Top AI Prompts for Clinical Trial Data Analysis

AI Prompts Revolutionizing Clinical Trial Data Analysis
Managing clinical trial data isn't just about collecting numbers—it's about making sense of complex information to drive breakthroughs.
From patient recruitment metrics to adverse event tracking and regulatory submissions, clinical trial data analysis encompasses numerous datasets and strict timelines. AI prompts are becoming indispensable in this landscape.
Clinical teams leverage AI to:
- Quickly identify patterns and anomalies in trial results
- Generate detailed reports and data summaries with minimal effort
- Interpret regulatory guidelines and compliance documents
- Transform raw datasets into clear action plans, dashboards, or follow-up tasks
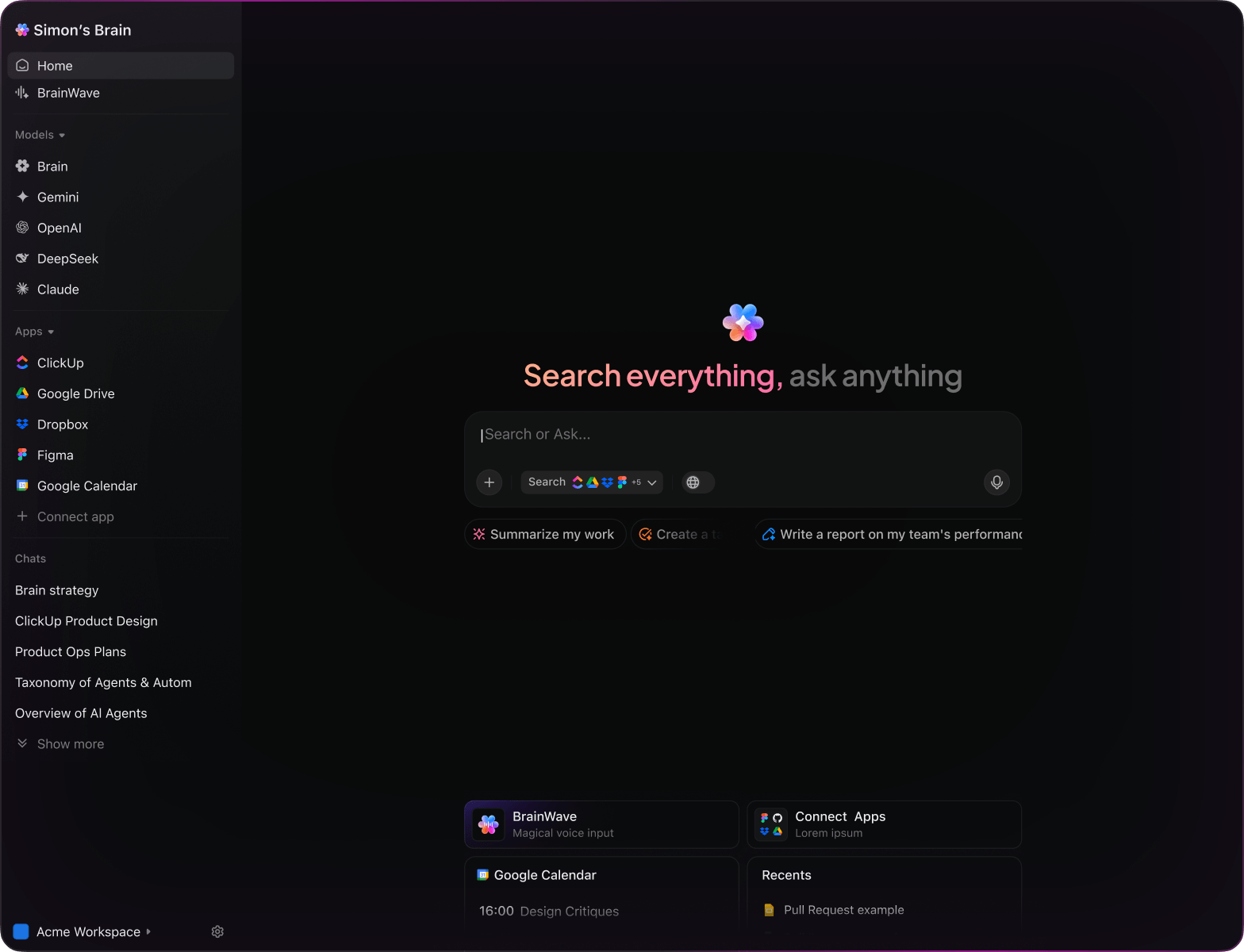
Integrated within familiar tools like documents, dashboards, and project trackers, AI in platforms such as ClickUp Brain acts as a powerful partner—turning scattered data into structured, impactful insights.
