for follow-ups, submissions, and compliance deadlines
- Attach research data, contracts, and regulatory documents directly to records
- Link tasks, notes, and next steps to stakeholders and projects
- Monitor milestones, ownership, and progress in real-time
- Leverage ClickUp Brain and Brain Max for smart insights and workflow optimization
Unlock Biotech Startup Success with CRM Use Cases
Centralize Investors, Regulators & Partners
Manage Funding Pipelines with Precision
Coordinate Clinical Trial Participants
Track Regulatory Submissions & Feedback
Keep detailed logs of submissions, agency feedback, and revision tasks to ensure compliance and audit readiness.
Facilitate Cross-Functional Team Collaboration
Align R&D, business development, and regulatory teams with shared dashboards, task assignments, and real-time updates.
Oversee Vendor & Equipment Management
Monitor orders, maintenance schedules, and communications to keep your lab running efficiently.
Monitor Intellectual Property & Licensing
Track patent applications, licensing agreements, and related correspondence all in one place.
Streamline Industry Collaborations & Partnerships
Manage NDAs, joint projects, and deliverables with automated follow-ups and clear accountability.
Convert Meetings into Actionable Tasks
Automatically link meeting notes to contacts and projects, assigning owners and deadlines to next steps.
Who Gains the Most from ClickUp CRM in Biotech?
If You’re Leading a Multi-Disciplinary Biotech Team
Use ClickUp CRM to unify communication across R&D, regulatory affairs, business development, and external partners, bridging time zones and expertise.
If You’re Managing Clinical Trials
Track participant recruitment, consent, follow-ups, and regulatory compliance seamlessly from a single dashboard.
If You’re a Startup Founder or CEO
Keep investor relations, funding pipelines, partnerships, and team progress transparent and easily accessible to drive growth.

How ClickUp CRM Transforms Biotech Startup Operations
Create a Comprehensive Stakeholder Network
Manage investors, regulators, collaborators, and vendors with customizable fields and detailed interaction histories.
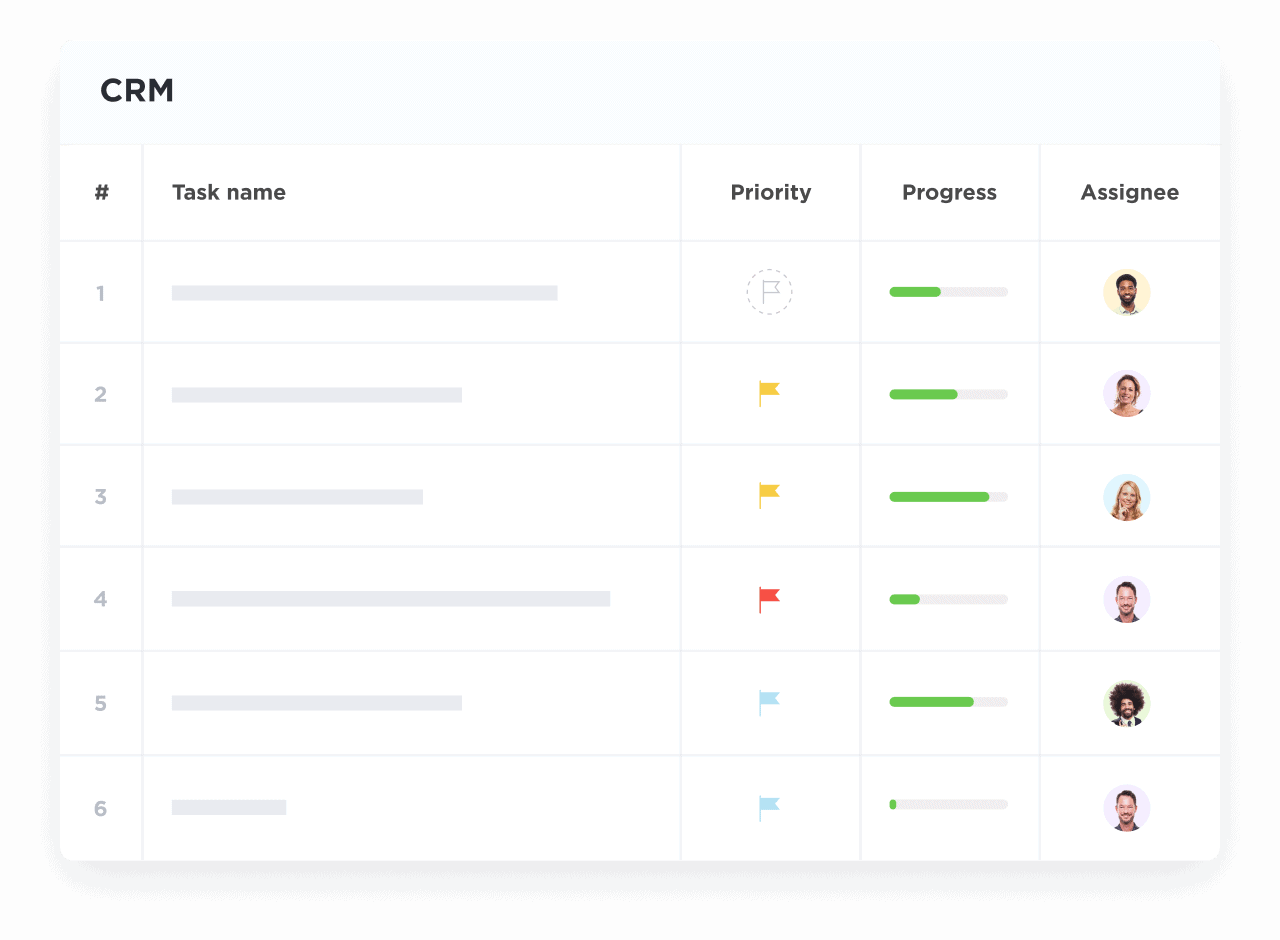
Visualize Funding and Research Pipelines
Track milestones from funding applications to clinical trial phases with clear, actionable views.