Why ClickUp Brain Stands Out
Standard AI Platforms
- Constantly switching apps to collect data context
- Repeating objectives with every query
- Responses lacking relevance to clinical specifics
- Hunting through multiple systems for trial files
- Interacting with AI that only processes input
- Manually toggling between different AI engines
- Merely a browser add-on without deep integration
ClickUp Brain
- Deeply connected to your clinical trial tasks, documents, and team communications
- Retains your project history and analysis goals
- Provides detailed, context-aware recommendations
- Consolidated search across all clinical data sources
- Supports voice commands with Talk to Text
- Automatically selects optimal AI model: GPT, Claude, Gemini
- Dedicated desktop app for Mac & Windows designed for performance
15 Essential AI Prompts for Clinical Trial Data Analysis
Identify 5 key patient demographic trends from the ‘Phase III Oncology Trial’ dataset.
ClickUp Brain Behavior: Analyzes linked trial data to highlight demographic patterns and participant characteristics.
What adverse event patterns are most frequent in cardiovascular studies under 2023 protocols?
ClickUp Brain Behavior: Integrates findings from internal safety reports; Brain Max can supplement with public pharmacovigilance data if accessible.
Draft a summary report for protocol deviations in the ‘Diabetes Control Study’ referencing monitoring logs and audit notes.
ClickUp Brain Behavior: Extracts relevant entries from linked documents to compile a structured deviation overview.
Compare efficacy endpoints between Drug A and Drug B using data from the ‘Immunotherapy Trials Q1’ folder.
ClickUp Brain Behavior: Pulls tabular and narrative data from internal sources to generate a concise comparative analysis.
List top biomarkers tracked in recent oncology trials, referencing lab reports and research summaries.
ClickUp Brain Behavior: Scans internal documents to identify frequently measured biomarkers and their clinical relevance.
From the ‘Data Quality Checks’ doc, create a checklist for common data validation steps in clinical trials.
ClickUp Brain Behavior: Recognizes validation criteria and formats them into a practical task list or document.
Summarize 3 emerging trends in patient-reported outcome measures from recent trial feedback and literature reviews.
ClickUp Brain Behavior: Extracts repeated themes and insights from linked research and survey documents.
From the ‘Site Monitoring Reports Q2’ folder, highlight key compliance issues affecting data integrity.
ClickUp Brain Behavior: Reviews monitoring notes to identify recurring compliance challenges and their impact.
Compose clear, patient-friendly consent form language based on guidelines in ‘EthicsBoard_2024.pdf’.
ClickUp Brain Behavior: Draws tone and style cues from the document to suggest accessible consent text variations.
Summarize upcoming changes in FDA 2025 data submission requirements and their implications for our trial databases.
ClickUp Brain Behavior: Reviews linked regulatory documents; Brain Max can incorporate public updates if available.
Generate guidelines for anonymizing patient data in compliance with HIPAA and GDPR, referencing internal policy docs.
ClickUp Brain Behavior: Extracts key rules and best practices to form a comprehensive compliance checklist.
Create a checklist for adverse event reporting timelines using FDA and EMA guidelines alongside our internal SOPs.
ClickUp Brain Behavior: Identifies critical deadlines and reporting steps from linked PDFs and workflow documents.
Compare data management approaches across recent oncology, cardiology, and neurology trials using internal audit reports.
ClickUp Brain Behavior: Summarizes documented practices into a clear, comparative format (summary/table).
What data visualization trends are emerging in clinical trial dashboards since 2023?
ClickUp Brain Behavior: Synthesizes insights from internal design notes, user feedback, and industry reports.
Summarize key data quality challenges reported by Southeast Asia trial sites, focusing on missing data and entry errors.
ClickUp Brain Behavior: Extracts and prioritizes issues from survey responses, monitoring notes, and flagged tickets.
AI Prompts for Clinical Trial Data Analysis with ClickUp Brain
ChatGPT Clinical Trial Prompts
- Summarize patient enrollment data into a concise 5-point report highlighting demographic trends.
- Draft patient communication templates emphasizing safety, compliance, and study benefits.
- Generate 3 alternative statistical analysis plans for efficacy endpoints and explain their strengths.
- Write a detailed workflow for integrating real-time adverse event monitoring in trial management.
- Compare last 3 trial site performances and summarize key operational challenges for improvement.
Gemini Clinical Trial Prompts
- Develop 3 alternative data visualization concepts for patient outcome metrics based on prior study feedback.
- List innovative patient engagement strategies focusing on retention and adherence.
- Produce a mood board description for a clinical trial dashboard highlighting clarity, accessibility, and data transparency.
- Suggest optimized scheduling layouts for multi-center trials and rank them by efficiency and patient convenience.
- Create a comparison table for three data management platforms focusing on compliance, usability, and integration.
Perplexity Clinical Trial Prompts
- List 5 emerging biomarkers relevant to oncology trials and rank them by validation status.
- Provide a comparison of electronic data capture systems, highlighting cost, usability, and regulatory compliance.
- Summarize global trends in decentralized clinical trials and their impact on patient recruitment.
- Generate a list of 5 innovative remote monitoring tools and rank them by data accuracy.
- Compare past trial protocols and summarize top 3 lessons for adaptive study designs.
ClickUp Brain Clinical Trial Prompts
- Transform this patient feedback log into prioritized action items for the clinical operations team.
- Summarize investigator meeting notes and create follow-up tasks with assigned roles and deadlines.
- Analyze annotated trial protocol documents and generate a checklist of compliance requirements for review.
- Develop a task list from this cross-department discussion on data quality improvements, including urgency levels.
- Summarize monitoring visit reports and produce actionable tasks for site coordinators and data managers in ClickUp.
Transform Raw Data Into Clear Insights
- Convert scattered trial notes into detailed analysis reports swiftly.
- Generate innovative hypotheses by reviewing previous study results.
- Build standardized templates to accelerate each trial phase.
Brain Max Boost: Quickly access historical trial data, participant feedback, and protocol documents to guide your next analysis.
Accelerate Clinical Trial Data Analysis
- Break down intricate trial data into manageable tasks.
- Transform raw datasets into actionable insights for your team.
- Automatically create detailed analysis summaries without extra effort.
Brain Max Boost: Instantly access historical trial results, protocol variations, or patient cohort details across studies.
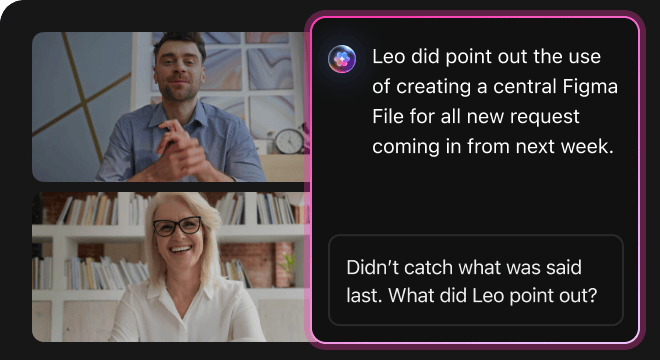
How AI Prompts Enhance Every Phase of Clinical Trial Data Analysis
Quickly Develop Robust Hypotheses
Researchers explore diverse data patterns rapidly, refine analytical approaches, and overcome data complexity challenges.