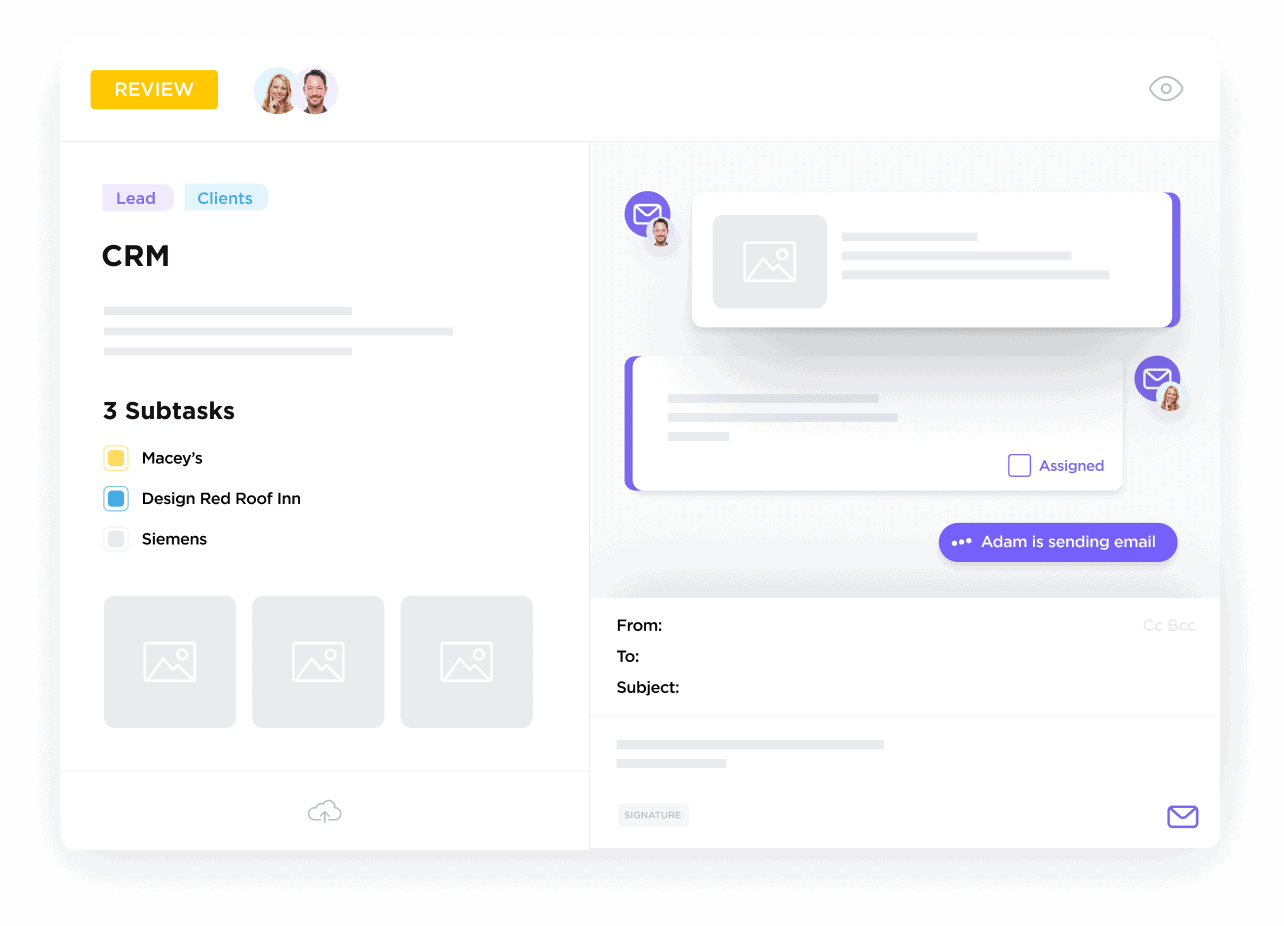
Maintain Complete Communication Logs
Sync emails, calls, and notes to provide full context for every project and stakeholder.
Turn Conversations into Manageable Tasks
Assign follow-ups with deadlines and notifications to keep your team accountable.
Attach Critical Documents Securely
Link proposals, regulatory filings, consent forms, and IP documents directly to CRM entries.
Stay Alert with Smart Reminders and Dashboards
Never miss a compliance deadline or funding opportunity with proactive notifications and real-time insights.